Clear Sky Science · en
Risk of retinal vein occlusions in patients taking systemic tyrosine kinase inhibitors
Why this matters for people living with cancer
Targeted cancer pills known as tyrosine kinase inhibitors (TKIs) have transformed treatment, often with fewer whole‑body side effects than traditional chemotherapy. Yet even these drugs are not completely risk‑free. This study explores a rare but serious eye problem—retinal vein occlusion, a kind of “blood clot in the vein of the eye”—that appeared in a group of patients taking TKIs. For anyone living with cancer, or caring for someone who is, understanding these risks can help protect vision while still benefiting from life‑saving cancer therapy.
Targeted drugs and a hidden eye risk
TKIs work by blocking key signaling switches inside cells that drive cancer growth. Because they hone in on specific pathways, they are generally thought to spare healthy tissues more than older chemotherapy drugs do. However, doctors have increasingly reported eye‑related side effects, including swelling of the retina and eyelids. Scattered case reports have also described retinal vein occlusion (RVO) in people taking TKIs. RVO occurs when a vein in the light‑sensing tissue at the back of the eye becomes blocked, which can suddenly blur or darken vision and sometimes cause permanent sight loss. The authors of this paper set out to see whether these events might be linked to TKI treatment rather than being coincidental.
What the doctors did and who was affected
Researchers at the Cleveland Clinic reviewed ten years of records to find patients who had both taken a TKI and developed an RVO. After carefully excluding people whose vein problem started before the drug, or who did not truly have RVO, they identified 12 affected eyes in 11 patients. Most were older adults, with an average age of about 76, and nearly three‑quarters were men. Every patient had high blood pressure, more than half had diabetes, and most had high cholesterol—well‑known risk factors for eye vein blockages. Their cancers included chronic myeloid and chronic lymphocytic leukemia, kidney cancer, and a gastrointestinal stromal tumor. The TKIs involved were imatinib, axitinib, ibrutinib, and regorafenib.
How and when vision problems appeared
On average, patients developed RVO roughly three years after starting a TKI, though the range was from just under one year to more than six years. Two‑thirds of the affected eyes had a central retinal vein occlusion, where the main drainage vein is blocked, while the rest had a branch occlusion involving a smaller side vein. Vision at the time of diagnosis ranged from mildly blurred to seeing only hand movements. Most eyes were treated with injections of drugs that block vascular endothelial growth factor (anti‑VEGF medicines), sometimes combined with a steroid implant or laser, to reduce swelling and leakage. At final follow‑up, half the eyes had improved vision, a quarter were unchanged, and a quarter had worsened. Decisions about whether to continue cancer therapy varied: in some patients the TKI was stopped immediately, in others it was held temporarily or continued, balancing cancer control against eye risk.
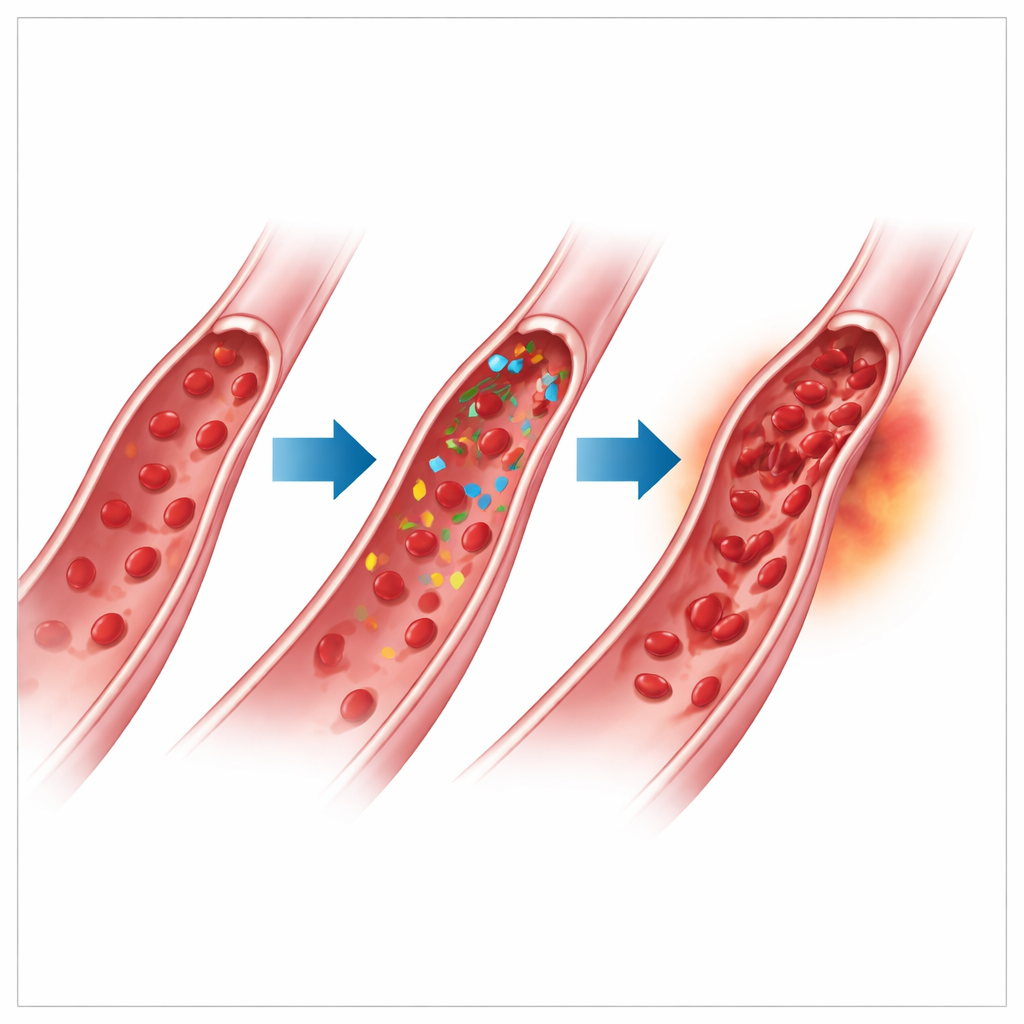
Clues that point toward the cancer drugs
Because these patients already had many reasons to form blood clots, the authors used a standard tool called the Naranjo Adverse Drug Reaction Scale to judge how likely it was that the TKI contributed to the RVO. The average score suggested a “probable” link. In one striking case, a woman on regorafenib developed a severe vein blockage in one eye and, months later, in the other eye while still on the drug. Her score was among the highest, in part because the problem recurred while treatment continued. The authors discuss how TKIs that interfere with vascular growth signals may disturb the normal balance between clot‑promoting and clot‑preventing factors in blood vessels, potentially tipping susceptible patients toward vein blockage in the eye. Still, they stress that aging, high blood pressure, diabetes, high cholesterol, and even the cancer itself likely all play a role.
What this means for patients and clinicians
Although the overall risk of RVO in people taking TKIs appears low—on the order of a small fraction of a percent—the consequences can be life‑altering for those who lose vision. This case series, the largest of its kind, does not prove that TKIs directly cause eye vein clots, but it strengthens the suspicion of an association, especially in older patients with multiple vascular problems or existing eye disease. The authors suggest that such high‑risk patients may benefit from a baseline eye exam and closer monitoring once TKI treatment begins. Most importantly, people on these medicines should be told to seek urgent eye care if they notice sudden blurring, dark spots, or distortion in their vision. With quick recognition and modern retinal treatments, some vision can often be preserved, allowing patients to continue benefiting from targeted cancer therapies as safely as possible.
Citation: Mohan, N., Srivastava, S.K., Duphare, C. et al. Risk of retinal vein occlusions in patients taking systemic tyrosine kinase inhibitors. Eye 40, 697–704 (2026). https://doi.org/10.1038/s41433-026-04240-7
Keywords: tyrosine kinase inhibitors, retinal vein occlusion, ocular toxicity, cancer therapy side effects, anti-VEGF treatment