Clear Sky Science · en
Tackling cuproptosis: from metabolic rewiring to therapeutic exploitation in cancer
When a Helpful Metal Turns Against Cancer
Copper is best known as a component of electrical wire and plumbing, but it also runs quietly through our cells, helping enzymes make energy and protect us from damage. This review article explores a surprising twist: under the right conditions, too much copper can push cancer cells into a special kind of death. Understanding this process—called “cuproptosis”—could open up new ways to treat tumors that have outsmarted standard therapies.
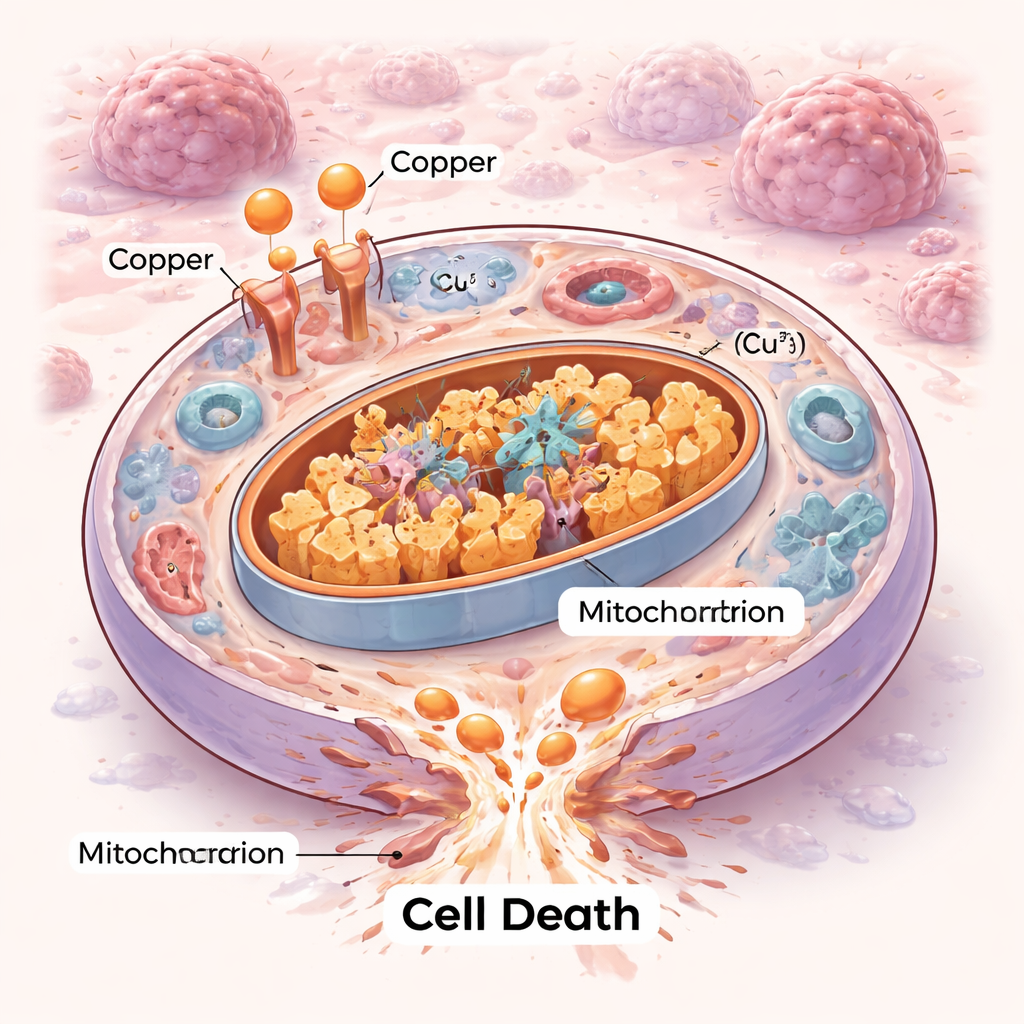
A New Way Cells Can Die
Cells do not all die the same way. For decades, scientists have mapped out orderly self‑destruct programs like apoptosis and newer forms such as ferroptosis. Cuproptosis adds a fresh chapter. Here, excess copper floods a cell’s power stations—the mitochondria—and binds to specific metabolic enzymes that normally help burn fuel. This binding causes those enzymes to clump together and destabilizes tiny iron–sulfur components the mitochondria need to function. The result is a kind of internal traffic jam and mechanical breakdown that the cell cannot repair, ending in death by proteotoxic stress rather than by the classic suicide routes.
How Cancer Cells Control Copper and Energy
Because copper is both essential and dangerous, cells maintain an elaborate logistics network to move it safely. Dedicated transporters bring copper into the cell, chaperone proteins escort it to mitochondria and other destinations, and storage molecules like metallothioneins and glutathione mop up any excess. Tumors often rewire this system. Some boost importers or chaperones to feed their high energy demands; others increase exporters or storage proteins to avoid copper overload. At the same time, many cancers shift their metabolism— toggling between sugar‑burning at the cell surface and deeper mitochondrial respiration. The review explains that cuproptosis strikes hardest in cells that still depend heavily on mitochondria, making tumors with this metabolic profile especially vulnerable.
Master Switches That Tune Copper Sensitivity
Major cellular “decision‑makers” influence whether cuproptosis can occur. The tumor suppressor p53, for example, tends to push cells away from fast sugar fermentation toward more orderly mitochondrial fuel use. By doing so, and by altering the handling of copper‑binding enzymes, p53 can make cancers more susceptible to copper‑induced death—while mutant forms of p53 often do the opposite. In contrast, the hypoxia factor HIF‑1α, activated in poorly oxygenated tumor cores, helps cells escape cuproptosis by dialing down key mitochondrial enzymes and boosting copper‑binding shields. Other pathways, such as Wnt/β‑catenin and AKT, promote resistance by driving copper export or chemically modifying crucial proteins so they no longer respond to copper overload. Together, these networks act like thermostats, raising or lowering a tumor’s sensitivity to copper‑based treatment.
Copper, the Immune System, and Smarter Drug Delivery
Copper’s role is not limited to killing cancer cells directly; it also shapes the immune landscape around tumors. Controlled copper stress can make dying cancer cells more “visible,” releasing danger signals that recruit and activate T cells, dendritic cells, and macrophages. Copper can also influence the levels of immune checkpoints like PD‑L1, which tumors use to turn off anti‑cancer T cells, suggesting chances to combine copper‑targeting drugs with modern immunotherapies. Because free copper can harm healthy tissues, researchers are building precision tools—small‑molecule ionophores that chaperone copper selectively into cancer cells, and nanomedicine platforms that pack copper or copper‑driven drugs inside targeted particles or hydrogels. These technologies aim to raise copper to lethal levels inside tumors while keeping the rest of the body in a safe range.
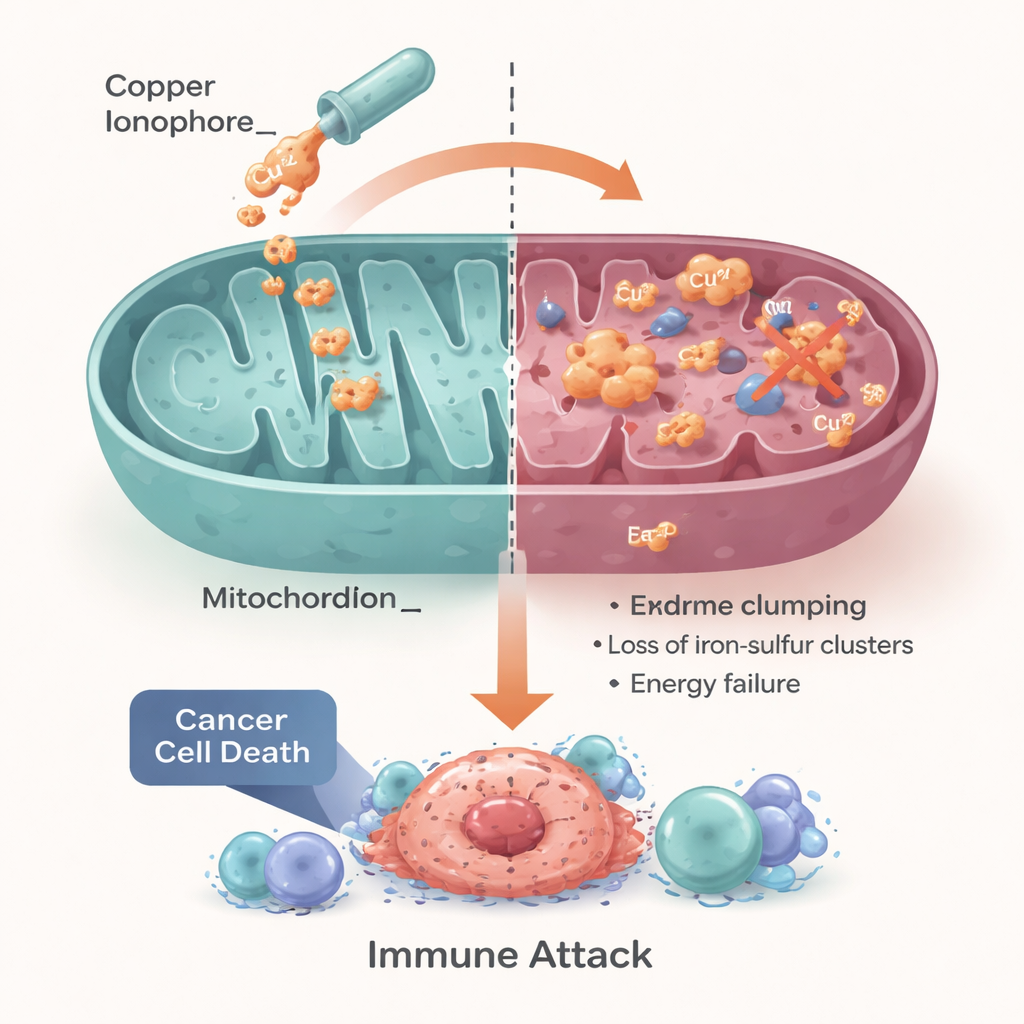
Turning a Cellular Weakness into a Treatment
The authors conclude that cuproptosis turns a basic cellular need—careful copper and energy management—into a potential Achilles’ heel for cancer. Cancers with disturbed copper handling or an overreliance on mitochondrial metabolism may be especially sensitive to copper‑based strategies, including repurposed drugs like disulfiram and elesclomol, as well as next‑generation nanoparticles. However, they stress that success will depend on matching the right patients to the right approach, using biomarkers that report on copper transporters, mitochondrial activity, and the tumor’s immune and oxygen status. If these hurdles can be cleared, harnessing cuproptosis could give oncologists a new, metal‑powered lever to treat tumors that currently have few good options.
Citation: Hao, Q., Gan, Y. & Zhou, X. Tackling cuproptosis: from metabolic rewiring to therapeutic exploitation in cancer. Cell Mol Immunol 23, 239–260 (2026). https://doi.org/10.1038/s41423-026-01387-x
Keywords: cuproptosis, copper metabolism, cancer therapy, tumor metabolism, cancer immunotherapy