Clear Sky Science · en
Nanopore sensing of protein and peptide conformation for point-of-care applications
Why tiny protein shapes could change your next checkup
As populations age, diseases such as Alzheimer’s, Parkinson’s, heart disease, and cancer are becoming more common. Many of these illnesses begin when vital proteins and small protein fragments (peptides) subtly change their shape long before symptoms appear. Today’s clinical tests usually measure how much of a biomarker is present, not whether its shape is normal or distorted. This article explores a technology called nanopore sensing, which can electrically “feel” the shape and chemistry of single protein molecules, and argues that it could become the basis of future point-of-care tests that run in minutes on a small device.
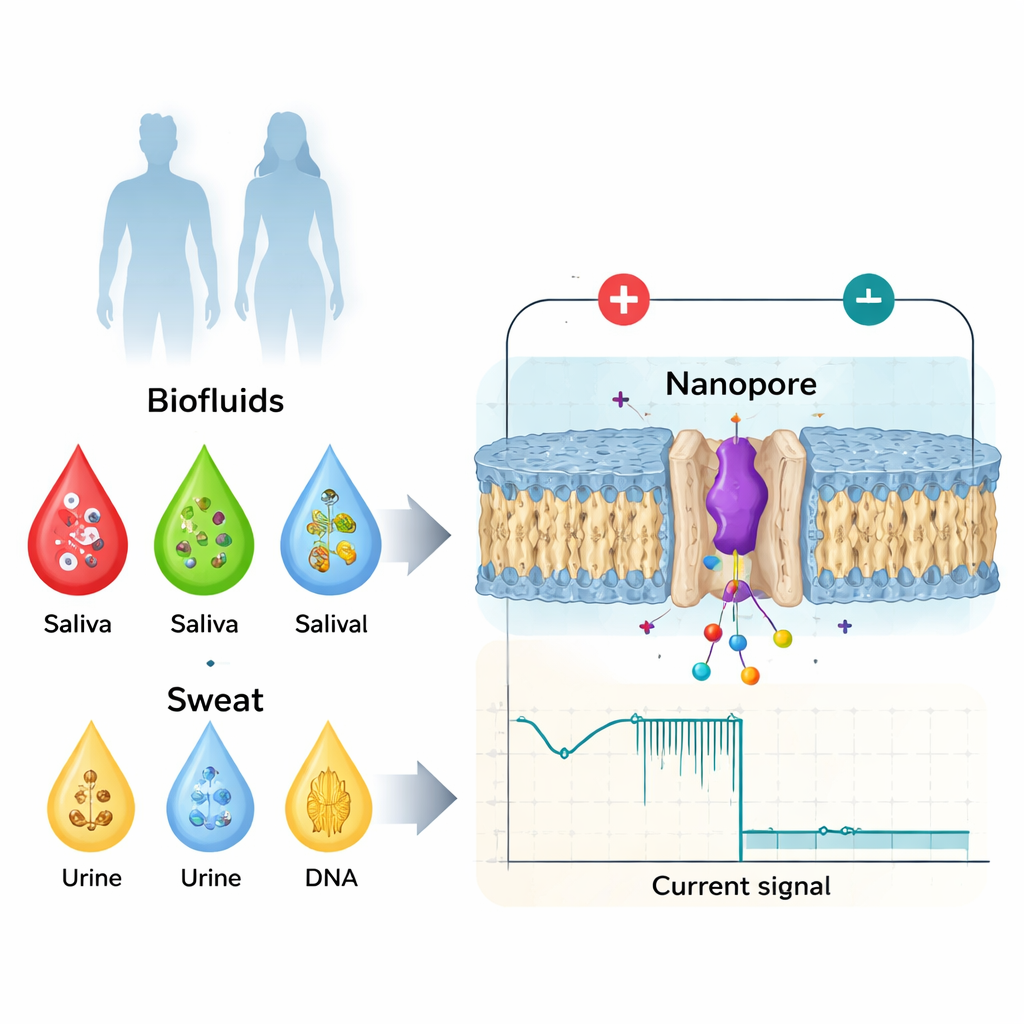
From simple blood tests to shape-aware biomarkers
Doctors already use many biomarkers taken from blood, saliva, urine, or sweat—such as proteins, hormones, or small molecules—to track health and disease. Traditionally, the focus has been on gross changes: extra copies of a protein, damaged DNA, or the presence of a viral component. But biology turns out to be more subtle. The same protein can be switched on or off by tiny changes in its structure, by chemical tags added after it is made, or by mirror-image versions of its building blocks. These small changes can alter how a protein binds to partners, forms clumps, or carries signals, and are linked to blood clotting problems, neurodegenerative diseases, and cancer. Standard hospital tools like mass spectrometry, antibody-based tests, and high-resolution imaging are powerful, but they are expensive, slow, require highly trained staff, and generally cannot be deployed as fast, simple point-of-care devices.
What a nanopore does differently
Nanopore sensing flips the problem on its head. Instead of averaging over trillions of molecules, it examines one at a time as it passes through a tiny hole in a membrane that is just a few billionths of a meter wide. A voltage is applied so ions flow through the pore and create a steady electrical current. When a protein or peptide enters the pore, it partially blocks that current. The depth of the current drop, how long it lasts, and the fine details of its shape depend on the molecule’s size, charge, and conformation (its three-dimensional fold). By carefully designing the pore—using either engineered proteins or solid-state materials—researchers can confine individual biomolecules long enough to create rich electrical “fingerprints” that distinguish not only different proteins, but also subtle variants of the same biomarker.
Reading disease-relevant changes, one molecule at a time
The review highlights how nanopores have already been used to resolve medically important differences that other methods struggle to see. They can tell apart peptides that differ by a single amino acid, detect disease-linked variants of hemoglobin directly from blood, and distinguish short hormone-like peptides that vary by only one building block or even by a mirror-image form of that building block. Nanopores can also sense post-translational modifications—small chemical tags such as phosphate, sugar, or sulfate groups—that help control whether proteins involved in Alzheimer’s, Parkinson’s, blood clotting, or cancer behave normally or pathologically. In some experiments, a single enzyme or binding protein is held inside the pore, and changes in its electrical signal reveal, in real time, how it binds partners or carries out reactions, potentially exposing malfunctioning pathways in disease.
Toward fast, bedside testing
Because each blockage event corresponds to one molecule, nanopore devices can be extremely sensitive, detecting as few as thousands of copies of a biomarker in a complex fluid. The authors discuss strategies to overcome key hurdles for clinical use: increasing the rate at which rare molecules are captured, stabilizing membranes or using hybrid solid–biological pores, and using machine learning to automatically classify complex electrical patterns into clear diagnostic categories. They also show how indirect approaches—such as attaching DNA tags or binding partners—can amplify weak signals or allow simultaneous measurement of multiple biomarkers, while still preserving much of the conformational detail that makes nanopores unique.
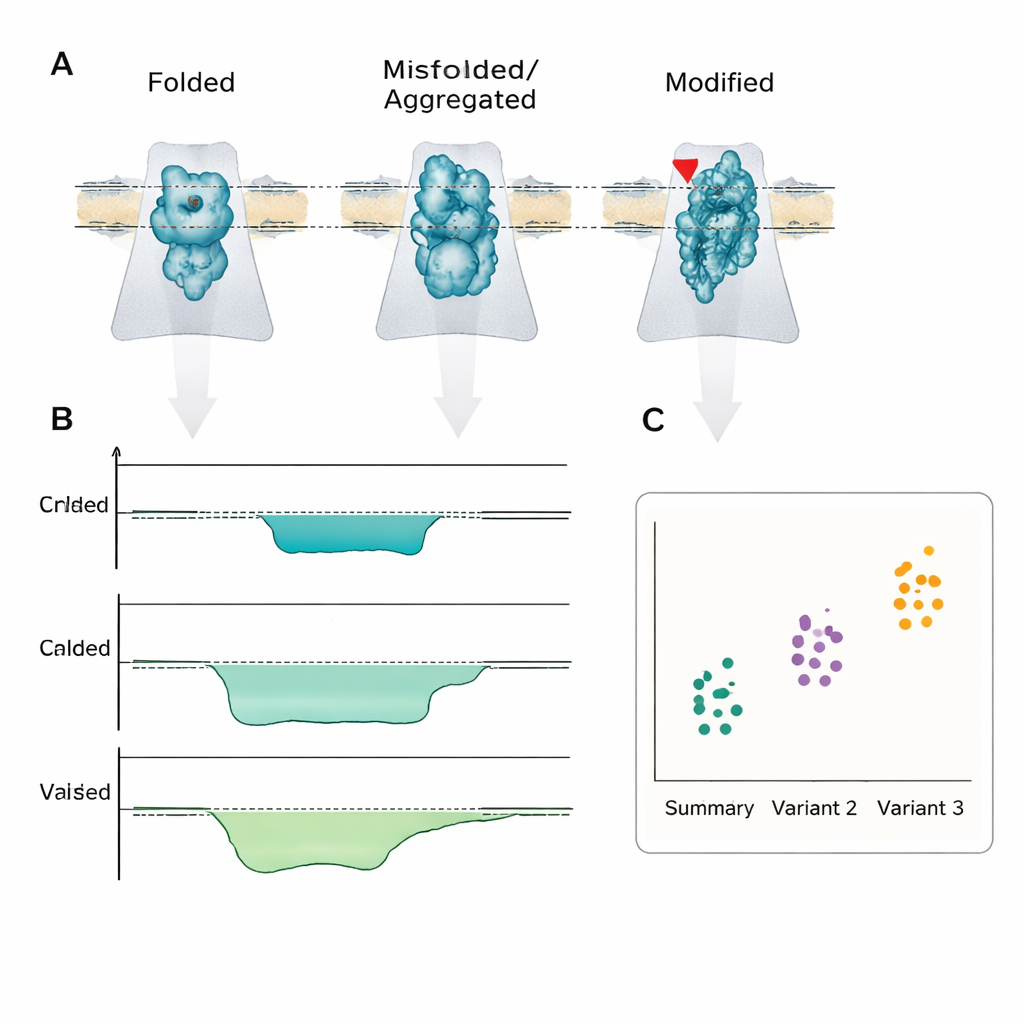
What this could mean for patients
The central message is that disease is often driven less by how much protein exists and more by which shape or chemical version is present. Nanopore sensing is one of the few technologies that can directly read these differences at the single-molecule level, fast enough and simply enough to be integrated into portable devices. Although significant engineering and standardization challenges remain, the authors argue that, much like nanopore DNA sequencers have moved into the clinic, nanopore protein and peptide sensors could eventually provide rapid, bedside tests that not only say “something is wrong” but also reveal the exact molecular misfolding or modification behind a patient’s condition.
Citation: Ratinho, L., Meyer, N., Greive, S. et al. Nanopore sensing of protein and peptide conformation for point-of-care applications. Nat Commun 16, 3211 (2025). https://doi.org/10.1038/s41467-025-58509-8
Keywords: nanopore sensing, protein biomarkers, point-of-care diagnostics, post-translational modifications, conformational diseases