Clear Sky Science · en
Pleiotropic genes linking congenital hypogonadotropic hypogonadism and cleft lip/palate: evidence from a genomic CHH cohort study
When Puberty and Smile Share the Same Roots
Most people think of delayed puberty and cleft lip or palate as completely separate health problems—one affects hormones and fertility, the other the shape of the face and mouth. This study reveals that, in many patients, these two conditions can spring from the same underlying genes. Understanding this hidden connection not only reshapes how doctors think about these disorders, but could also help families receive earlier diagnoses and more personalized care.
Two Conditions That Start Before Birth
Congenital hypogonadotropic hypogonadism (CHH) is a rare condition in which puberty never fully starts because the brain does not release enough of a key hormone that controls the ovaries or testes. Cleft lip and palate (CLP), by contrast, are among the most common birth differences, in which the upper lip or roof of the mouth do not fuse completely during early development. On the surface these problems seem unrelated. Yet both depend on precisely timed events in the embryo: specialized nerve cells must travel to the brain’s hormone centers, and facial tissues must grow and join along the midline of the face. The authors suspected that common genetic instructions might guide both processes.
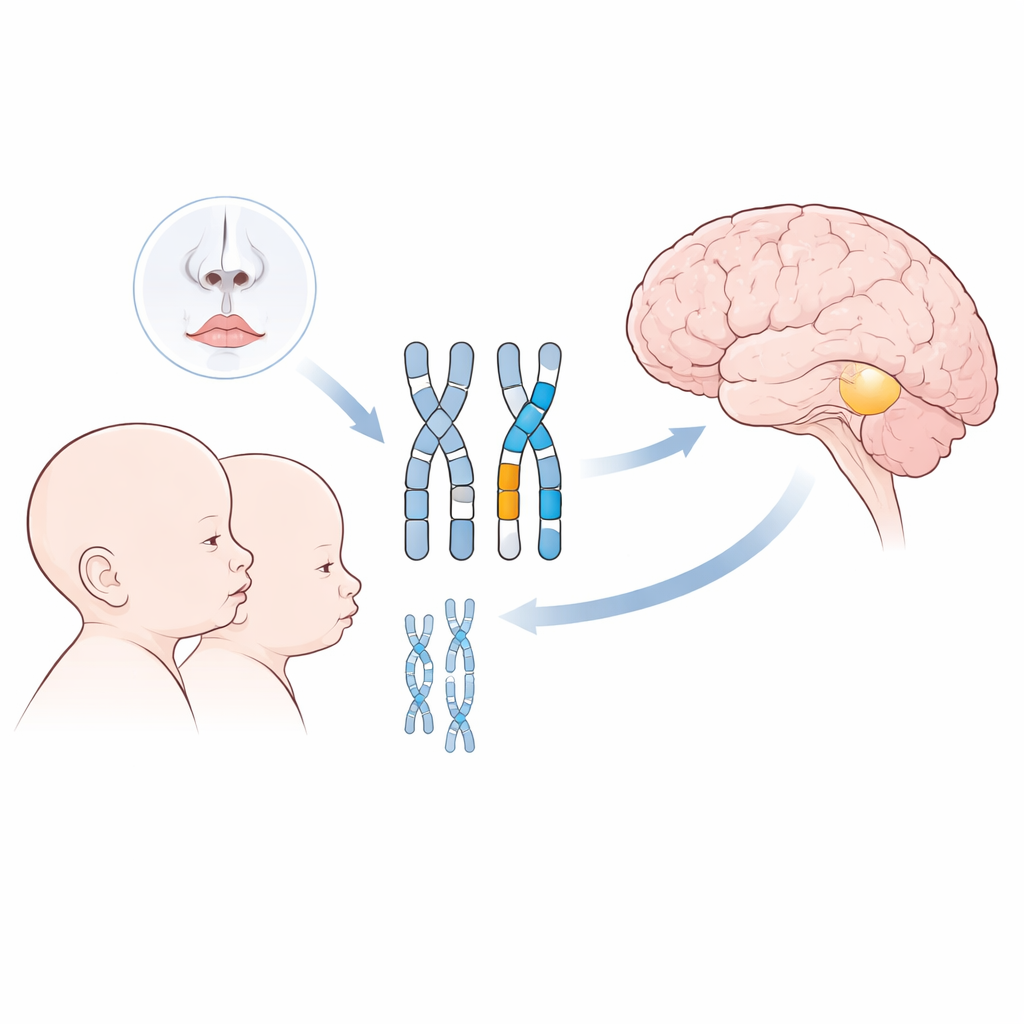
Searching the Genome for Shared Clues
The researchers studied 336 people diagnosed with CHH, carefully documenting whether they also had cleft lip or palate or other developmental differences. They then used high-throughput DNA sequencing to search for rare, damaging changes in two large sets of genes: one set already tied to CHH and another linked to clefting. Each genetic variant was graded for how likely it was to cause disease, following international guidelines. By comparing clinical records with genetic findings, the team could see when the same genes seemed to influence both lack of puberty and facial formation.
Key Genes That Do Double Duty
Among all participants with CHH, about 6 percent also had a cleft lip or palate. In half of these, the team found clearly harmful variants in genes previously connected to clefting, and most of those genes—especially FGFR1 and CHD7—were also known players in CHH. Changes in FGFR1 were the most frequent, appearing in seven patients with both CHH and CLP and in many others with CHH alone. These individuals often had a poor or absent sense of smell and sometimes additional features such as hearing loss, tooth anomalies, growth problems, or brain structure differences. Similar patterns were seen with variants in CHD7, SOX9, and CTNND1, genes already recognized for their roles in complex developmental syndromes affecting the face, ears, skeleton, and heart.
Hidden Cleft Genes in Patients Without a Cleft
Strikingly, the story did not stop with patients who visibly had clefts. Among the 315 CHH patients without any cleft lip or palate, nearly 16 percent still carried harmful variants in genes that have been associated with clefting. Some of these genes—such as DVL3, PLCB4, NIPBL, and EDNRA—had not previously been linked to CHH at all. Their usual roles include shaping the skull, jaw, and limbs, and they are known to cause rare syndromes that sometimes involve small testes, undescended testes, or subtle brain differences. The finding that changes in these genes appear in people whose main problem is absent puberty suggests they may also influence the hormone system, even when the face looks typical.
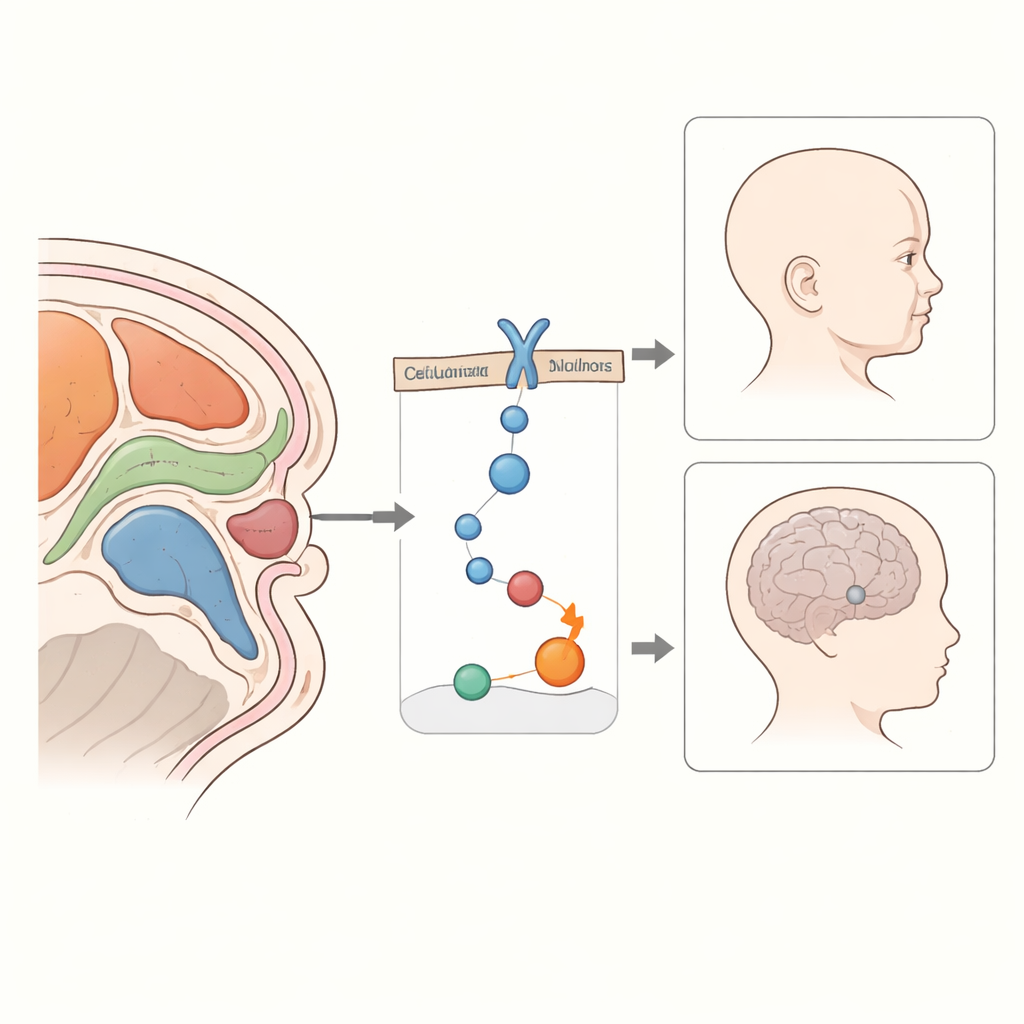
Many Genes, Many Combinations
Another insight from this work is that some patients carried harmful variants in more than one relevant gene. For example, several individuals had changes in FGFR1 together with variants in other developmental genes, hinting that a combination of hits may be needed to produce the full picture of symptoms. This “multi-gene” or oligogenic pattern could explain why one family member has a severe syndrome with facial clefts, while a relative with the same variant shows only mild delay of puberty or a slightly reduced sense of smell. Incomplete penetrance—where some carriers remain outwardly healthy—is a recurring theme for these genes.
What This Means for Patients and Families
The study supports the idea that CHH and cleft lip or palate are often different faces of the same developmental story, shaped by shared genes such as FGFR1 and CHD7. For families, this means that a child born with a cleft, especially when accompanied by hearing problems, smell loss, or other anomalies, might also be at higher risk for delayed puberty and fertility issues later in life. Conversely, teenagers evaluated for absent puberty may carry genetic changes first discovered in cleft research. Recognizing this overlap can prompt earlier genetic testing, closer hormone monitoring, and more tailored counseling, turning what once seemed like isolated problems into a more understandable—and more manageable—whole.
Citation: Correa, F.d.A., Habibi, I., Zhai, J. et al. Pleiotropic genes linking congenital hypogonadotropic hypogonadism and cleft lip/palate: evidence from a genomic CHH cohort study. Eur J Hum Genet 34, 340–347 (2026). https://doi.org/10.1038/s41431-025-02005-6
Keywords: congenital hypogonadotropic hypogonadism, cleft lip and palate, FGFR1, developmental genetics, oligogenic inheritance