Clear Sky Science · en
Nanozymes expanding the boundaries of biocatalysis
Small helpers that speed up chemistry
From brewing beer to digesting dinner, our lives rely on invisible helpers called catalysts that make chemical reactions go faster. For more than a century, biology’s catalysts were thought to be only fragile proteins and, later, catalytic RNA. This review introduces “nanozymes” – tiny engineered particles that behave like enzymes but are built from robust materials such as metals, oxides, and carbon. They can survive heat, cold, and harsh conditions, and are beginning to reshape how we think about chemistry in living systems, disease, industry, and even the origin of life.
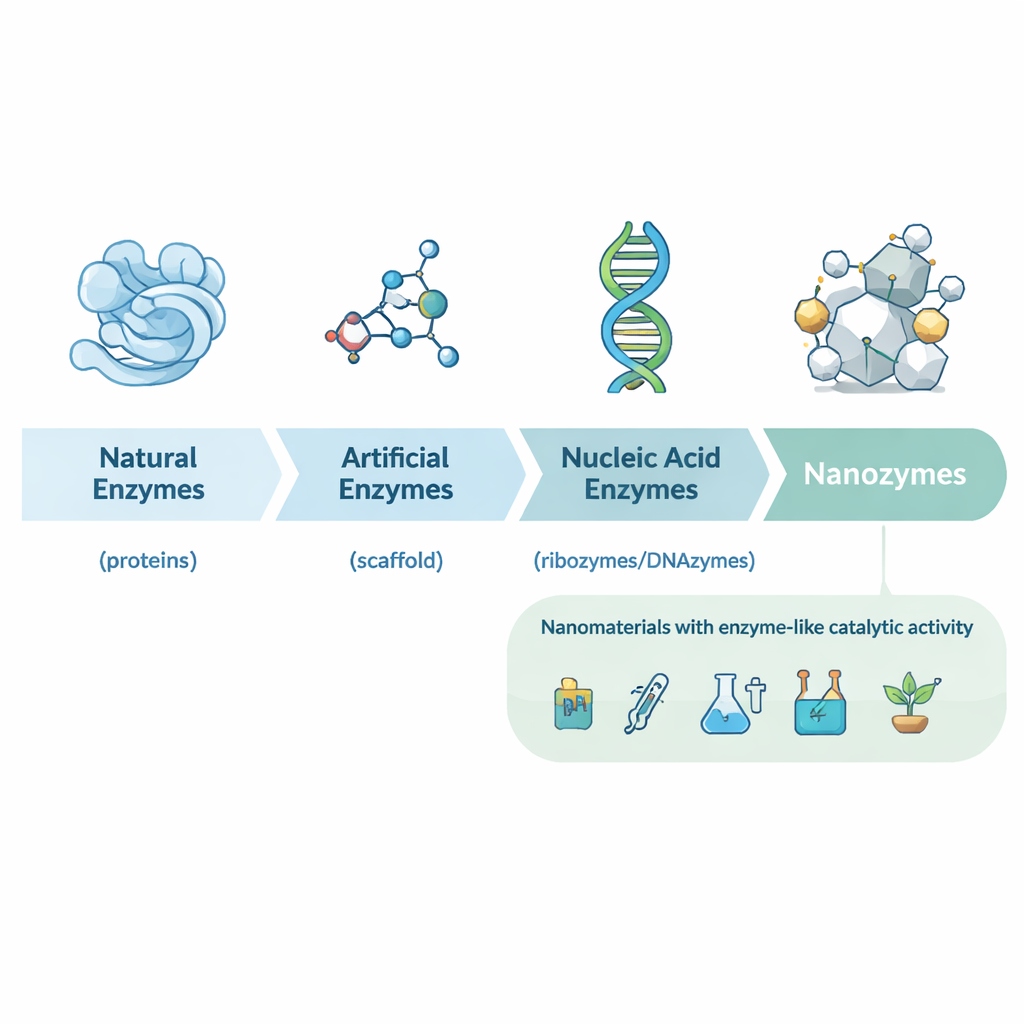
From natural enzymes to designer nanocatalysts
The article first walks through the history of biocatalysis, from early discoveries of protein enzymes in yeast and stomach juices to ribozymes (catalytic RNA) and a wide range of artificial enzymes built from small molecules and antibodies. These traditional catalysts are powerful but often expensive, unstable, and hard to mass‑produce. In the early 2000s, researchers found that some nanomaterials – for example, iron oxide nanoparticles – could mimic the behavior of classic enzymes such as horseradish peroxidase. This led to the concept of nanozymes: nanometer‑scale particles whose own structure gives them enzyme‑like activity, rather than attached protein or small‑molecule catalysts.
What makes nanozymes different
Unlike enzymes, which have a single, precisely shaped active pocket, nanozymes expose many active spots across their surfaces. These spots can sit at crystal edges, defects, or interfaces between two materials, and collectively they can turn over many molecules at once. A single active site on a nanozyme is usually less efficient than an enzyme’s, but one nanoparticle can host thousands of such sites, so its overall catalytic power can rival or exceed that of natural enzymes. Their activity can also be tuned by changing size, shape, chemical doping, and surface coatings – much like adjusting the architecture and wiring of a tiny machine. Because they are made from robust solids rather than delicate folded proteins, nanozymes keep working at high temperatures, low temperatures, or high salt levels that would quickly disable most enzymes.
How nanozymes work and how they are measured
The authors show that nanozymes often follow the same basic kinetic rules that biochemists use for enzymes, such as Michaelis–Menten behavior, where reaction speed depends on how easily a catalyst binds and converts its substrate. But there are important twists. One particle can host multiple types of active sites, so a single nanozyme may carry out different reactions or even conflicting ones, like both making and destroying reactive oxygen species. This can produce complex “non‑ textbook” kinetic curves that hide multiple processes under one smooth trend. The review explains how researchers dissect these behaviors, estimate how many active sites really exist, and compare the overall activity of different nanozymes using standardized units, all of which are crucial for turning an intriguing material into a reliable tool.
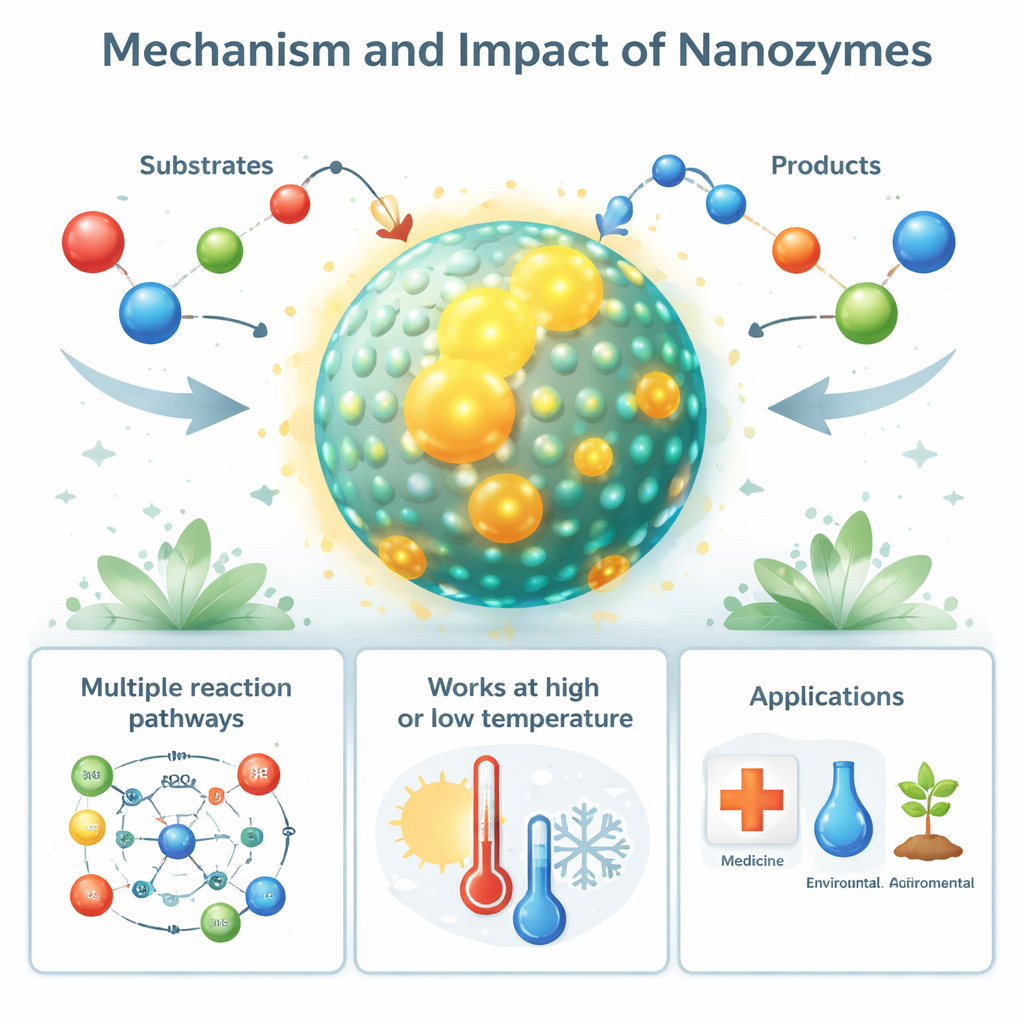
From natural nanozymes to real‑world uses
Intriguingly, nanozyme‑like behavior is not limited to the lab. Certain biological nanostructures, such as iron‑filled protein cages (ferritin) and magnetic particles in bacteria (magnetosomes), act as natural nanozymes that help control harmful reactive oxygen species. Even disease‑related protein fibers, like amyloid deposits in Alzheimer’s disease, can adopt nanozyme‑like peroxidase activity that damages nearby cells. On the applied side, synthetic nanozymes are being used as cheaper, tougher stand‑ins for enzymes in diagnostic tests, biosensors, and industrial assays. Their ability to either generate or remove reactive oxygen species is being harnessed to kill tumor cells and bacteria, protect tissues from oxidative stress, and help crops withstand drought, salinity, and pollution.
Designing the next generation of smart catalysts
Looking ahead, the authors highlight key challenges: pinpointing the exact atomic structures that act as active sites, improving reaction selectivity so that nanozymes act only where and when they are wanted, and ensuring safety and stability inside the body. They point to advanced imaging, quantum‑level simulations, and machine learning as powerful tools for predicting and optimizing nanozyme performance. Because nanozymes can work in extreme conditions, respond to light, heat, sound, or magnetic fields, and sometimes perform several reactions in sequence, they could underpin new therapies, environmental technologies, and industrial processes. The review concludes that nanozymes broaden the very definition of biocatalysis and may even offer clues to how primitive catalysts once powered the chemistry of early life.
Citation: Zhang, R., Yan, X., Gao, L. et al. Nanozymes expanding the boundaries of biocatalysis. Nat Commun 16, 6817 (2025). https://doi.org/10.1038/s41467-025-62063-8
Keywords: nanozymes, biocatalysis, nanoparticles, enzyme mimics, reactive oxygen species