Clear Sky Science · en
Illuminating the impact of N-terminal acetylation: from protein to physiology
How tiny chemical tags can change protein behavior
Every cell in your body is packed with proteins that must fold correctly, go to the right place, assemble into molecular machines, and be removed when damaged. This review article explores a subtle but enormously common chemical change at one end of many proteins—called N-terminal acetylation—and shows how this small “cap” helps control everything from cell growth and stress responses to heart development and brain function.
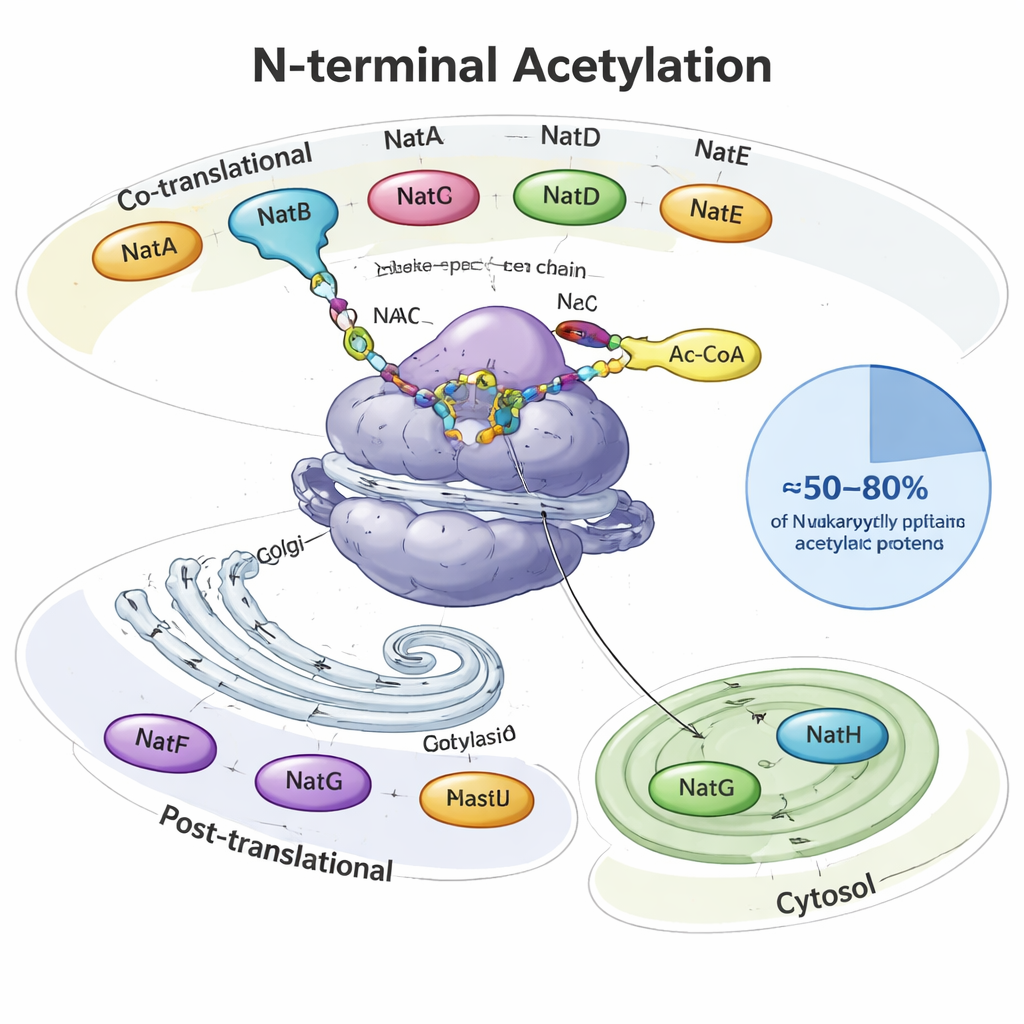
The protein cap that most of us never heard of
When a protein is made, it emerges from the cell’s protein factory, the ribosome, like a growing thread. At its very beginning—the N-terminus—many eukaryotic proteins receive an acetyl group, a tiny two‑carbon cap. Specialized enzymes called N-terminal acetyltransferases (NATs) perform this job, using a common metabolic molecule (acetyl‑CoA) as the donor. In yeasts, plants, and animals, eight main NAT types (NatA–NatH) divide the labor. Some act while the protein is still being made, others work afterward and are stationed at specific locations such as the Golgi apparatus or plant plastids. Together they modify roughly half to four‑fifths of all cellular proteins, each NAT recognizing preferred starting sequences of two to four amino acids.
Many enzymes, each with its own specialty
NatA, NatB and NatC are the heavy lifters, acting right at the ribosome and covering large swaths of the “N‑terminal acetylome.” NatA alone can modify around 40% of human proteins, usually those whose first methionine has been trimmed away. NatB and NatC act on proteins that keep their initial methionine, but differ in which neighboring amino acids they prefer. Other NATs are more selective: NatD focuses on just a few histone proteins that package DNA, NatF targets transmembrane proteins at the Golgi, NatG works inside plant plastids, and NatH (NAA80) finishes a special two‑step maturation process on actin, a major component of the cell’s skeleton. The article pulls together structural and biochemical studies showing how helper subunits anchor these enzymes to ribosomes or membranes and fine‑tune which proteins they act on.
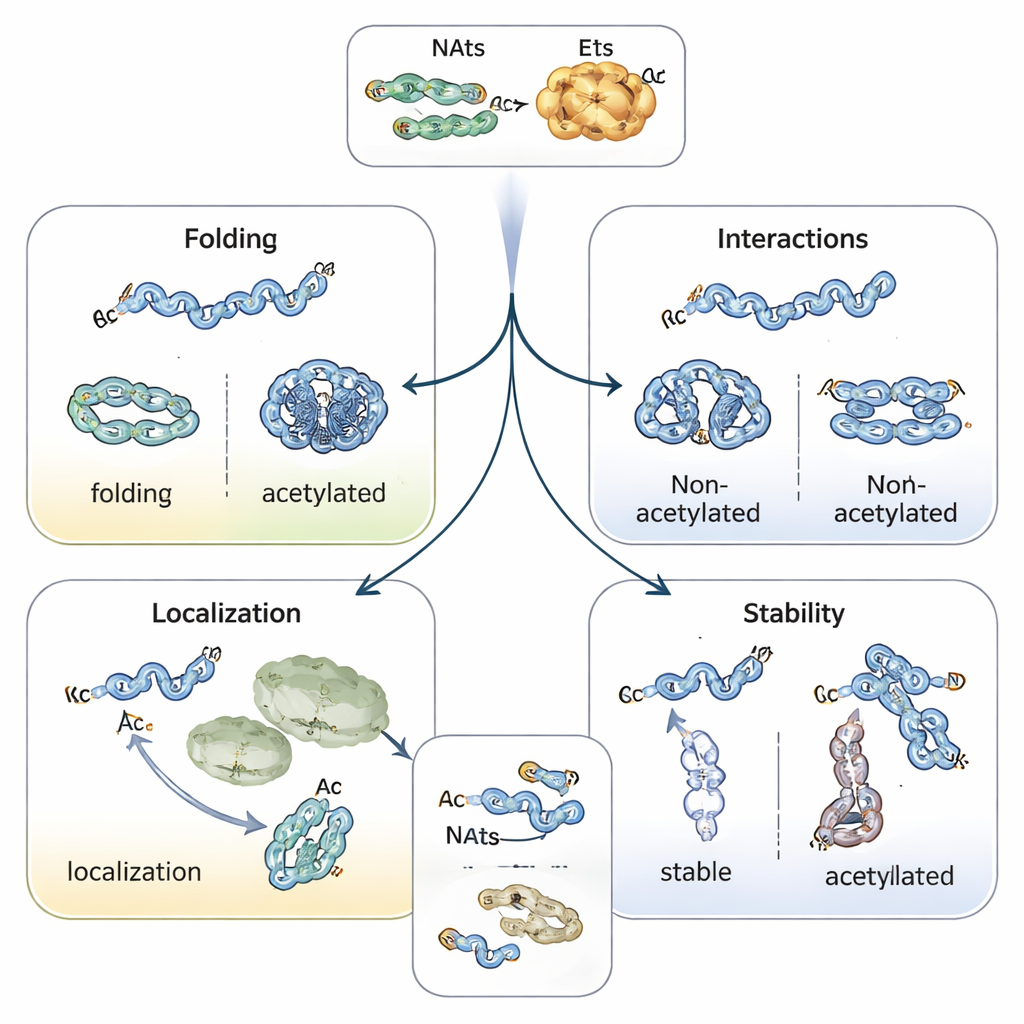
Steering protein folding, location, and lifespan
Adding an acetyl cap changes the chemical nature of the protein’s beginning, often stabilizing helical structures and influencing how proteins fold. For example, the Parkinson’s‑linked protein alpha‑synuclein tends to clump less and adopt a safer shape when its N‑terminus is acetylated. Tropomyosin, a partner of actin, binds and regulates actin filaments much better when properly acetylated. In plants and animals, N‑terminal acetylation can serve as a kind of address label, helping proteins find chloroplasts, lysosomes, or the Golgi. Perhaps most striking is its role in determining how long proteins survive: an unprotected N‑terminus can be read as a “degradation signal” by ubiquitin‑tagging enzymes, while acetylation can mask that signal and stabilize the protein—or, in some cases, create a conditional tag that is only recognized when the protein is misfolded or not in the right complex.
From cell physiology to development and disease
Because N-terminal acetylation touches so many proteins, disrupting NATs has broad consequences for whole organisms. Removing NatA, NatB, or NatC in model species often causes severe developmental defects, altered stress responses, or even lethality, while more specialized enzymes such as NatF and NatH produce narrower problems like brain calcifications or hearing loss. In humans, inherited mutations in several NAT genes underlie rare neurodevelopmental syndromes with intellectual disability, heart defects, or muscle weakness. The same machinery is frequently overactive in cancers, where elevated NAT levels correlate with poor prognosis and may drive tumor growth, metastasis, and resistance to therapy. Changes in N‑terminal acetylation also influence aggregation‑prone proteins such as alpha‑synuclein and huntingtin, linking this modification to neurodegenerative disease mechanisms.
Future directions and medical promise
The authors conclude that while we now know most of the core enzymes that add these N‑terminal caps, we still understand only a fraction of the specific protein targets and regulatory rules. A major theme is that acetylation at the protein’s beginning often shields it from premature destruction, while also creating context‑dependent signals for quality control. Pinpointing which ubiquitin ligases read which N‑terminal patterns, and discovering whether any enzyme can remove these caps, are key open questions. Because NATs sit at the crossroads of protein stability, gene regulation, and cell stress responses, selective NAT inhibitors—and perhaps future tools that tweak N‑terminal acetylation of specific disease‑related proteins—are emerging as promising avenues for new therapies in cancer, heart disease, and neurodegeneration.
Citation: McTiernan, N., Kjosås, I. & Arnesen, T. Illuminating the impact of N-terminal acetylation: from protein to physiology. Nat Commun 16, 703 (2025). https://doi.org/10.1038/s41467-025-55960-5
Keywords: protein modification, N-terminal acetylation, protein quality control, neurodegenerative disease, cancer biology