Clear Sky Science · en
Aerosol assisted chemical vapor deposition of cobalt-based co-catalysts on bismuth vanadate-based photoelectrodes for solar water splitting systems
Turning Sunlight and Water into Clean Fuel
Imagine making hydrogen fuel directly from sunlight and water, using a solid panel much like a solar cell. This study explores a new way to build the crucial "helper" layer on such panels, using a method that could be scaled up to cover large surfaces at low cost. By improving how efficiently the panel pulls electrical charges apart and how long it survives in water, the work moves solar-made hydrogen a step closer to practical reality.
A Special Solar Panel for Splitting Water
The core of the device is a light-absorbing material called bismuth vanadate, which behaves a bit like a solar panel but is tailored for water instead of wires. When sunlight hits this layer in contact with water, it creates positive and negative charges that, in principle, can pull water molecules apart into oxygen and hydrogen. In practice, however, many of these charges quickly recombine and vanish as waste heat, and the surface of the material can slowly dissolve. To help, researchers add a thin "co-catalyst" coating on top that encourages the desired reaction and shields the surface from damage.
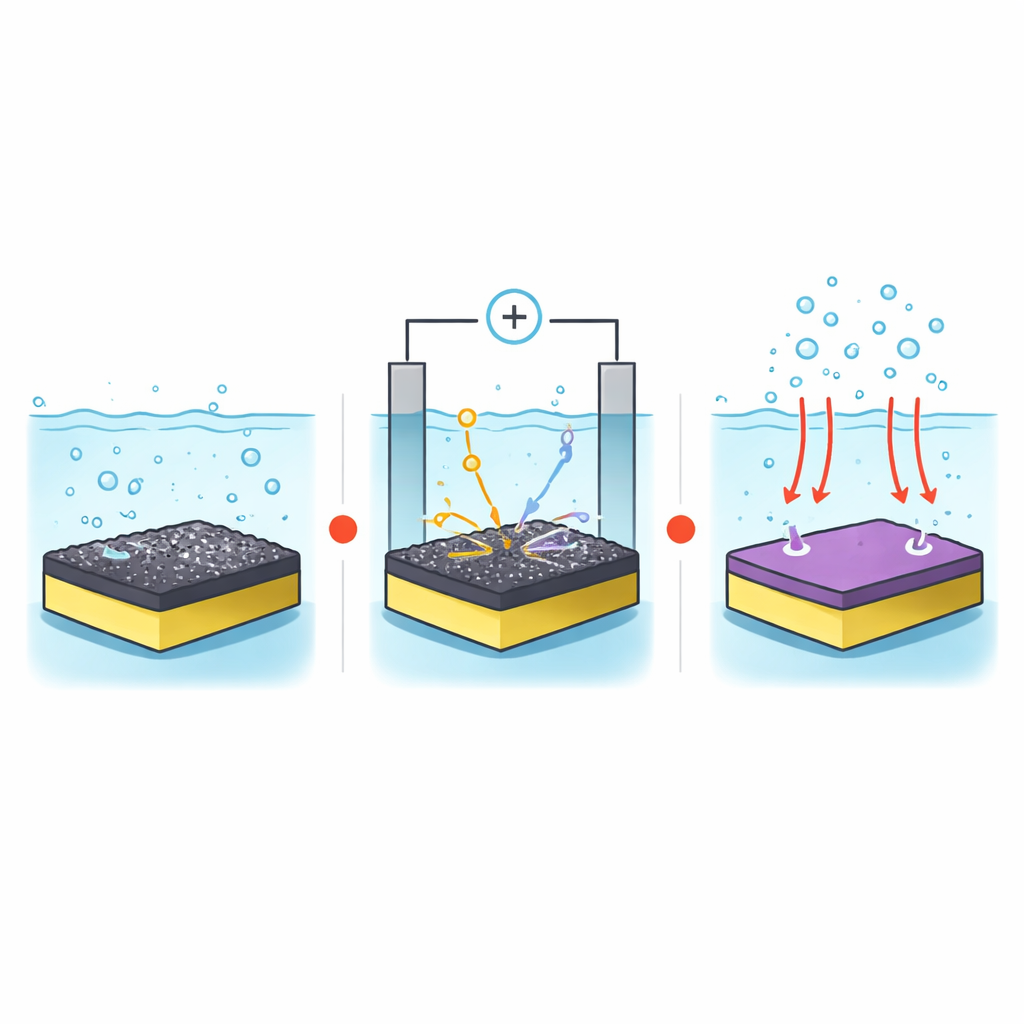
Building the Helper Layer by Spraying from the Air
Traditionally, the cobalt-based co-catalyst layer known as cobalt phosphate is grown in a liquid bath under light and electric bias, a process that is difficult to apply evenly over large areas. In this work, the team instead first deposits a cobalt-oxide film using aerosol-assisted chemical vapor deposition: a fine mist of a cobalt-containing solution is carried in hot air over glass coated with the bismuth vanadate layer, forming a uniform cobalt oxide skin. They then place the coated sample in a phosphate-containing salt solution and apply voltage in the dark, transforming only the outer surface of the cobalt oxide into cobalt phosphate. This two-step "spray then tune" process happens at normal air pressure, making it more compatible with industrial coatings.
How the New Coating Boosts Performance
The researchers compared their sprayed-and-treated cobalt phosphate films with the standard films made entirely by growth in liquid under light. Although the new films contain only a very thin phosphate-rich surface, they adhere more tightly and cover the underlying bismuth vanadate more smoothly. Electrical tests under simulated sunlight showed that the new coating more than doubled the solar-to-hydrogen efficiency of simple bismuth vanadate panels, raising it from 0.21% to 1.16%. It also shifted the voltage at which water splitting begins to lower values and cut the resistance to charge flow at the surface. Measurements of how efficiently incoming light is converted into electric current reveal that the sprayed films improve both the separation of charges inside the panel and the ease with which those charges trigger the oxygen-forming reaction at the surface.
Stability and Advanced Designs
A crucial question for any water-splitting panel is whether it can last. Bare bismuth vanadate electrodes rapidly lost most of their performance over just four hours of operation, as their surface corroded in contact with the electrolyte solution. Panels coated with the new sprayed cobalt phosphate layer retained about 90% of their initial current over the same period and largely recovered their output after resting, suggesting that the coating both speeds the useful reaction and physically protects the underlying material. In contrast, the conventional cobalt phosphate films developed cracks and gaps and eventually failed completely. When the team combined bismuth vanadate with an additional light-absorbing underlayer and then added the cobalt-based coatings, they achieved still higher current and efficiency, showing that the method can integrate into more advanced, multi-layer designs.
Why This Matters for Future Clean Energy
This study demonstrates that a scalable "spray and transform" method can create effective, durable cobalt-based helper layers for solar water-splitting devices. While the absolute efficiencies remain below those needed for commercial hydrogen production, the approach delivers large performance gains, strong medium-term stability, and compatibility with complex electrode structures, all using processes that operate at atmospheric pressure and lend themselves to large glass sheets. For a lay reader, the takeaway is that engineers are learning not just how to make clever materials, but how to manufacture them in a way that could eventually fill rooftops or solar farms with panels that turn sunlight and water directly into clean fuel.
Citation: Huang, M., Creasey, G., Lin, Z. et al. Aerosol assisted chemical vapor deposition of cobalt-based co-catalysts on bismuth vanadate-based photoelectrodes for solar water splitting systems. NPG Asia Mater 18, 8 (2026). https://doi.org/10.1038/s41427-026-00641-y
Keywords: solar water splitting, hydrogen fuel, photoelectrodes, cobalt phosphate catalyst, bismuth vanadate