Clear Sky Science · en
Performance benchmarking and analysis of lithium-sulfur batteries for next-generation cell design
Why new batteries matter
The batteries that power our phones, laptops, and electric cars are reaching their limits. To go farther on a single charge and store more renewable energy, researchers are turning to lithium–sulfur batteries, a chemistry that could pack far more energy than today’s lithium‑ion cells while using cheaper, more abundant materials. But thousands of laboratory experiments report performance in different, often incompatible ways. This article pulls those scattered results together, creating a common yardstick to see what really works—and what holds lithium–sulfur batteries back.
Building a better sulfur battery
Lithium–sulfur batteries replace the heavy metal‑oxide cathode in standard lithium‑ion cells with elemental sulfur, paired with a lithium metal anode and a liquid organic electrolyte. On paper, this simple swap could more than double the energy stored per kilogram. In practice, sulfur brings headaches: it and its discharge products conduct electricity poorly; intermediate “polysulfide” molecules dissolve into the electrolyte and wander through the cell, wasting active material and corroding the lithium anode; and the sulfur electrode swells and shrinks as the battery charges and discharges. To tame these effects, many researchers embed sulfur in an engineered “host” material that can conduct electrons, trap polysulfides, and provide room for the material to expand and contract.
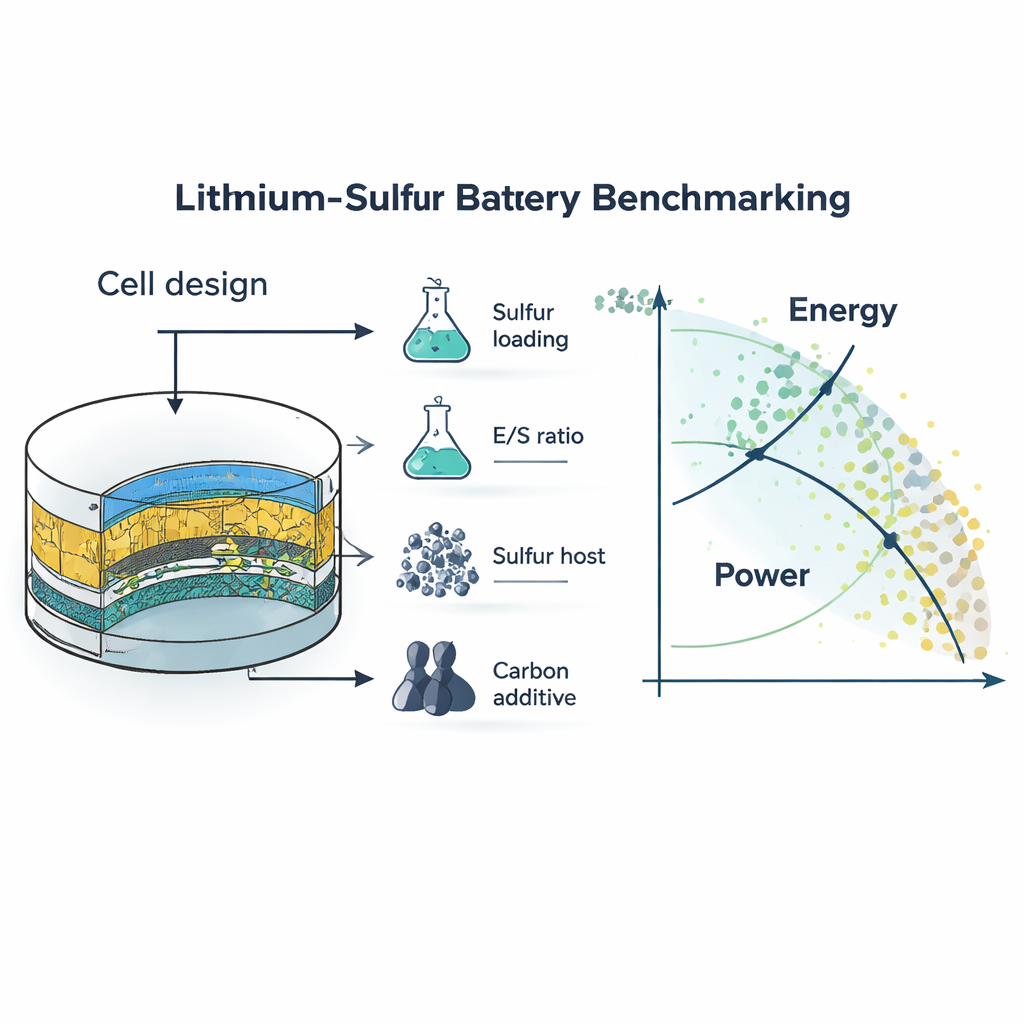
Turning scattered studies into a common map
The authors reviewed 184 recent papers, digitally extracting data from 866 battery test plots. For each cell they reconstructed key design choices—how much sulfur was loaded into the electrode, how much electrolyte was used per unit sulfur (the E/S ratio), how much carbon additive was present, and what type of host structure and surface area were employed. They then converted all results into cell‑level specific energy (watt‑hours per kilogram) and specific power (watts per kilogram), approximating how a real device, not just a single electrode, would perform. This data‑driven approach produces a “map” of the field, showing which combinations of ingredients and design parameters truly push performance forward.
Finding the sweet spot in cell design
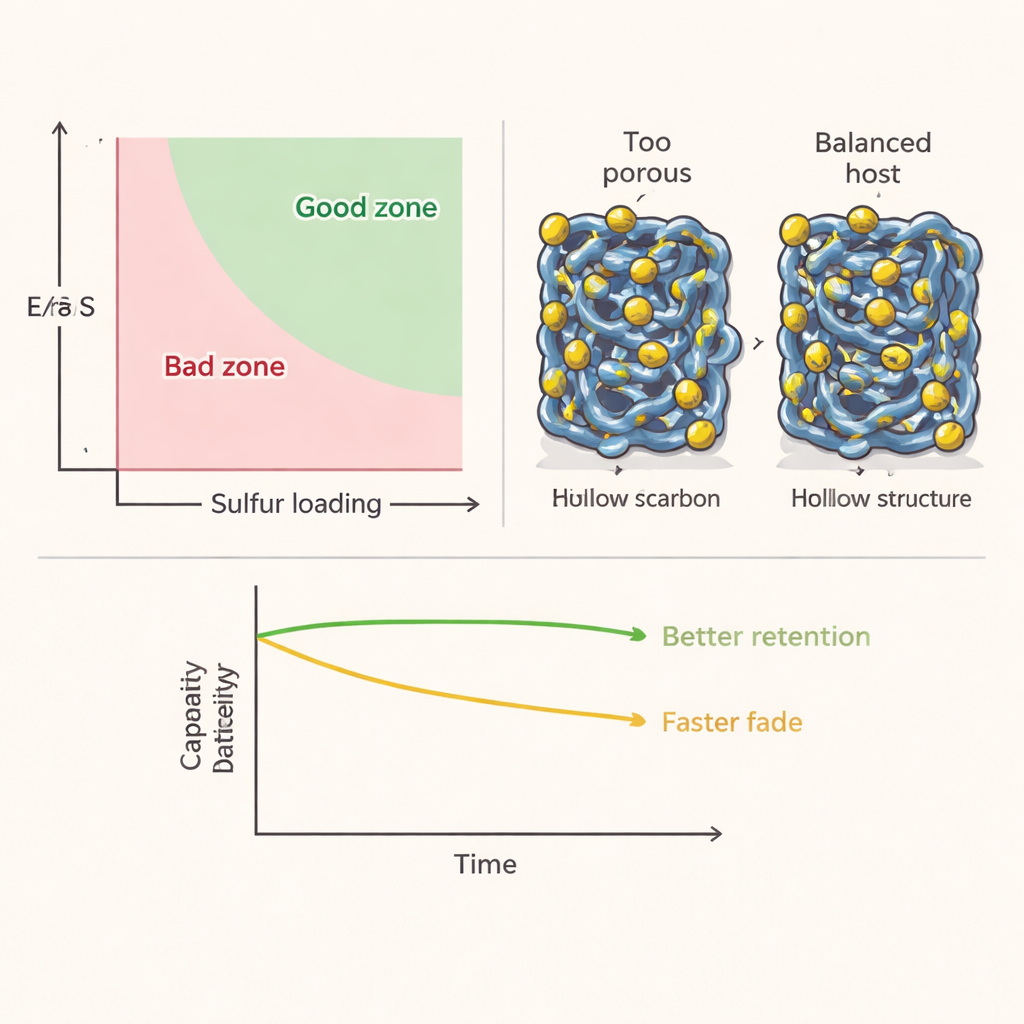
One of the clearest lessons concerns the balance between sulfur loading and the electrolyte‑to‑sulfur ratio. Thicker sulfur electrodes and less electrolyte should, in theory, boost energy per kilogram by cutting dead weight. The database shows a more nuanced reality: when sulfur loading rises much above roughly 6 milligrams per square centimeter, transport of ions and electrons through the electrode becomes sluggish and usable capacity plunges. By contrast, carefully reducing the E/S ratio has a strong, favorable correlation with higher specific energy, yet only a modest impact on how well the battery keeps its capacity over many cycles. In other words, trimming excess electrolyte is usually more beneficial than simply cramming in more sulfur, and there is a practical optimum where energy, stability, and power can all be balanced.
What makes a sulfur host truly helpful
The review also dissects the properties of the sulfur host materials themselves. Porous carbons, metal‑organic‑framework‑derived scaffolds, hollow particles, flat two‑dimensional sheets, and complex three‑dimensional assemblies were all compared by surface area and their tendency to bind polysulfides. Surprisingly, the highest surface areas did not give the best batteries: extremely fine pores and tortuous pathways hinder ion movement, soak up too much electrolyte, and can trap sulfur where it cannot be fully used. The best results tended to cluster around moderate surface areas and moderate binding strengths—strong enough to hold polysulfides near the reaction sites, but not so strong that they become immobilized. Hollow and two‑dimensional host structures often struck this balance, combining accessible space for sulfur and lithium transport with sufficient anchoring sites.
Speed, lifetime, and real‑world prospects
By comparing rate‑capability tests, the authors show that well‑designed lithium–sulfur cells can deliver respectable power: in typical “standard” cells with modest sulfur loading and ample electrolyte, most of the theoretical capacity is still available at one to two times the current used for slow tests. However, when sulfur loading is pushed higher and electrolyte volumes are cut—conditions needed for practical, high‑energy packs—maintaining both power and long life becomes much harder, especially in larger pouch cells. High carbon contents, often used to improve electronic conductivity, can actually worsen ion transport and hurt performance under lean‑electrolyte conditions. The analysis highlights that relatively low carbon fractions, carefully optimized sulfur‑to‑host ratios, and improved lithium‑metal anodes are key to sustaining capacity over many fast charge–discharge cycles.
What this means for future batteries
Taken together, the curated data show that lithium–sulfur cells built with advanced sulfur hosts are already surpassing today’s commercial lithium‑ion batteries in energy per kilogram, with some lab‑scale designs reaching around 440 watt‑hours per kilogram and pointing toward the long‑sought 500 watt‑hour mark. The study makes clear that there is no single magic material; instead, success depends on hitting the right combination of sulfur loading, electrolyte volume, host structure, and carbon content, all while protecting the lithium metal anode. By providing quantitative benchmarks and exposing which design choices pay off—or backfire—this work offers a practical roadmap for turning lithium–sulfur batteries from promising laboratory curiosities into reliable power sources for electric vehicles, aircraft, and grid storage.
Citation: Yari, S., Conde Reis, A., Pang, Q. et al. Performance benchmarking and analysis of lithium-sulfur batteries for next-generation cell design. Nat Commun 16, 5473 (2025). https://doi.org/10.1038/s41467-025-60528-4
Keywords: lithium-sulfur batteries, energy storage, sulfur host materials, battery design, electrolyte-to-sulfur ratio