Clear Sky Science · en
The Estonian Biobank’s journey from biobanking to personalized medicine
Why this matters for your health
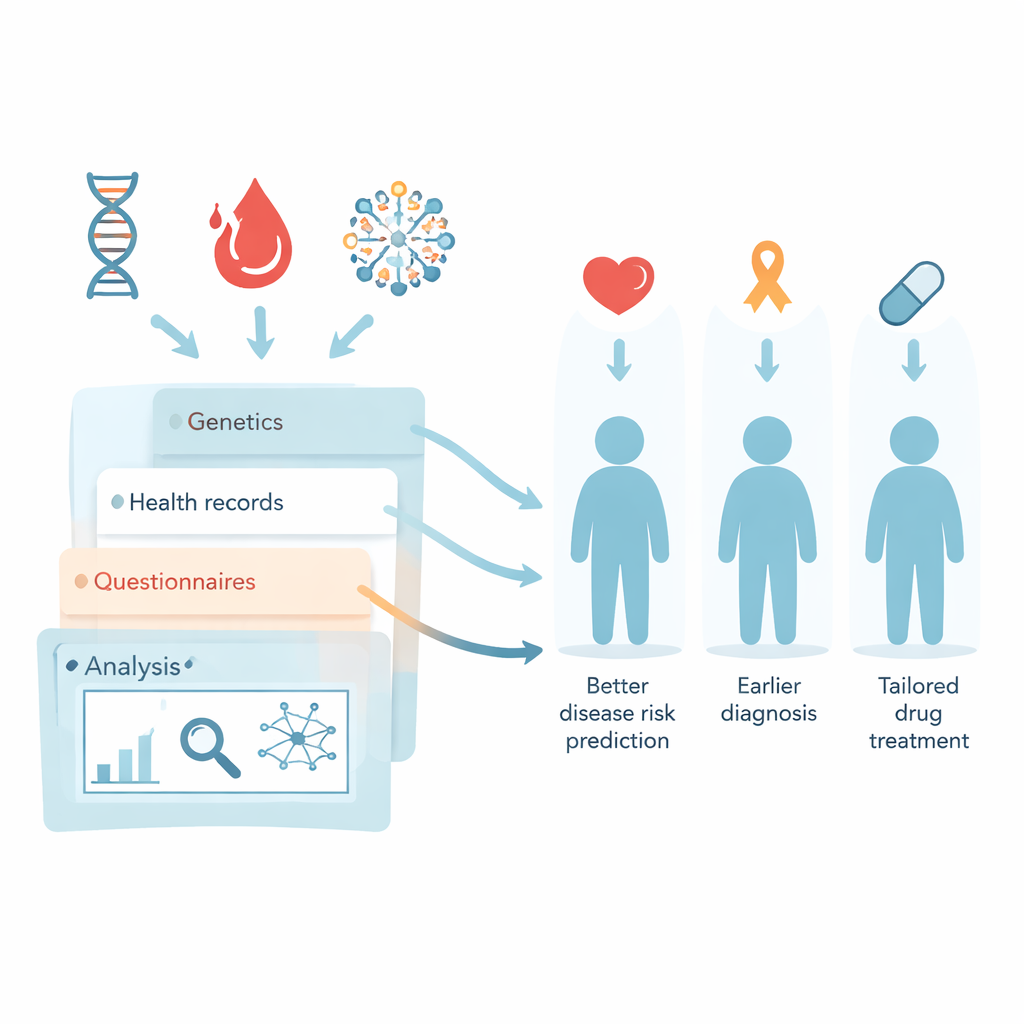
The Estonian Biobank is a national effort in which about one in five Estonian adults has donated blood and allowed their medical data to be used for research. This review article explains how, over 25 years, that resource has grown from frozen tubes in freezers into a powerful engine for personalized medicine—using your genes, health records, and lifestyle to predict disease risk, choose safer drugs, and design prevention strategies tailored to each person.
Building a nationwide health resource
The biobank began by recruiting tens of thousands of volunteers through family doctors and special clinics. Participants signed broad consent forms, gave blood, and answered hundreds of questions about their health, habits, and backgrounds. Crucially, their data are regularly linked to Estonia’s highly digital health system, which includes insurance claims, hospital records, prescriptions, and even causes of death. Over time, this has created rich, lifelong health profiles for 212,000 participants—about 20% of Estonia’s adult population—spanning everything from common infections and back pain to cancer, heart disease, and mental health conditions.
From blood samples to deep biological insights
Beyond basic DNA testing, the biobank has layered on many types of biological measurements. High‑coverage genome sequencing in thousands of people helped build a reference for the Estonian population, revealing rare genetic variants not seen elsewhere and improving the accuracy of cheaper DNA chip tests used on everyone. Other projects have measured hundreds of blood metabolites, proteins, immune markers, and even the bacteria living in the gut. These data have been used to find biomarkers that predict five‑year risk of death or heart disease, uncover long‑term damage to the gut microbiome after repeated antibiotic use, and identify new genetic links to thyroid function, autoimmune disease, and women’s reproductive disorders.
Making sense of everyday health data
Because nearly all prescriptions and doctors’ notes in Estonia are digital, researchers can study how real people move through the health system. The biobank team has transformed messy electronic health records and free‑text notes into standardized, research‑ready formats. This lets them track patterns such as which drugs are most commonly used, how one diagnosis tends to lead to another, and which side effects are missed in routine coding but hidden in doctors’ narratives. Powerful algorithms and newer artificial intelligence methods are being trained on this combined genetic and clinical information to detect early warning signs of diseases like stroke, to follow weight changes across years, and to map complex chains of illnesses and treatments.
Calling people back based on their genes
One of the most distinctive features of the Estonian Biobank is its “recall by genotype” strategy: re‑contacting participants who carry important genetic variants. People with inherited high cholesterol, Wilson’s disease, or BRCA1/2 mutations linked to breast and ovarian cancer have been invited for follow‑up testing, counselling, and tailored care. Similar projects use polygenic risk scores—summing the tiny effects of many DNA changes—to flag those at particularly high risk of heart disease or breast cancer. Trials show that when people and their family doctors receive clear genetic risk information, they are more likely to start preventive treatments such as cholesterol‑lowering drugs, and many serious conditions are caught earlier than they would have been under standard screening rules.
From research results to everyday tools
To bring these ideas to everyday life, the biobank has launched an online MyGenome Portal. Participants who log in with Estonia’s secure electronic ID can view their genetic risk for common conditions like type 2 diabetes and coronary artery disease, explore how changing weight or quitting smoking might shift those risks, and see guidance on how their body may handle certain medicines. Fun features such as ancestry breakdowns and caffeine metabolism sit alongside more serious reports. Studies of how people react to receiving such information suggest that most welcome it, even when it reveals higher risk, and that clear counselling and follow‑up options are key to avoiding unnecessary worry.
What this means for the future of care
Overall, the article shows how a mid‑sized country can turn a volunteer biobank into a test bed for personalized medicine at national scale. Estonia’s combination of dense genetic data, detailed health records, and the ability to re‑contact participants has already uncovered hidden high‑risk patients, refined disease prediction tools, and supported smarter use of medicines. As new layers such as long‑read genome sequencing and environmental data are added, the Estonian Biobank is poised to deepen our understanding of how genes, lifestyle, and surroundings interact—and to help move healthcare from one‑size‑fits‑all treatment toward prevention and care tailored to each person.
Citation: Milani, L., Alver, M., Laur, S. et al. The Estonian Biobank’s journey from biobanking to personalized medicine. Nat Commun 16, 3270 (2025). https://doi.org/10.1038/s41467-025-58465-3
Keywords: biobanks, personalized medicine, genetics, Estonian Biobank, health data