Clear Sky Science · en
Artificial chaperones: from conventional designs to smart systems
Helping Molecules Fold the Right Way
Inside every living cell, long chains of proteins and genetic material must bend and twist into just the right shapes to work properly. When they misfold, they can clump together, lose their jobs, and even trigger diseases such as neurodegeneration. Nature relies on “chaperone” molecules to keep this microscopic crowd in order, but these natural helpers are expensive, fragile, and hard to use outside cells. This article explores how scientists are now building artificial chaperones—engineered helpers that can coax wayward molecules back into shape and can even be switched on and off like smart tools.
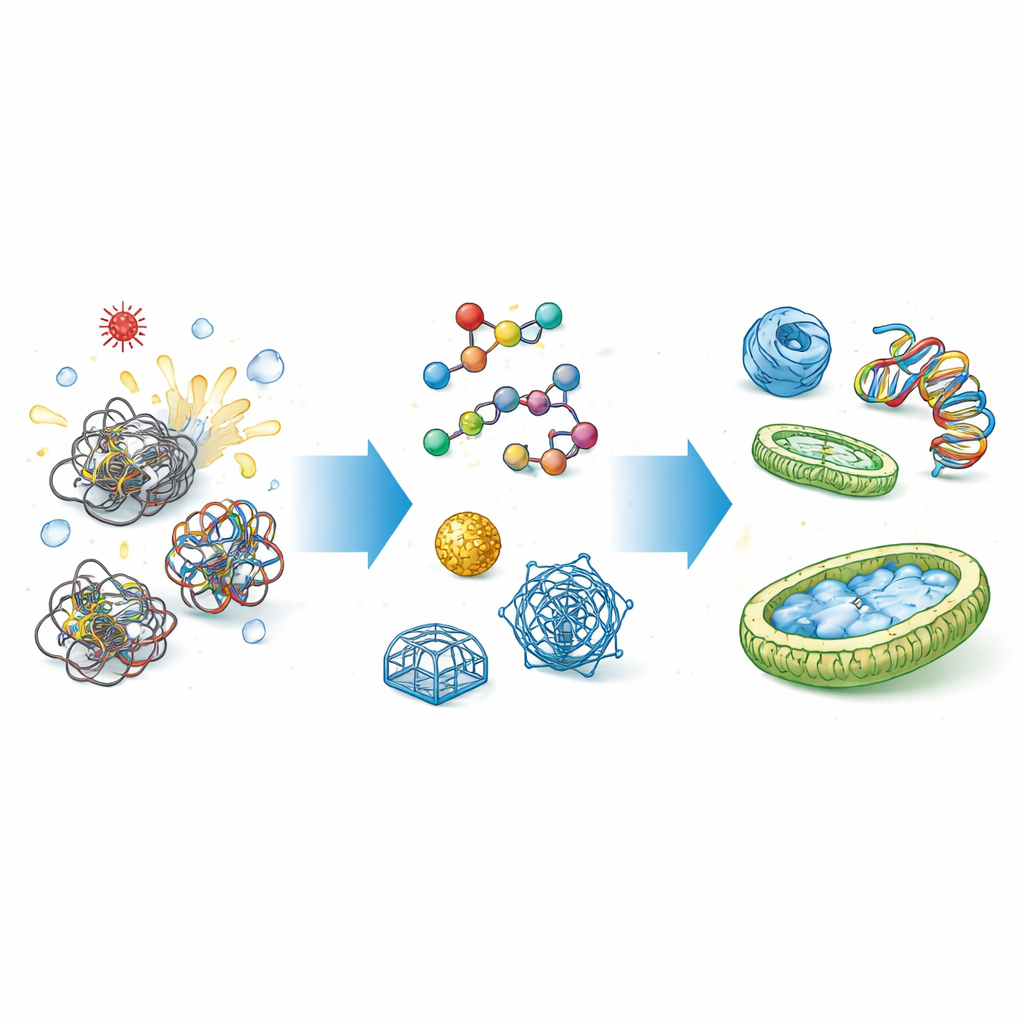
Why Cells Need Folding Helpers
Proteins and nucleic acids (like DNA and RNA) are not born ready-made; they must fold into intricate three-dimensional shapes. The route to those shapes is filled with pitfalls, where chains can get stuck or stick together. Natural chaperones, such as heat-shock proteins, grip these floppy chains, shield their sticky portions, and give them a protected space to fold correctly. Similar guiding roles are played for nucleic acids, where chaperones help unravel knots and rearrange base pairs. When this control fails, the result can be lost function, toxic clumps, disease, and costly failures in industrial protein production.
From Simple Chemicals to Designer Polymers
The first artificial chaperones were relatively simple chemicals that change how molecules feel in their surroundings. Certain solvents and detergents can hide sticky regions on proteins, prevent clumps, and give them a second chance to refold. Some small fatty acids, for example, help mutant collagen fold more correctly and ease stress inside cells affected by inherited disease. But these chemical helpers usually need to be added at high doses, are hard to tune for specific targets, and may disturb other components in complex mixtures. To gain more control, researchers turned to larger, tailor‑made polymers—flexible chains that can be decorated with charged or water‑loving branches to cradle DNA, RNA, and proteins more selectively.
Polymer and Nanogel Toolkits
Designer polymers can latch onto charged backbones of DNA or exposed patches on proteins, smoothing the energy landscape so these molecules find their preferred shapes faster and more reliably. For nucleic acids, cationic (positively charged) polymers can neutralize repulsion between strands, speeding up base pairing, strand exchange, and the formation of exotic structures used in DNA computing and biosensors. For proteins, polymers such as specially shaped polyethylene glycol loops or synthetic polyanions can outperform some natural chaperones in preventing heat‑induced aggregation. Other systems pair flexible chaperone polymers with short peptides to remodel cell‑like membranes into flat nanosheets or vesicles, creating reversible two‑dimensional platforms for cargo delivery and tissue engineering.

Building Smart, Switchable Chaperones
Beyond simply holding and releasing their clients, modern artificial chaperones can now respond to their environment. Researchers have built nanogels, nanoparticles, and polymer networks whose grip on proteins tightens or loosens with changes in acidity, temperature, light, or specific chemicals like glucose. For instance, some micelles become more hydrophobic at mildly acidic pH, mimicking natural chaperones by capturing threatened proteins and then releasing them when the pH changes again. Temperature‑responsive polymers collapse or expand as they cross a threshold, allowing them to sequester damaged proteins at one temperature and release refolded ones at another. Light‑sensitive groups can be used as molecular switches, turning DNA assembly pathways on or off with a flash, while sugar‑responsive particles can protect a therapeutic hormone and then release it preferentially when blood sugar is high.
What This Means for Medicine and Technology
The article concludes that artificial chaperones are evolving from simple chemical stabilizers into highly programmable “molecular tools” that can recognize, protect, and remodel many kinds of biological structures on demand. By carefully balancing attractive and repulsive forces, and by wiring in sensitivity to cues such as pH, heat, light, or mechanical stress, these systems can fold target molecules at the right place and time. In the long run, such smart helpers could make industrial protein production more efficient, sharpen biosensors, power molecular computers, and offer new ways to tackle diseases tied to misfolded proteins—potentially including conditions like Alzheimer’s disease—while closely imitating the subtle control that living cells already achieve.
Citation: Zhang, W., Maruyama, A. Artificial chaperones: from conventional designs to smart systems. NPG Asia Mater 18, 12 (2026). https://doi.org/10.1038/s41427-026-00640-z
Keywords: artificial chaperones, protein folding, smart polymers, nanomedicine, stimuli-responsive materials