Clear Sky Science · en
Endogenous opioid receptors and the feast or famine of maladaptive feeding
Why our brains can push us to overeat or hardly eat at all
Most people have felt both strong cravings for rich foods and, at other times, a loss of appetite under stress. This article explains how the brain’s own opioid system—the same family of chemicals involved in pain relief and drug addiction—can push eating habits to dangerous extremes. By looking at both obesity and anorexia nervosa, the authors show how similar brain circuits and chemical signals can produce opposite outcomes: chronic overeating or severe self-starvation.

The brain’s pleasure and pain chemicals
The body naturally produces opioid-like molecules such as endorphins, enkephalins, and dynorphins. These chemicals act on opioid receptors spread across the brain, especially in areas that control reward, motivation, hunger, and pain. When we eat, particularly tasty, high-calorie foods, these systems respond with signals that can make food feel rewarding and ease discomfort. Under normal conditions, this helps us seek enough food without going to extremes. But when these receptors are overactive, underactive, or wired differently, they can distort how rewarding food feels, how hungry we think we are, and how much pain or stress we notice.
How natural opioids can fuel overeating and obesity
In obesity, the brain’s reward circuits appear to be reshaped in ways that resemble changes seen with addictive drugs. Opioid receptors in a key reward hub called the nucleus accumbens help drive the urge to seek and enjoy palatable foods rich in fat and sugar. Genetic studies suggest that certain variants in the mu-opioid receptor gene may protect some people from gaining weight, while other patterns of gene regulation are linked to obesity. Experiments in rodents show that blocking opioid receptors reduces the appeal of sweet, high-fat diets, while removing these receptors can make animals resistant to diet-induced obesity. At the same time, people with obesity often show lower availability of mu-opioid receptors in the reward system but higher levels in hypothalamic regions that sense hunger and satiety, hinting at a long-term shift in how the brain values and regulates food.
When the same system supports self-starvation
Anorexia nervosa, though outwardly the opposite of obesity, also involves altered opioid signaling. Genetic studies repeatedly point to the delta-opioid receptor gene as a risk factor, and brain imaging reveals overall reduced opioid receptor availability in regions that process reward, fear, and aversion. A long-standing theory proposes that starvation in susceptible individuals triggers a self-reinforcing “high”: natural opioids released by dieting and intense exercise dampen stress and produce emotional relief, encouraging further weight loss rather than normal refeeding. Animal models in which food restriction is combined with voluntary running show similar patterns—hyperactivity, weight loss, and signs that the opioid system has been driven to such a high baseline that external opioids lose their impact. Importantly, blocking opioid receptors in some studies appears to help patients with anorexia gain weight and encourages rodents to increase fat intake, suggesting a potential treatment avenue.
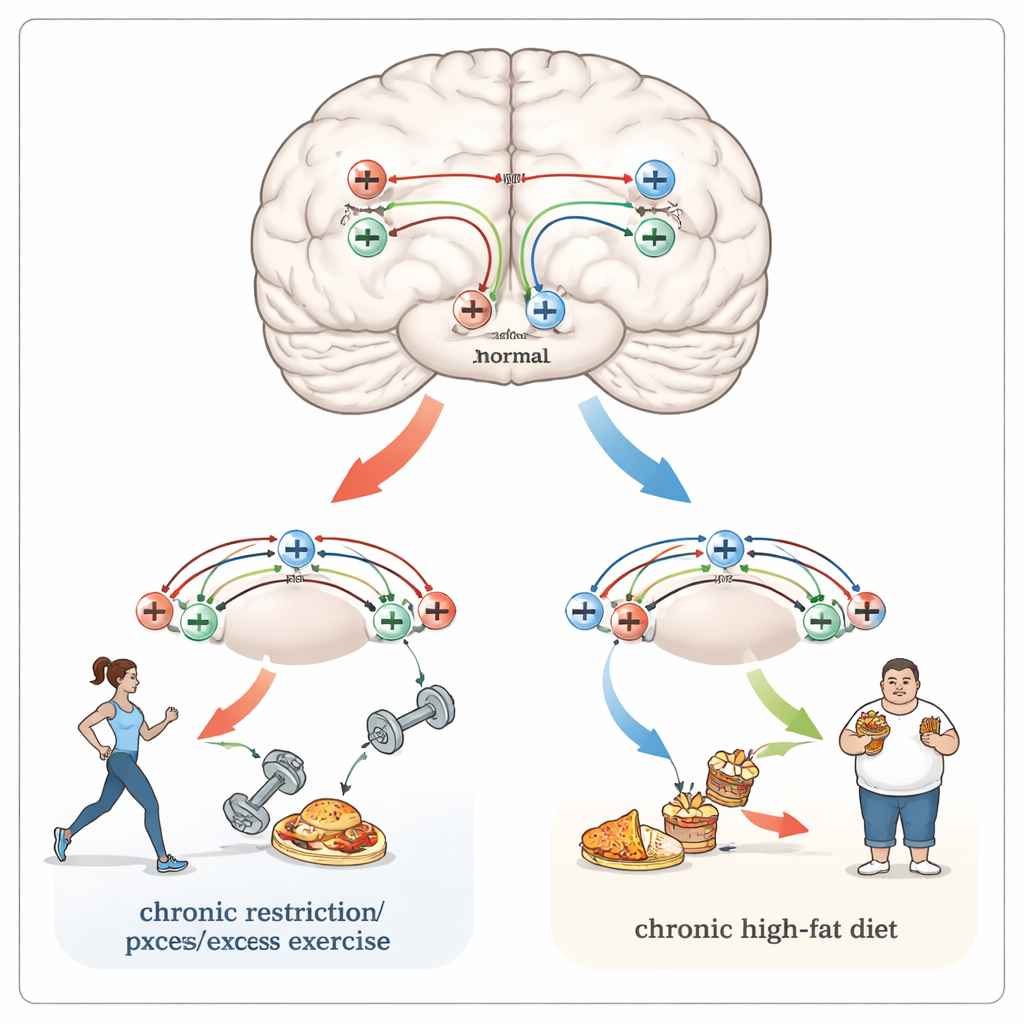
Shared links to pain, addiction, and rigid habits
The same brain regions and receptors that shape eating also process pain and drug rewards. People with obesity tend to experience more pain, including migraines, while those with long-standing anorexia often show a blunted response to painful stimuli. Natural opioids, acting in regions like the hypothalamus, brainstem, and nucleus accumbens, can both soften pain and shift attention toward urgent needs such as hunger. These circuits overlap with pathways that are reshaped by addictive drugs, and chronic food restriction is known to heighten sensitivity to substances like opioids in animal studies. Both obesity and anorexia are also tied to reduced cognitive flexibility—the ability to adapt habits to new circumstances—which may reflect deeper, long-lasting changes at connections between nerve cells driven in part by opioid receptors.
What this means for understanding and treating extreme eating
Taken together, the evidence suggests that the brain’s own opioid system helps determine whether changes in eating—such as exposure to rich foods or periods of dieting—remain temporary and adaptive or spiral into long-term, harmful patterns. In obesity, repeated overeating appears to remodel reward and hunger circuits so that high-calorie foods become especially compelling. In anorexia, starvation and excessive exercise may hijack the same opioid-driven learning processes to make self-denial and hyperactivity feel rewarding despite dangerous weight loss. By pinpointing where and how these receptors change, the authors argue, future research may guide more precise treatments—such as targeted receptor blockers, brain stimulation, or drugs that normalize synaptic plasticity—to nudge the brain’s feast-or-famine circuitry back toward balance.
Citation: Sutton Hickey, A.K., Matikainen-Ankney, B.A. Endogenous opioid receptors and the feast or famine of maladaptive feeding. Nat Commun 16, 2270 (2025). https://doi.org/10.1038/s41467-025-57515-0
Keywords: endogenous opioids, eating disorders, obesity, anorexia nervosa, brain reward circuits