Clear Sky Science · en
Drug repurposing for Alzheimer’s disease and other neurodegenerative disorders
Why Old Medicines Matter for Brain Health
As Alzheimer’s disease and related brain disorders rise worldwide, scientists are searching not only for brand-new drugs, but also for fresh uses of medicines we already have. This strategy, known as drug repurposing, asks a simple question: can a pill approved for conditions like diabetes, high blood pressure, or depression also help protect memory, thinking, or behavior in Alzheimer’s disease and other neurodegenerative disorders? Because these older drugs have known doses and safety records, repurposing them could bring helpful treatments to patients faster and at lower cost, including in low- and middle-income countries where dementia is growing most rapidly.
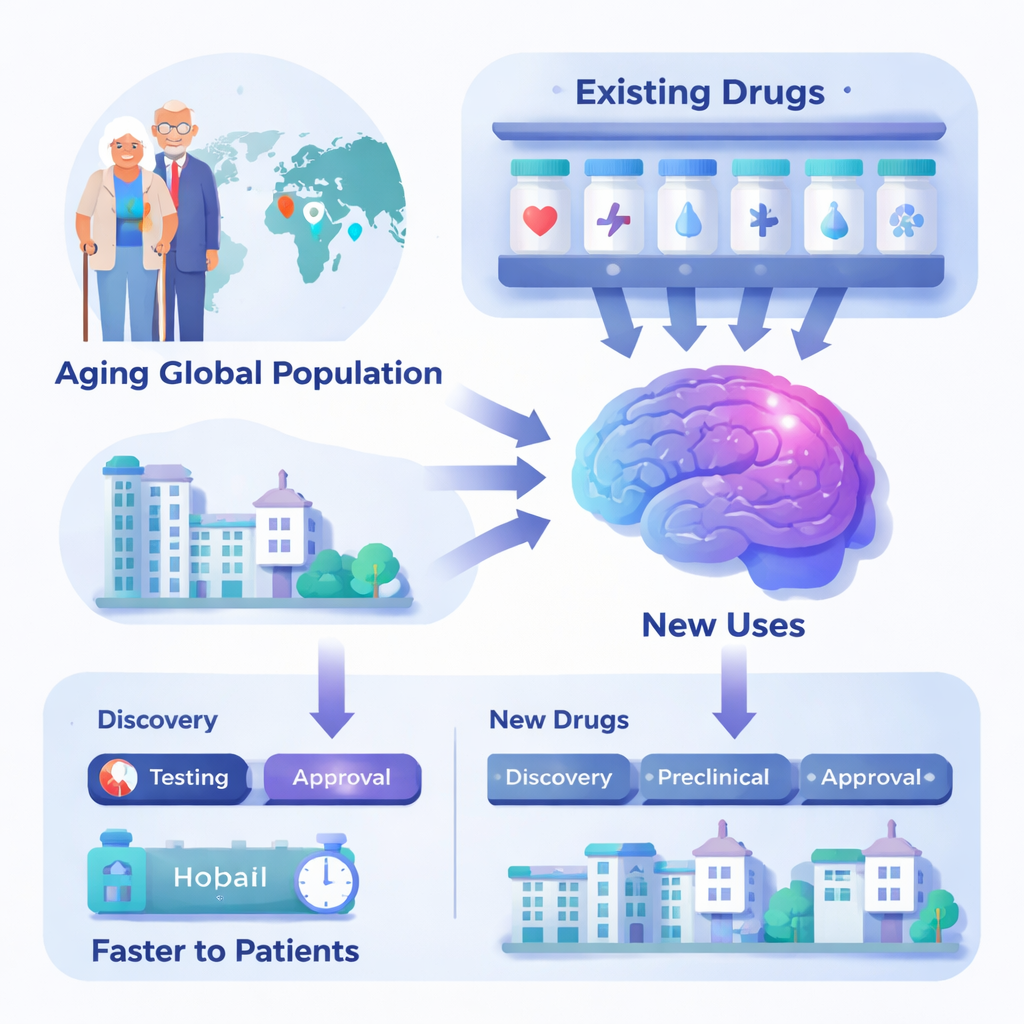
The Growing Weight of Memory Disorders
Alzheimer’s disease already affects tens of millions of people, and that number is expected to nearly triple by 2050, with most new cases occurring in less wealthy regions. Many additional people live with milder problems such as mild cognitive impairment or early, symptom-free brain changes. Other degenerative brain diseases, like Parkinson’s disease, frontotemporal dementia, and amyotrophic lateral sclerosis, are also becoming more common as populations age. Current treatments, including recently approved antibody drugs that target amyloid plaques, help only certain patients in early stages and are expensive and hard to deliver widely. At the same time, traditional drug development is slow, risky, and costly, often taking more than a decade and failing in late stages.
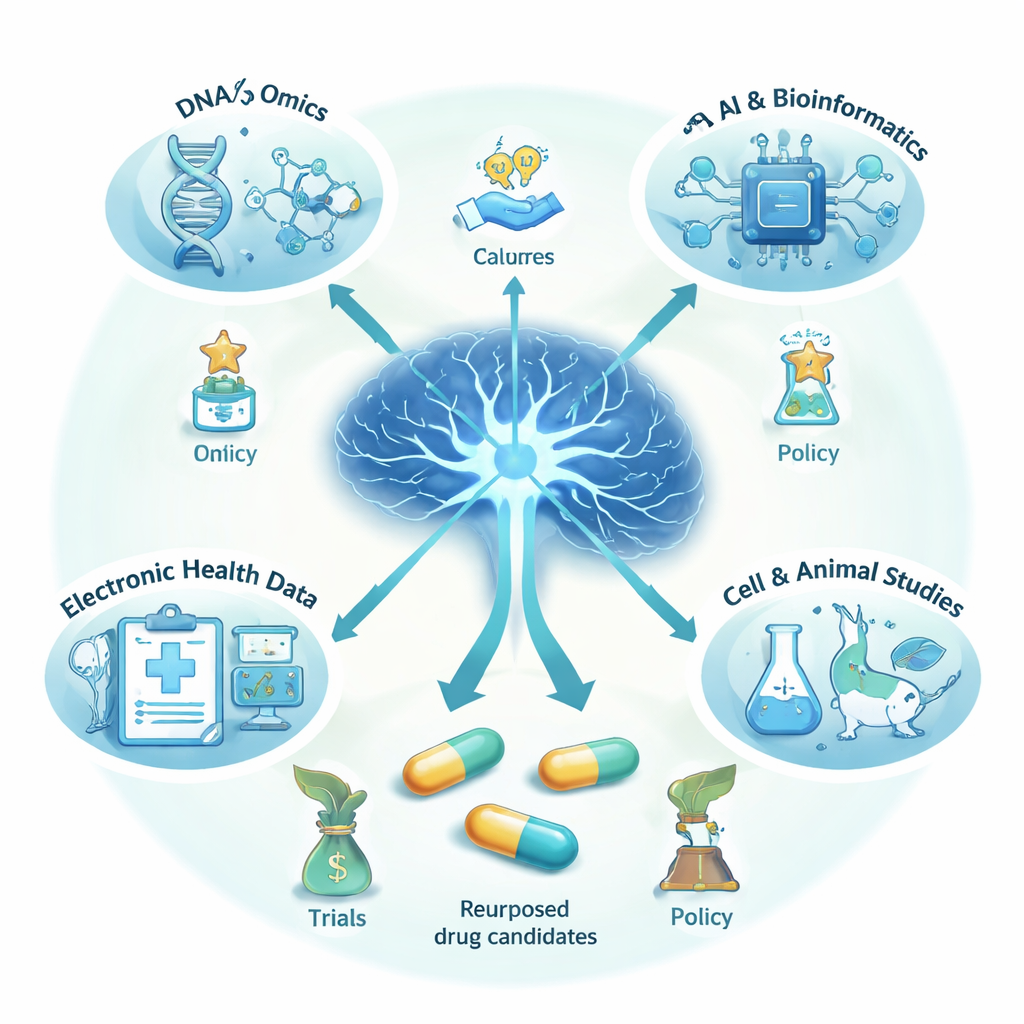
How Researchers Find New Uses for Old Drugs
Instead of starting from scratch, scientists now mine huge sources of data to look for existing medicines that might help the brain. They use genetic and other “omics” information from thousands of human brains, advanced computer models, and networks that link diseases, proteins, and drugs. They also study electronic health records and insurance claims to see whether people taking certain medications, such as heart pills or asthma sprays, are less likely to develop dementia. In some cases, they recreate trial-like conditions with these real-world data to estimate how a drug might perform in a formal study. Promising signals are then tested in lab-grown nerve cells, brain organoids, and animal models to see whether a drug can reduce harmful proteins, calm inflammation, or protect connections between brain cells.
Why Repurposed Drugs Offer a Head Start
Repurposed medicines come with major built-in advantages. Their basic safety, side effects, and dosing were already established when they were first approved, so researchers can sometimes skip early safety trials and move directly into studies that test whether the drug helps memory, thinking, or daily function. This can save several years and tens or even hundreds of millions of dollars compared with developing a brand-new compound. Many of these agents are generic, meaning they are inexpensive to manufacture and could be more accessible globally if proven effective. Trials of repurposed drugs also help build clinical research centers, train specialists, and refine new brain scans, blood tests, and digital tools that benefit all dementia research.
Roadblocks: Money, Patents, and Policy
Despite their promise, generic repurposed drugs face tough obstacles. Because most are off patent, companies have little financial incentive to pay for large, late-stage trials, regulatory applications, and marketing. There is no easy way to charge higher prices for a “new use” of a cheap generic without others simply selling the same pill for less. The authors discuss creative solutions, including special legislative protections, tax credits, temporary exclusivity linked to the new indication, innovation fees on generic sales, and novel partnerships among governments, charities, universities, and industry. Scientific strategies such as new formulations, pro-drugs, and combination therapies can also create limited but meaningful intellectual property that attracts investment.
What This Means for Patients and Families
The article concludes that repurposing is not a shortcut around rigorous science, but a powerful tool for speeding safer, affordable treatments for Alzheimer’s disease and other brain disorders. Even when a specific repurposed drug does not reach the market, the trials themselves sharpen diagnostic tools, improve study design, expand research sites worldwide, and train the next generation of dementia scientists. With smarter use of big data, laboratory models, real-world health records, and supportive policy changes, repurposed drugs could become an important pillar of global efforts to delay, prevent, or ease the burden of neurodegenerative diseases.
Citation: Cummings, J.L., Zhou, Y., Van Stone, A. et al. Drug repurposing for Alzheimer’s disease and other neurodegenerative disorders. Nat Commun 16, 1755 (2025). https://doi.org/10.1038/s41467-025-56690-4
Keywords: drug repurposing, Alzheimer’s disease, dementia, neurodegenerative disorders, clinical trials