Clear Sky Science · en
The nerve center of organ engineering
Why wiring up organs matters
Scientists are getting closer to building replacement organs in the lab to tackle the severe shortage of donor hearts, livers, kidneys, and more. But most artificial organs focus on blood vessels and ignore another crucial ingredient: nerves. This review article explains why the body’s electrical wiring is essential for organ growth, everyday function, and healing, and how researchers are learning to weave nerve networks into lab-grown organs.
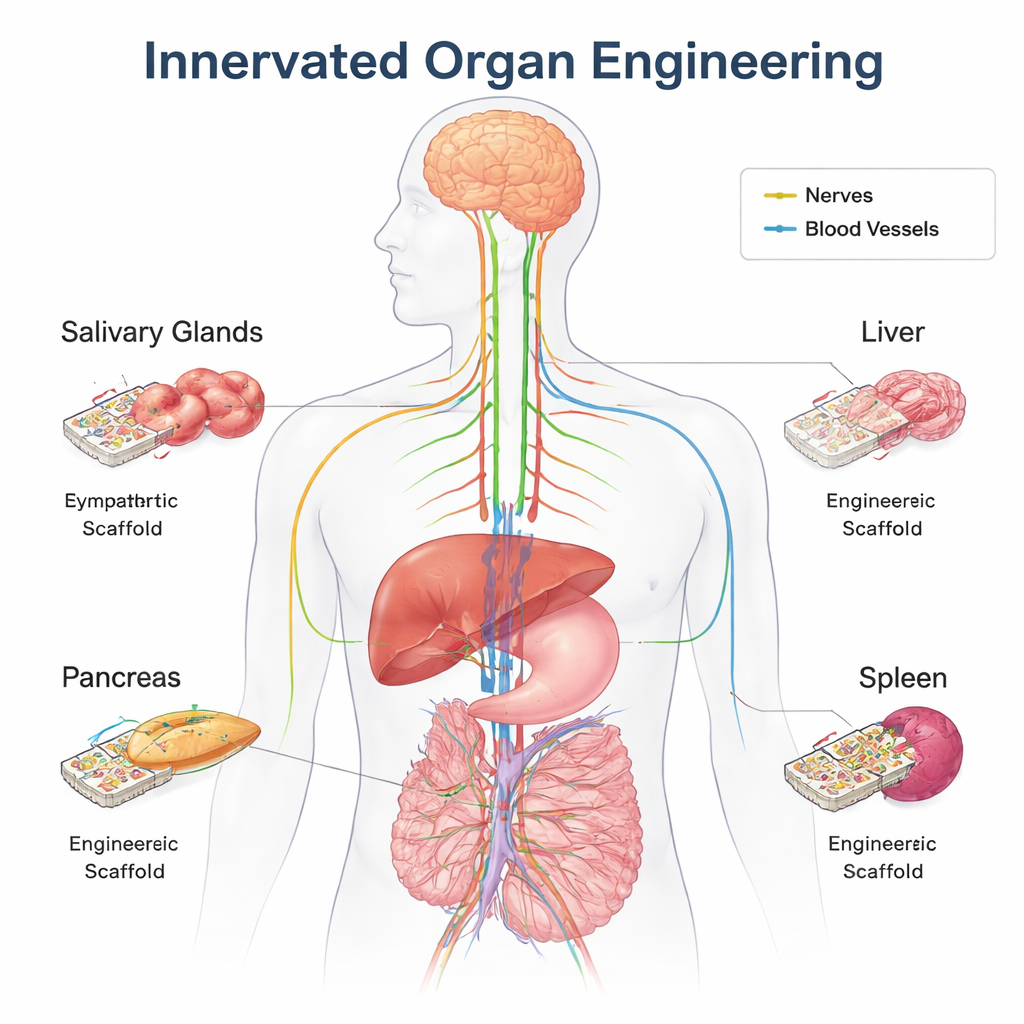
The body’s hidden control network
Every major organ is threaded with nerve fibers that constantly talk to the brain and spinal cord. Through the autonomic nervous system—the “fight-or-flight” and “rest-and-digest” branches—these nerves quietly regulate blood sugar, digestion, immune responses, blood flow, and more. They do this by sending chemical messages such as acetylcholine and norepinephrine, and by sensing changes like stretch, temperature, or nutrient levels. Beyond day-to-day control, nerves also help shape organs before birth, guide wound healing, and support stem cells that repair tissues. In many places, nerves grow alongside blood vessels, using similar guidance cues, so wiring and plumbing develop together.
Why transplants cope without nerves—but lab organs cannot
Traditional organ transplants are usually implanted without their original nerves. A transplanted liver or kidney can still work because it receives hormones and other signals through the bloodstream, and over time new nerve fibers can grow in from the recipient. That “grace period” does not exist for engineered organs built from scratch. These constructs typically lack the full mix of mature cell types and the complex supporting matrix found in a natural organ. Precise nerve connections are especially important for coordinating the many specialized cells inside organs like the pancreas, liver, salivary glands, and spleen. The authors argue that for bottom-up organ engineering—where organs are assembled from small building blocks—preplanned innervation should be treated as a design requirement, not an optional add-on.
How nerves shape four key organs
The review explores in detail how nerves influence four example organs. In the pancreas, sympathetic and parasympathetic fibers help sculpt the architecture of the insulin-producing islets during development and later fine-tune insulin and glucagon release as blood sugar rises and falls. In the salivary glands, early parasympathetic input keeps epithelial stem cells alive and guides the branching ducts that ultimately secrete saliva; cutting these nerves at the wrong time disrupts normal gland formation. The liver’s nerves sense salt, water, sugar, and fat levels in the blood and adjust metabolism, blood pressure, and regeneration after injury. In the spleen, sympathetic fibers talk directly to immune cells, dampening or boosting inflammation and helping the body respond to infection and stress. Across these organs, losing or miswiring nerves is linked to diabetes, digestive problems, liver disease, and impaired immunity.
Building organs with built-in wiring
To bring innervation into bioengineered organs, scientists are combining advanced 3D printing, clever scaffold design, and stem-cell–derived nerve cells. Extrusion and light-based bioprinting can place different cell types and materials in precise patterns, while specialized hydrogels and polymers are tuned to support long, branching nerve fibers. Studies have already created printed neural networks, nerve–muscle units, and miniature brain-like tissues that show realistic electrical activity. The authors outline practical blueprints for innervated versions of the pancreas, salivary gland, liver, and spleen: isolate or derive the key organ cells, mix them with autonomic neurons (sympathetic and parasympathetic) in compatible bioinks, print them into organ-shaped structures, and mature them in bioreactors so nerves can extend into the tissue before implantation.
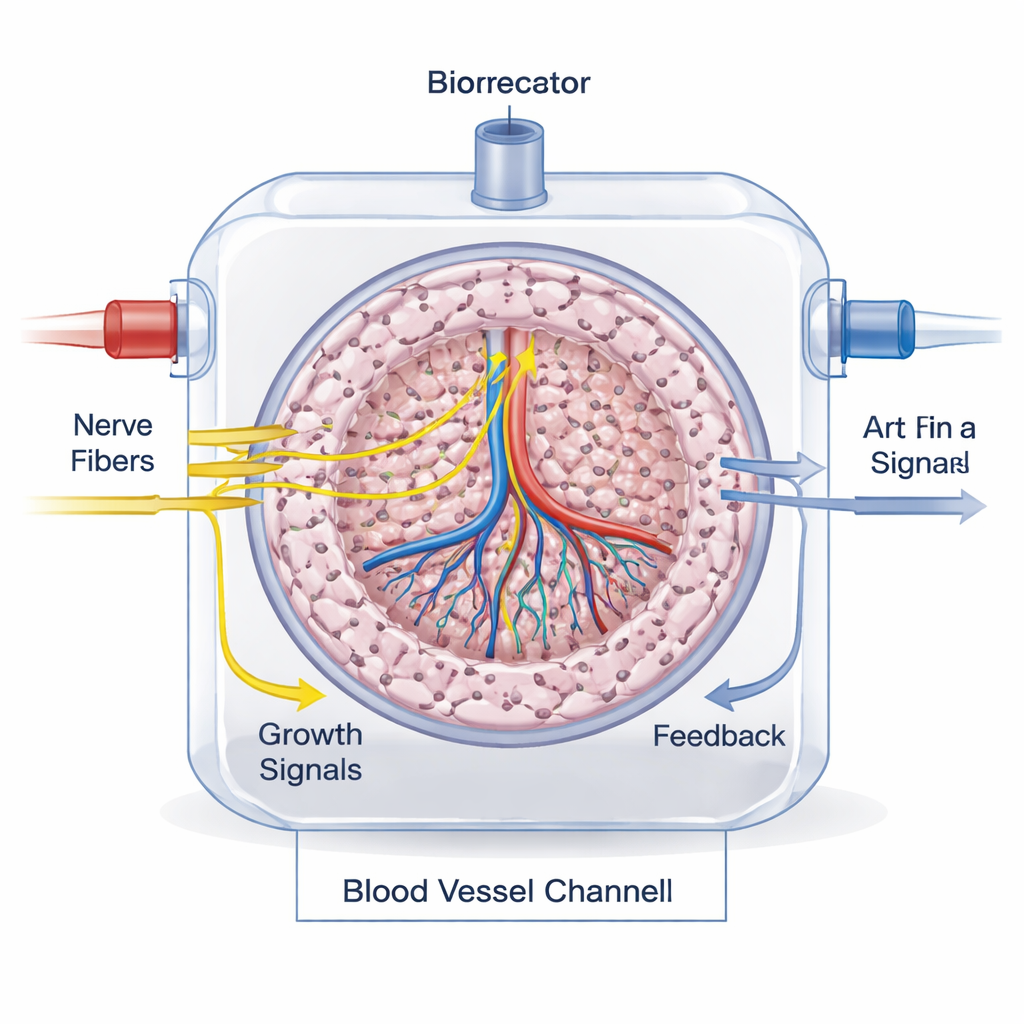
Measuring whether the wiring works
Checking that nerves are truly integrated and functional is a major challenge. In the lab, researchers stain for nerve-specific proteins, measure neurotransmitters with biochemical tests, and use live imaging to watch axons grow and cells respond. Microfluidic “organ-on-a-chip” platforms help model realistic blood flow and nerve–organ signaling. In animals and, eventually, in patients, innervation can be tracked by combining tissue staining, nerve tracers, and modern imaging methods such as MRI, CT, and PET scans with specialized radioactive compounds that highlight sympathetic or parasympathetic fibers.
From idea to therapy
The article concludes that nerves are not a luxury feature but a core requirement for truly functional artificial organs. To move from promising lab demos to real therapies, future work must improve large-scale bioprinting of intricate nerve networks, refine cell sources (especially human stem-cell–derived autonomic neurons), and develop practical clinical imaging tools to monitor neural integration after implantation. If these hurdles are overcome, next-generation engineered organs could not only replace lost tissue but also plug seamlessly into the body’s own control circuits, offering more reliable and lifelike treatments for diabetes, liver failure, dry mouth disorders, immune deficiencies, and beyond.
Citation: Das, S., Gordián-Vélez, W.J., Dave, J.R. et al. The nerve center of organ engineering. Nat Commun 16, 9834 (2025). https://doi.org/10.1038/s41467-025-64801-4
Keywords: organ engineering, innervation, biofabrication, 3D bioprinting, autonomic nervous system