Clear Sky Science · en
Superatomic molecules: natural and non-natural atom-like bonding between superatoms
Tiny Building Blocks That Behave Like Atoms
Most of us learn that atoms are the basic building blocks of matter. This review article shows that, at very small scales, groups of dozens of metal atoms can themselves act like “super‑atoms,” and that these superatoms can join together into “superatomic molecules.” By understanding and designing these unusual building blocks, scientists hope to create new materials with finely tuned light, electronic, and catalytic properties that ordinary molecules cannot provide.
Clusters That Imitate Single Atoms
Superatoms are ultra‑small metal clusters—often made of gold, silver, or copper—whose electrons arrange themselves into neat, shell‑like patterns, much like the shells in a textbook diagram of an atom. When the shells are completely filled, at certain “magic numbers” of electrons, the cluster becomes especially stable. Chemists can stabilize these clusters further by wrapping them in organic molecules called ligands, turning them into precise nano‑objects with well‑defined sizes and shapes. Many of these superatoms are nearly spherical, and their stability can be explained by a simple model in which the metal’s free electrons swirl around inside as if in a smooth, positively charged droplet.
When Superatoms Bond Like Ordinary Molecules
Some metal clusters are not spherical at all. Instead, they look like two or more superatoms fused together, forming what the author calls superatomic molecules. To explain these structures, researchers developed “super valence bond” theory, which treats each superatom like a giant atom with its own orbitals, and describes how these orbitals mix to create shared “superatomic molecular orbitals.” In many cases these combinations behave just like familiar chemical bonds. For example, pairs of gold superatoms can form superatomic versions of the bond in the fluorine molecule, while more complex fusions give analogues of oxygen molecules with higher bond orders, or even three‑center bonds reminiscent of ozone. These arrangements create rich patterns of bonding—single, multiple, and multi‑center—that echo the bonding rules of ordinary chemistry, but now at the scale of clusters containing dozens of atoms. 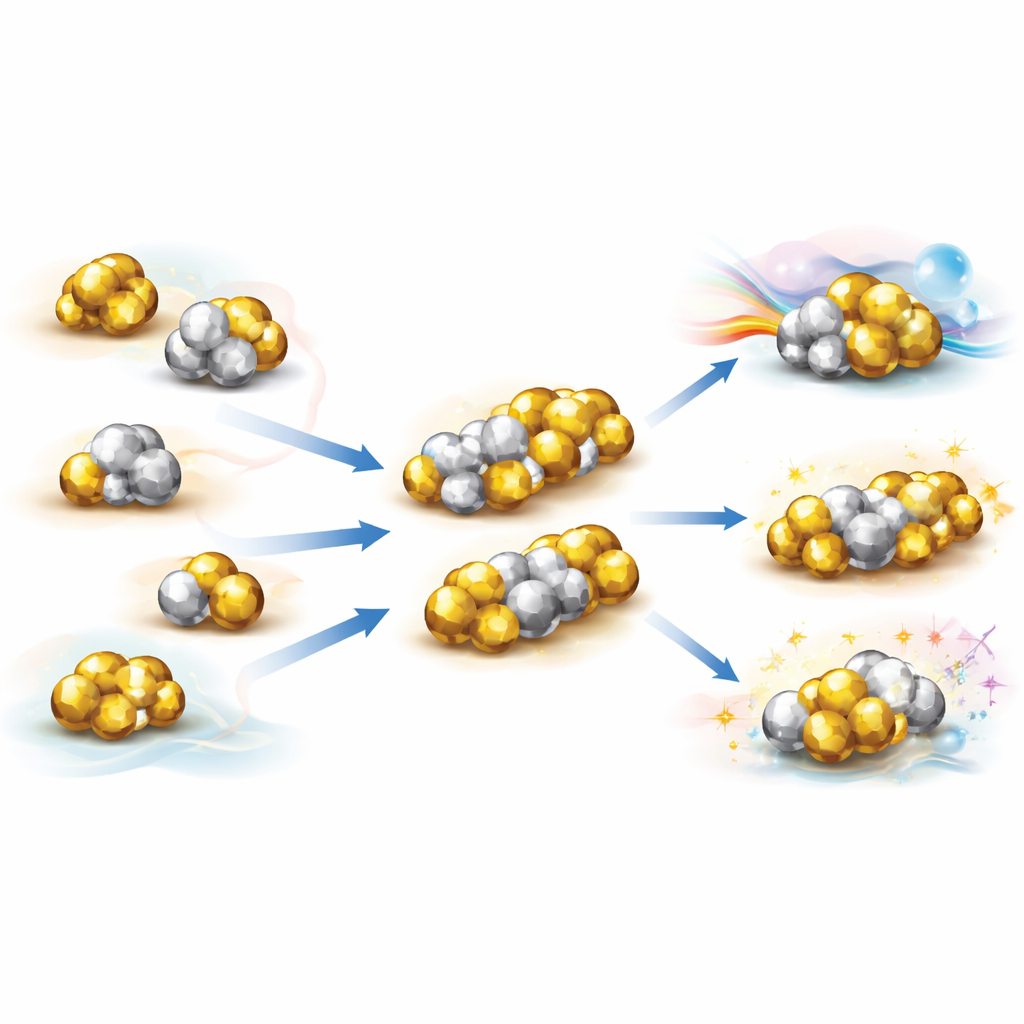
Strange Bonds With Noble‑Gas‑Like Units
Superatomic molecules are not limited to neat analogies with everyday bonds. The review highlights “non‑natural” bonding motifs where superatoms with closed electronic shells—similar in spirit to noble gases like helium or neon—combine into larger structures even though, by standard counting rules, there should be no bond at all. In these systems, superatoms may share a single metal atom, sit edge‑to‑edge, or link into cyclic and rod‑like assemblies. Formally, their bond order is zero, yet interactions between their electron clouds and the surrounding ligands stabilize the overall structure. Remarkably, these assemblies show new absorption bands and other electronic features that are absent in the isolated superatoms, revealing that subtle orbital interactions can produce entirely new optical responses.
From Super Rings to Super Chains
The article also examines more exotic examples where superatoms form rings and chains with collective behavior. One notable case is a gold cluster built from five icosahedral superatoms arranged in a ring; theoretical studies suggest that, with the right number of electrons, this “super‑ring” can become aromatic, spreading its electrons around the loop in a way directly analogous to classic aromatic molecules such as benzene or the cyclopentadienyl ion. Another family of structures links tiny three‑atom gold units into rod‑shaped chains. Even though the connections between neighboring units are weak and partly antibonding, the repeating pattern of interacting orbitals acts like a “superatomic polymer,” giving rise to strong absorption in the near‑infrared region and pointing toward applications in photothermal and optoelectronic devices. 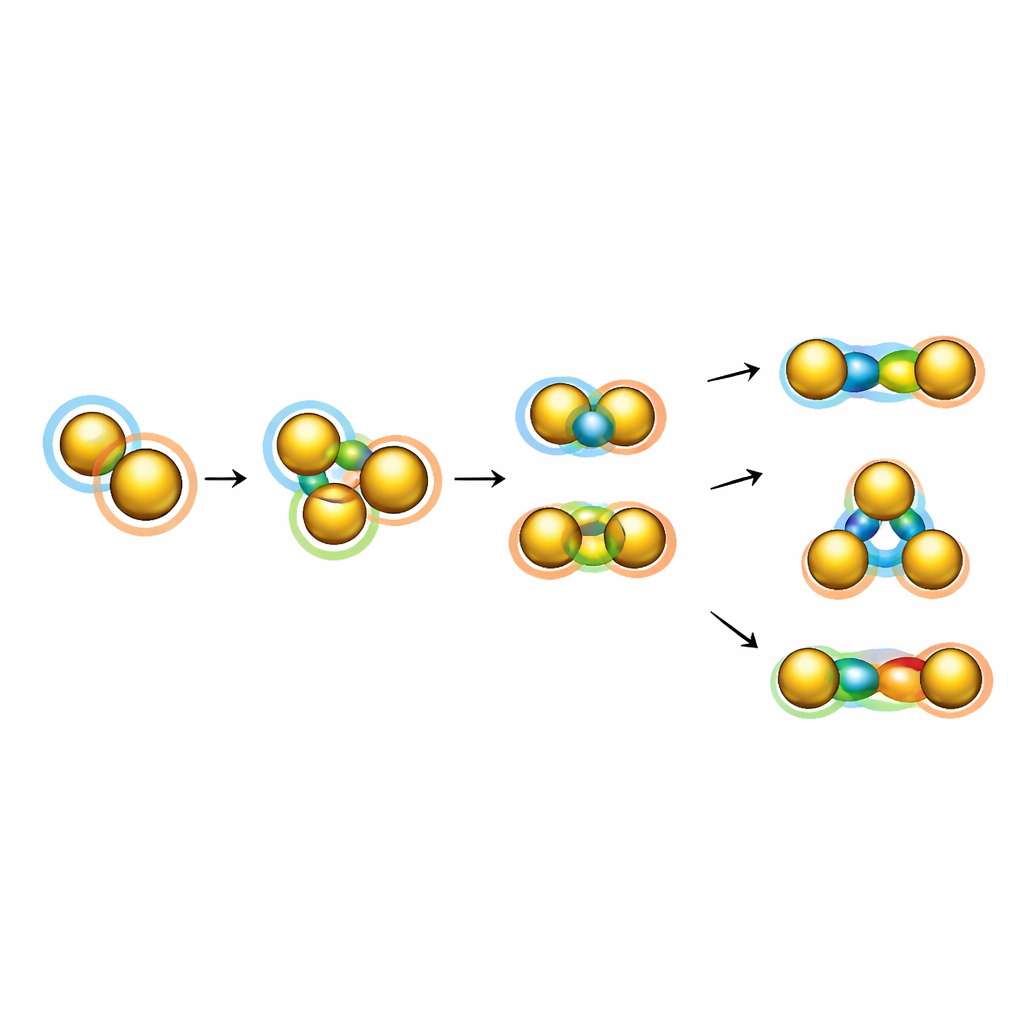
Why These Tiny Super‑Molecules Matter
Overall, the review argues that treating metal clusters as atom‑like units that can bond into superatomic molecules provides a powerful design language for new materials. By choosing how many electrons each superatom carries, how they are linked, and which ligands surround them, scientists can engineer not only stability but also light absorption, charge transport, magnetism, and catalytic activity. Natural‑style bonding gives familiar design rules, while non‑natural and weakly bonded assemblies unlock behaviors with no direct analogue in ordinary chemistry. As this understanding matures, superatomic molecules may become a toolbox for crafting next‑generation catalysts, light‑harvesting systems, and electronic components built from precisely arranged clusters of atoms.
Citation: Isozaki, K. Superatomic molecules: natural and non-natural atom-like bonding between superatoms. NPG Asia Mater 18, 9 (2026). https://doi.org/10.1038/s41427-026-00636-9
Keywords: superatoms, metal nanoclusters, superatomic molecules, nanomaterials, optoelectronic properties