Clear Sky Science · en
Dynamic magneto-mechanical force in lysosomes induces durable macrophage repolarization for antitumor immunity
Using Gentle Forces to Wake Up the Immune System
Cancer often survives by turning the body’s defenses to sleep. This study explores an unexpected way to wake them up again: by physically tugging on tiny compartments inside immune cells with magnetically driven nanoparticles. Instead of relying only on drugs, the researchers use carefully tuned mechanical forces inside cells to reprogram immune cells called macrophages into long‑lasting tumor fighters.
Why Solid Tumors Are Hard to Treat
Modern cancer immunotherapies, such as checkpoint‑blocking antibodies and engineered immune cells, have transformed treatment for some blood cancers. Yet for many people with solid tumors like lung cancer, these approaches help only a minority. One major reason is the tumor microenvironment. Tumors are surrounded by a protective niche filled with cells and signals that dampen immunity. Macrophages, which can either attack tumors (an M1‑like state) or support them (an M2‑like state), are often pushed into the tumor‑aiding M2 mode. Reversing this “bad” programming in a durable way has been difficult using conventional biochemical drugs alone.
Turning Nanoparticles into Tiny Mechanical Motors
The team designed magnetic nanomotors—nanoscale particles of zinc‑doped iron oxide coated with a positively charged polymer—that are readily swallowed by macrophages and end up concentrated in lysosomes, the cell’s recycling and signaling hubs. Once inside, an external rotating magnetic field makes these particles line up into rod‑like chains and spin, stirring the thick fluid within lysosomes and creating tiny vortices. Computer simulations and model membrane experiments showed that by carefully tuning the rotation frequency, the shear stress on the lysosomal membrane can be set to a sweet spot: strong enough to make the membrane briefly leaky, but not so strong that it is permanently destroyed.
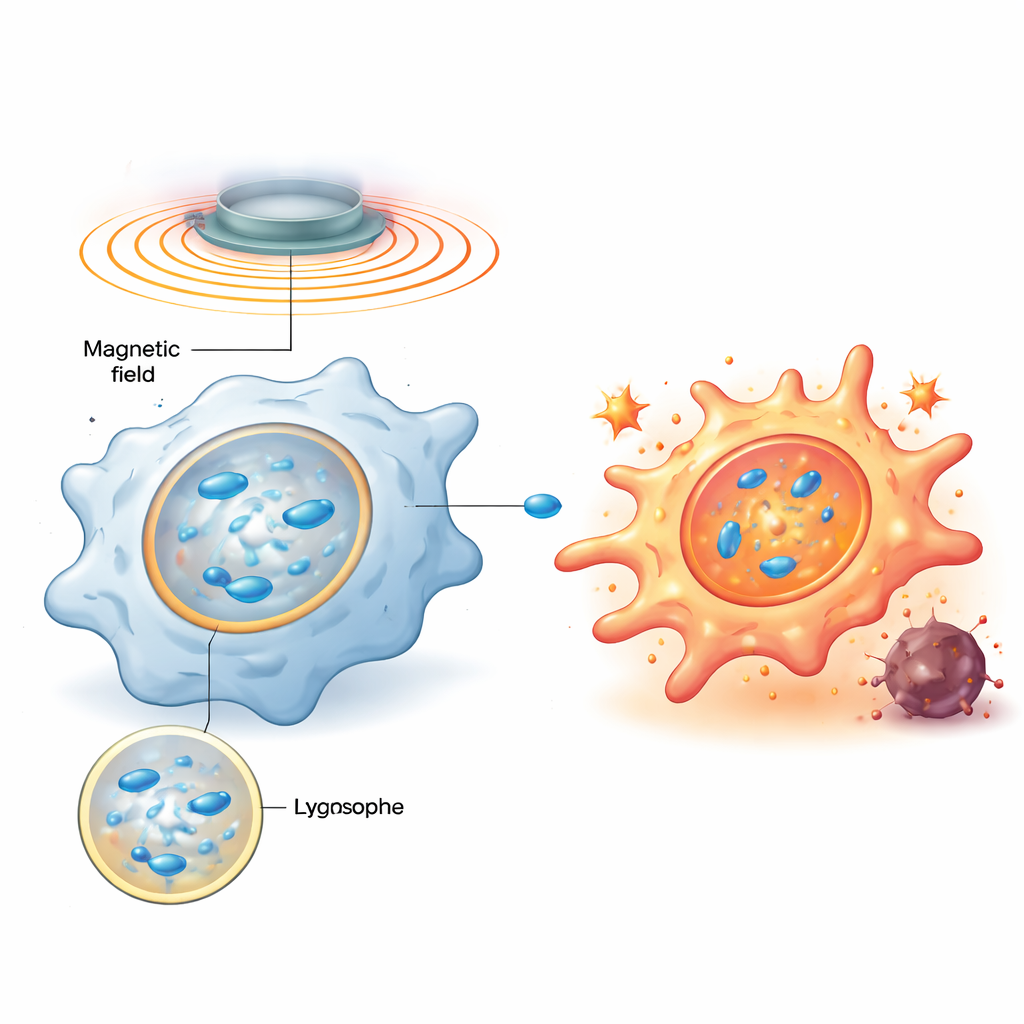
Mechanical Nudge Triggers a Biochemical Alarm
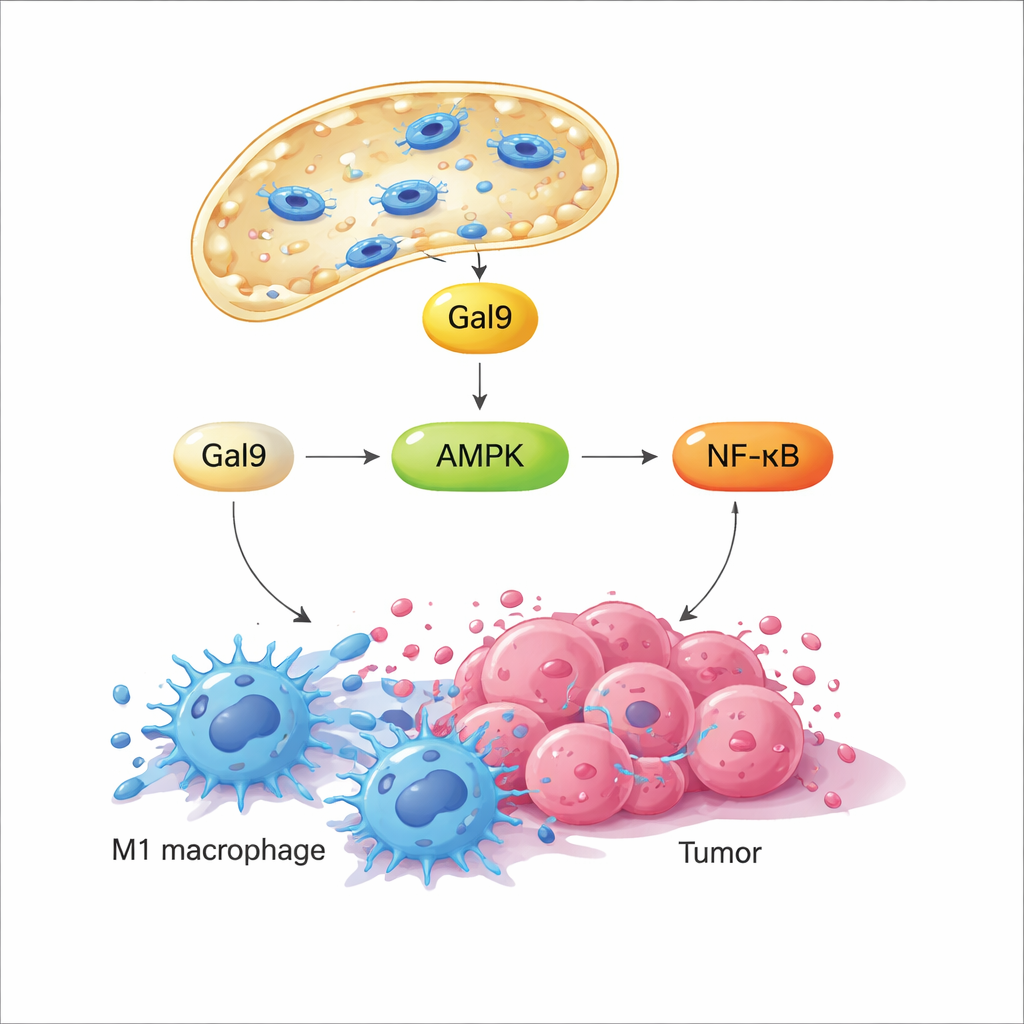
When the lysosomal membrane is briefly disturbed, a sugar‑binding protein called galectin‑9 rushes to the damaged spots, acting as a sensor for this mechanical stress. The study shows that this, in turn, activates a key energy‑sensing enzyme, AMPK, which then boosts the NF‑κB signaling pathway—well known for driving inflammatory and antimicrobial responses. Together, these signals rewire macrophage metabolism away from a quiet, fuel‑efficient state and toward fast glycolysis, a pattern linked with hostile, M1‑like behavior. Importantly, when the magnetic field is turned off, the lysosome repairs itself and signaling subsides; when the field is turned back on later, the same pathway is re‑activated. This programmable on‑off cycling lets researchers repeatedly “tap” the same mechanical switch without killing the cells.
Re‑Educating Macrophages to Fight Tumors
In cell cultures, this magnetically induced lysosomal permeabilization—termed MagLMP—converted macrophages pushed into a tumor‑supporting M2‑like state back into a pro‑inflammatory M1‑like state. These reprogrammed cells produced more inflammatory cytokines and retained their M1‑like profile for days, even in tumor‑derived culture media that usually drives them toward M2. In mouse tumor models, embedding magnetic nanomotors into tumors and applying daily cycles of the rotating field slowed growth of several cancer types. When macrophages were experimentally depleted, the benefit largely disappeared, showing that these cells are central to the treatment’s effect. Single‑cell RNA sequencing of tumor tissue further revealed that MagLMP increased the proportion of M1‑like macrophages, boosted antitumor neutrophils and effector CD8 T cells, and upregulated NF‑κB‑related and glycolysis genes within macrophages.
From Local Control to Systemic Therapy
The researchers then asked whether this approach could work more like a real‑world treatment, delivered through the bloodstream. They injected the nanomotors intravenously and used a simple external magnet to concentrate them within tumors, demonstrating efficient uptake by macrophages in the tumor bed. Subsequent MagLMP treatment again shifted macrophages toward the M1‑like state and curbed tumor growth. In a mouse model of early‑stage lung cancer growing within the lung itself, combining magnetic guidance with cyclic MagLMP led to markedly prolonged survival: about one‑third of treated mice lived more than 300 days, compared with only weeks in control animals. Combining MagLMP with a PD‑1 checkpoint inhibitor further enhanced tumor control in difficult‑to‑treat models.
A New Way to Control Immunity from the Inside Out
This work shows that tiny, well‑controlled mechanical forces inside a specific organelle can be harnessed to steer immune behavior in living animals. By repeatedly and reversibly “poking” lysosomes, MagLMP activates a galectin‑9–AMPK–NF‑κB axis that reprograms macrophages into durable tumor fighters without broadly damaging cells. For non‑specialists, the key idea is that physical cues—delivered by magnetically driven nanomotors—can act like a dial on the immune system, offering a new class of tools to complement drugs in future cancer immunotherapies.
Citation: Li, Y., Zheng, M., Zhu, Z. et al. Dynamic magneto-mechanical force in lysosomes induces durable macrophage repolarization for antitumor immunity. Cell Res 36, 197–218 (2026). https://doi.org/10.1038/s41422-025-01217-1
Keywords: cancer immunotherapy, macrophage reprogramming, magnetic nanoparticles, mechanotransduction, lysosome signaling