Clear Sky Science · en
m6A RNA methylation modulates antiviral response in celiac disease
When a Virus and Gluten Team Up
Celiac disease is often thought of as a simple food problem: eat gluten and your gut gets hurt. This study shows a deeper story, where a common type of gut virus and a tiny chemical tag on our RNA molecules work together to push the immune system toward long‑lasting damage. Understanding this hidden layer of control may help explain why only some people develop celiac disease and point to new treatments that go beyond the gluten‑free diet. 
A Hidden Tag on Our Genetic Messages
Our cells constantly read genes and copy them into RNA, short “messages” that tell the cell which proteins to make. These messages are not fixed; they can be decorated with chemical marks that act like highlighters, changing how strongly a message is read. One of the most common marks is called m6A, a tiny change to the RNA letters. Viruses are known to alter these RNA marks in infected cells, and earlier work had already linked m6A to the way gluten sparks inflammation in the celiac gut. The authors of this paper asked whether a virus‑driven change in m6A could help flip the immune system from tolerance of food to attack.
Evidence from Patients with Celiac Disease
The researchers first looked at blood and intestinal biopsies from people with active celiac disease and from non‑celiac controls. They found that celiac patients carried higher levels of antibodies against reovirus, a double‑stranded RNA virus that infects the gut, suggesting more frequent or stronger exposure. In the gut tissue of these patients, key antiviral warning genes, especially one called IRF7, were switched on at higher levels, along with inflammatory molecules such as STAT1 and the immune‑cell attractant CXCL10. At the same time, the machinery that adds and reads m6A marks was more active, and total m6A levels on RNA were higher. The expression of antiviral genes rose and fell together with m6A‑related genes, hinting that the same control system might be driving both viral defense and autoimmune damage.
How Gluten Amplifies the Viral Alarm
To probe cause and effect, the team built a celiac‑like model in intestinal cells grown in the lab. They mimicked a viral infection using a synthetic double‑stranded RNA, then added digested gluten fragments similar to what would appear in the gut after a meal. Viral mimic alone boosted both IRF3 and IRF7, two master switches for antiviral responses. But when gluten was added on top, IRF7 showed a strong extra surge, while IRF3 did not. The researchers discovered that, in these conditions, m6A marks increased on a specific region within the IRF7 RNA, and that this RNA physically interacted with an m6A‑writing enzyme (METTL3) and an erasing enzyme (ALKBH5). Elevating m6A, either by adding more METTL3 or by reducing ALKBH5, drove IRF7 levels still higher, tying the gluten‑plus‑virus synergy directly to this RNA modification. 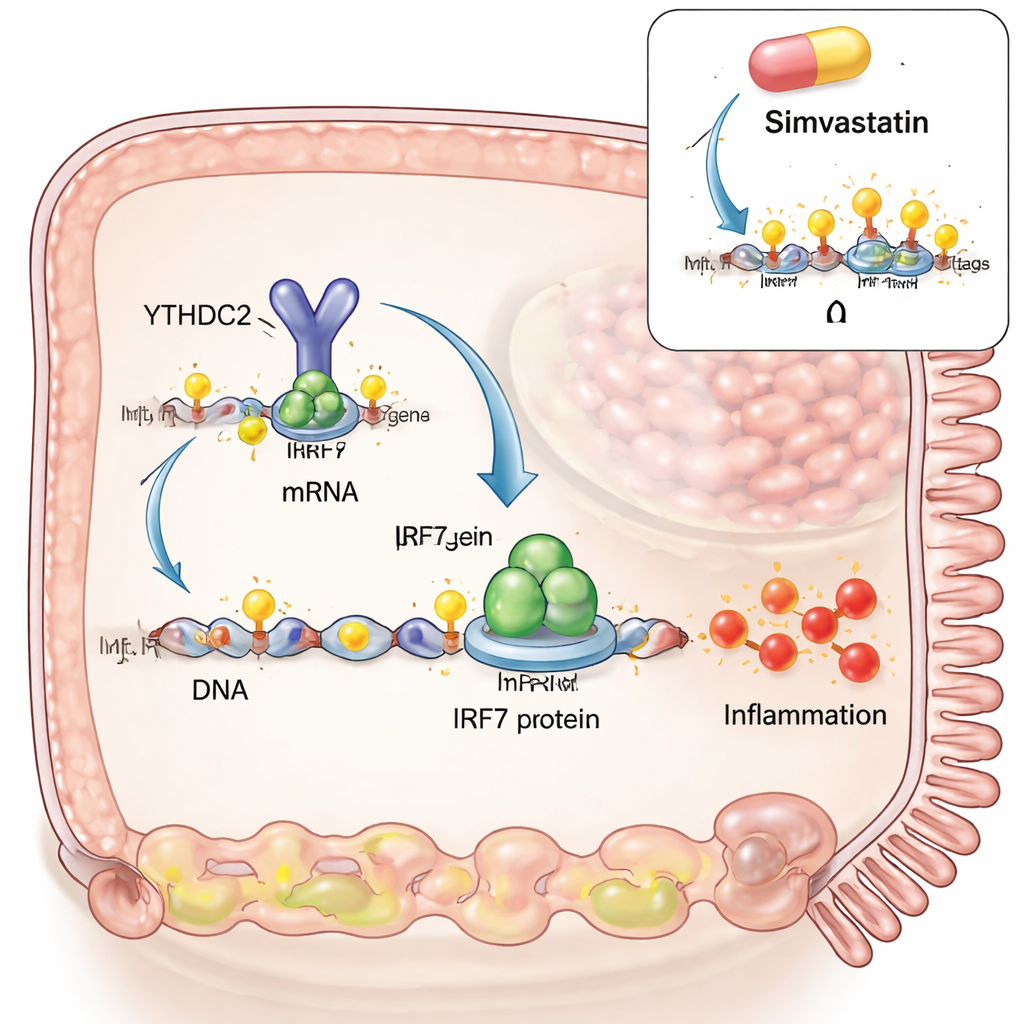
The Molecular Middleman and a Surprising Drug
Next, the authors asked how m6A marks on IRF7 RNA actually change the cell’s behavior. They engineered a version of IRF7 RNA with the same protein code but without the m6A sites. This RNA made similar amounts of transcript, but far less IRF7 protein, and it failed to trigger downstream inflammatory genes like STAT1 and CXCL10. The missing link was a reader protein called YTHDC2, which normally binds m6A within the protein‑coding region and helps drive efficient translation into protein. Without the m6A sites, YTHDC2 could no longer latch onto IRF7 RNA. Blocking the m6A writer METTL3 in the cell model reduced IRF7 protein and CXCL10, lowering the inflammatory output. A cholesterol‑lowering drug, simvastatin, which has recently been shown to reduce m6A in other tissues, also cut m6A on IRF7 RNA, reduced IRF7 protein in cells, and dampened IRF7‑related genes in freshly obtained celiac gut biopsies grown ex vivo. Large‑scale gene‑expression comparisons suggested that simvastatin pushes celiac tissue toward a profile more like that of patients on a gluten‑free diet.
What This Means for People with Celiac Disease
This work paints a picture in which a gut virus and gluten act together on a fine‑tuned layer of RNA control. In susceptible people, viral infection appears to boost m6A tagging of IRF7 RNA in intestinal cells. Gluten exposure then further amplifies that tagged signal, leading to more IRF7 protein, stronger antiviral alarms, and higher levels of inflammatory messengers that help drive the autoimmune attack on the gut lining. By dialing down m6A marks—either genetically or with a drug like simvastatin—the chain reaction can be softened, at least in lab and ex vivo models. For lay readers, the key message is that celiac disease is not just about what you eat, but also about how infections and subtle chemical marks on RNA shape the immune system’s decision to tolerate or attack. Targeting these RNA marks could someday complement the gluten‑free diet and inspire new treatments for a broader range of autoimmune diseases.
Citation: Sebastian-delaCruz, M., Olazagoitia-Garmendia, A., Pascual-Gonzalez, I. et al. m6A RNA methylation modulates antiviral response in celiac disease. Genes Immun 27, 130–139 (2026). https://doi.org/10.1038/s41435-025-00373-z
Keywords: celiac disease, RNA methylation, antiviral immunity, IRF7, simvastatin