Clear Sky Science · en
Oncolytic virus M1 reinvigorates CD8+ T-cell immunity against glioblastoma through B-cell-dependent antigen cross-presentation in the spleen
Turning the Immune System Back On
Glioblastoma is one of the deadliest brain cancers, in part because it is remarkably good at switching off the body’s defenses. This study explores a new way to flip those defenses back on using a specially designed virus, delivered through the bloodstream, that both attacks tumor cells and rallies immune cells to fight the cancer. The work reveals an unexpected star player in this response: immune cells in the spleen that help awaken tumor-killing T cells.
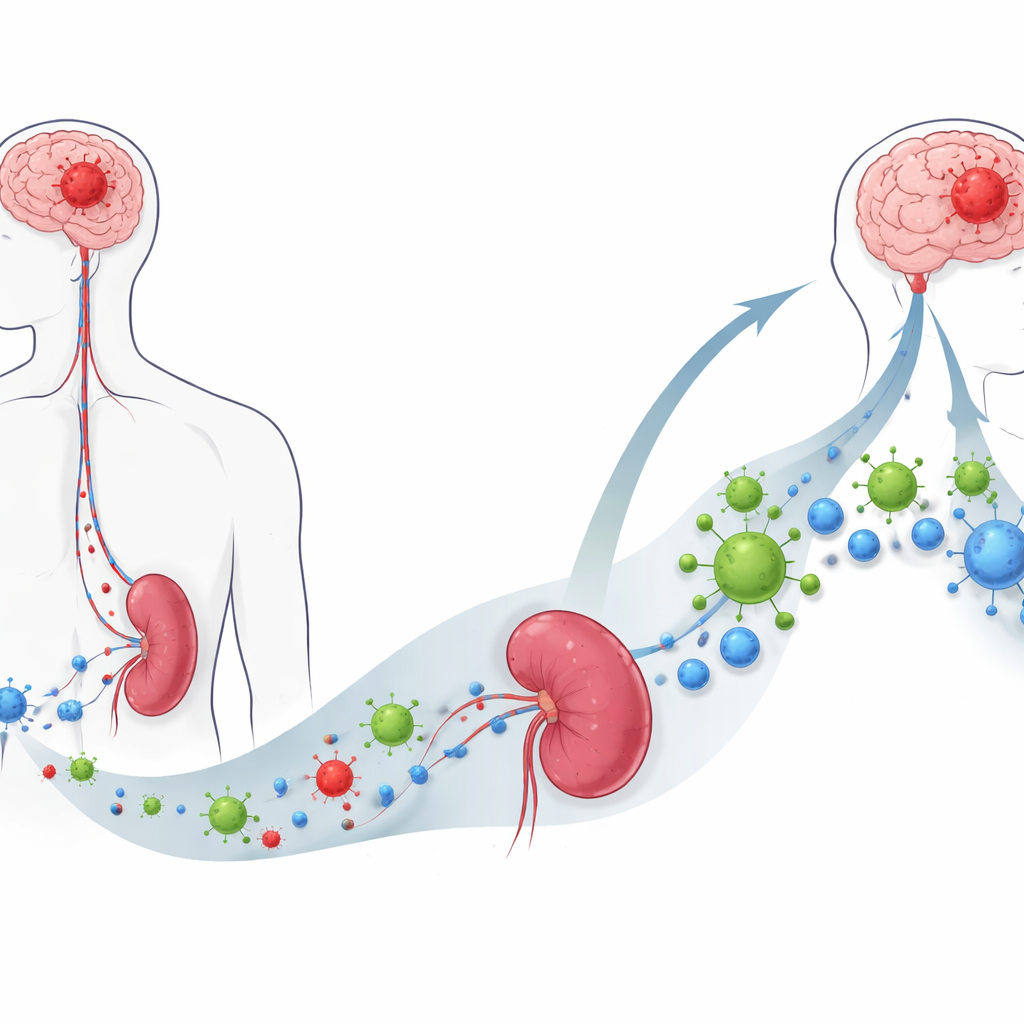
A Virus That Prefers Cancer Cells
The researchers studied an experimental therapy called oncolytic virus M1, or OVM, a modified alpha virus that prefers to infect and kill cancer cells while sparing normal tissue. In lab dishes, OVM efficiently invaded a range of mouse and human glioblastoma cell lines and drove them into a form of cell death that leaves behind molecular “danger signals.” When tested on freshly removed human tumor fragments, OVM replicated strongly in glioblastoma tissue but not in neighboring noncancerous brain samples, suggesting it can distinguish malignant from healthy cells better than standard chemotherapy, which damaged both.
Reversing Whole-Body Immune Shutdown
Beyond the brain, glioblastoma causes widespread immune collapse: patients and mouse models show shrunken spleens and a sharp loss of circulating T cells. After intravenous OVM treatment in tumor-bearing mice, this picture changed dramatically. Spleens regained their size, blood levels of CD4 and CD8 T cells rebounded, and immune-dampening molecules such as IL-10 and TGF-beta dropped. At the tumor site, more T cells—especially CD8 “killer” T cells that recognize a model tumor antigen—moved into the brain, turning a previously “cold” tumor environment into one packed with immune fighters.
Why the Spleen Matters
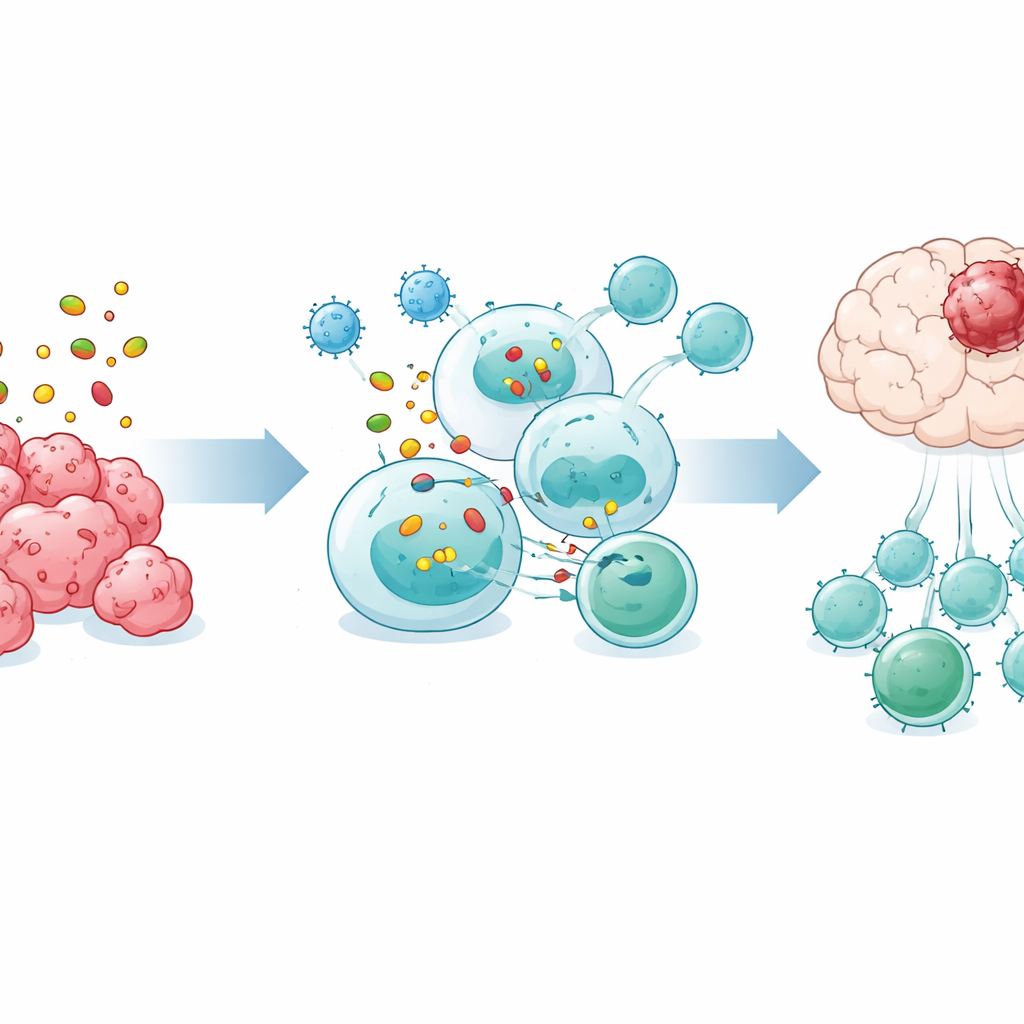
Surprisingly, the spleen turned out to be essential for OVM’s success. When the spleen was surgically removed, the virus no longer slowed tumor growth or extended survival, and the surge in T cells in blood and brain largely disappeared. Single-cell RNA sequencing of splenic immune cells showed that after OVM treatment, B cells in the spleen formed especially strong communication networks with T cells. In dishes, B cells from OVM-treated mice made CD8 T cells proliferate and activate far more effectively than B cells from untreated animals, whereas dendritic cells did not show the same boost, pointing to B cells as key intermediaries.
Special B Cells That Prime Tumor Killers
Diving deeper, the team discovered a distinct B-cell subgroup marked by a surface molecule called Bst2 that expanded in the spleen after OVM treatment. These Bst2-positive B cells were enriched for genes involved in taking up tumor material, processing it inside the cell, and displaying fragments on their surface in a way that CD8 T cells can “see.” Experiments showed that these cells captured tumor antigens more efficiently, formed tight contact zones with CD8 T cells, and required direct cell–cell contact and a specific protein-processing pathway to activate T cells. When purified Bst2-positive B cells were transferred into B-cell–deficient mice with brain tumors, they significantly prolonged survival, whereas other B-cell types did not.
Boosting the Power of Checkpoint Therapy
Today’s leading immunotherapies—checkpoint inhibitors that block PD-1 or PD-L1—have had disappointing results in glioblastoma. In the mouse models used here, PD-1 or PD-L1 antibodies alone did little. However, combining OVM with a PD-1–blocking antibody substantially increased the number of CD8 T cells in the brain tumors, shifted circulating T cells from a naïve to an activated state, restored spleen size, and extended survival. OVM treatment also raised PD-1 levels on tumor-infiltrating CD8 T cells, making them more susceptible to rescue by PD-1 blockade.
What This Could Mean for Patients
Taken together, this work suggests that an intravenously delivered oncolytic virus can do more than simply infect and lyse glioblastoma cells. By recruiting a specialized group of B cells in the spleen to process tumor material and show it to CD8 T cells, OVM rebuilds systemic immunity and drives killer T cells back into the brain to attack the tumor. In preclinical models, this strategy not only suppresses tumor growth on its own but also makes resistant brain tumors respond to PD-1 checkpoint therapy. If these findings translate to humans, OVM and Bst2-positive B cells could form the backbone of new combination treatments—and even personalized vaccines—that finally give the immune system a fighting chance against glioblastoma.
Citation: Han, Y., Guo, C., Chen, C. et al. Oncolytic virus M1 reinvigorates CD8+ T-cell immunity against glioblastoma through B-cell-dependent antigen cross-presentation in the spleen. Cell Mol Immunol 23, 349–366 (2026). https://doi.org/10.1038/s41423-026-01396-w
Keywords: glioblastoma, oncolytic virus, tumor immunology, B cells, immune checkpoint therapy