Clear Sky Science · en
CRISPR-AuNP: physicochemical optimization of a gold nanoparticle platform for cost-effective and modular non-viral gene editing in HSPCs
Bringing Gene Editing to More Patients
Gene-editing tools like CRISPR have already changed the lives of people with inherited blood diseases, but the treatments are still expensive, technically demanding, and largely limited to a few specialized centers. This paper describes a new way to deliver CRISPR into blood-forming stem cells using tiny gold-based particles that can be mixed on a lab bench in just a couple of hours. The goal is to make powerful gene-editing approaches simpler, cheaper, and easier to deploy in more places around the world.
Why Blood Stem Cells Matter
Blood-forming stem and progenitor cells live in the bone marrow and continually refill all the different types of blood cells in the body. If doctors can safely fix disease-causing mutations in these cells, a one-time treatment could provide a lifelong cure for conditions such as sickle cell disease and certain immune deficiencies. Today, the most common way to get CRISPR into these cells relies on electroporation, a process that uses electric pulses to open holes in cell membranes. While powerful, electroporation is harsh on the cells, requires expensive equipment and specialized reagents, and is not easily scaled or exported to resource-limited settings. Safer viral vectors are excellent for many gene therapies, but they are less suited for CRISPR delivery because they keep producing editing tools for too long, raising the risk of off-target changes.
Building a Tiny Gold Delivery Vehicle
The researchers set out to improve a previously reported gold nanoparticle system that carried CRISPR components on its surface. In earlier designs, the RNA that guides the CRISPR cutting protein to its target was anchored directly onto the gold core and then paired with the Cas9 protein. This worked reasonably well for one type of CRISPR enzyme but performed poorly with Cas9, which is central to many therapeutic strategies. By closely examining how Cas9 and its guide RNA behave under the acidic conditions used during particle assembly, the team discovered that the guide RNA duplex became unstable on the gold surface, leading to little or no active Cas9 attached to the particle. Confocal microscopy also suggested that the polymer coating intended to help the particles escape from cellular compartments peeled away before it could do its job.
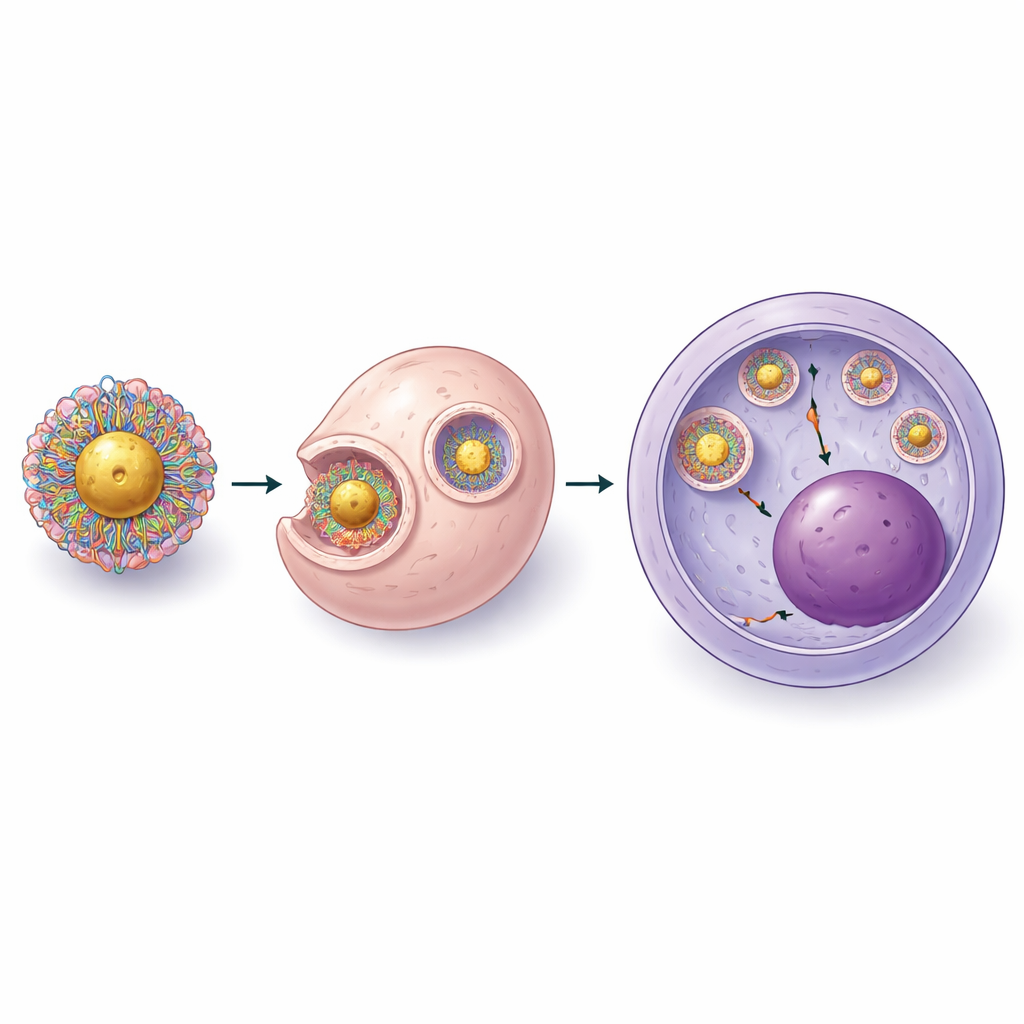
Stepwise Redesign of the Platform
To overcome these problems, the scientists rebuilt the system from the ground up in three stages. First, instead of anchoring bare RNA to the gold and then adding protein, they formed complete CRISPR ribonucleoprotein complexes (protein plus guide RNA) before bringing them into contact with the gold surface. They identified a slightly acidic condition that allowed these complexes to bind the particles while still keeping the cutting protein active. Next, they wrapped the loaded particles with specially engineered polymers that combined positively charged chains—needed to escape cellular compartments—with flexible, water-loving polyethylene glycol segments that prevent clumping. By measuring size, surface charge, and protein content, they showed they could reproducibly load dozens of CRISPR complexes per particle while maintaining stability in solution.
Cracking the Cell’s Defenses
Even with better loading, the second-generation particles still struggled with a key hurdle: getting CRISPR out of the cell’s internal vesicles and into the part of the cell where DNA lives. The team therefore systematically tuned the chemistry of the polymer layer and the ratio between positive groups on the polymer and negative groups on the CRISPR complex. They discovered that adding more short polyethylene glycol chains and a controlled number of sulfur-containing anchors improved both particle stability and the ability to disrupt endosomal membranes without harming the cells. In the final, third-generation design, they first created compact complexes of CRISPR and polymer, then attached these to concentrated gold cores. Electron microscopy showed a thin shell of CRISPR and polymer around each gold sphere, and imaging in live stem cells confirmed that the cargo could escape into the cell’s interior and reach the nucleus.
Editing Results Across Multiple CRISPR Tools
To test how well the optimized particles worked, the researchers targeted a gene called beta-2-microglobulin, which produces a protein found on the surface of nearly all blood cells and can be easily measured by flow cytometry. Using their third-generation gold platform, they achieved more than 10% precise disruptions of this gene in primary human blood stem and progenitor cells—levels considered meaningful for many therapeutic applications—while maintaining high cell survival. Although this efficiency is lower than what can be achieved with electroporation, the gold system required no specialized equipment and caused minimal stress to the cells. Importantly, the same platform could be used with different CRISPR proteins, including Cas9, Cas12a, and a newer enzyme called Cas12a-M29-1, with similar editing levels and no detectable toxicity, highlighting the modular nature of the design.
What This Could Mean for Future Therapies
In plain terms, this work shows that it is possible to build a small, carefully engineered gold-based carrier that can bring CRISPR tools into sensitive blood stem cells without electricity or viruses, using off-the-shelf materials for under seventy dollars per million cells. While further improvements are needed to match the efficiency of today’s best clinical methods, particularly for diseases that demand very high levels of corrected cells, this bench-assembled platform lowers practical barriers to performing sophisticated gene editing. With continued refinement and safety testing, such nanoparticles could help spread the benefits of CRISPR-based therapies beyond specialized centers, making curative treatments more attainable for patients worldwide.
Citation: Gottimukkala, K.S.V., Lane, D.D., Cunningham, R. et al. CRISPR-AuNP: physicochemical optimization of a gold nanoparticle platform for cost-effective and modular non-viral gene editing in HSPCs. Gene Ther 33, 188–202 (2026). https://doi.org/10.1038/s41434-025-00591-0
Keywords: CRISPR gene editing, gold nanoparticles, hematopoietic stem cells, non-viral delivery, nanomedicine