Clear Sky Science · en
Transient mechanical activation of the Piezo1 channel facilitates ex vivo expansion of hematopoietic stem cells
Making More Lifesaving Blood Stem Cells
Bone marrow transplants can cure certain blood cancers and inherited disorders, but doctors are often limited by how few true blood-forming stem cells they can obtain from a donor. This study reveals a surprising way to grow these rare cells outside the body by gently “touching” them with tiny plastic beads that nudge a mechanical sensor on their surface. The work suggests a new, potentially safer route to generating enough stem cells for more patients to benefit from transplantation.
Why Growing Blood Stem Cells Is So Hard
Blood stem cells sit deep inside our bones and quietly supply all of our red cells, white cells, and platelets for a lifetime. Transplanting these cells can reset a damaged blood system, as in leukemia treatment, but only if enough healthy stem cells are available and they remain fully functional. Efforts to expand them in laboratory dishes have often fallen short: the cells tend to either stop renewing themselves or lose their long-term ability to rebuild the blood system. Most current methods rely on cocktails of growth factors and genetic tweaks, but they do not fully recreate the complex physical environment of the bone marrow, where stem cells constantly feel pushes, pulls, and pressure.
A Hidden Mechanical Switch on Stem Cells
The researchers focused on a protein called Piezo1, a tiny valve-like channel in the cell membrane that opens when the membrane is bent or stretched, letting calcium ions rush in and start internal signals. By examining many blood cell types, they found that Piezo1 is especially abundant in true blood stem cells. When they removed Piezo1 genetically, or blocked it with a drug, stem cells grown in dishes could no longer expand well or repopulate the blood of mice after transplantation. Interestingly, stimulating Piezo1 continuously with chemical activators was also harmful: the cells accumulated excessive calcium, produced damaging reactive molecules, and were less able to engraft. These results suggested that stem cells need brief, well-timed mechanical activation of Piezo1, not constant chemical prodding.
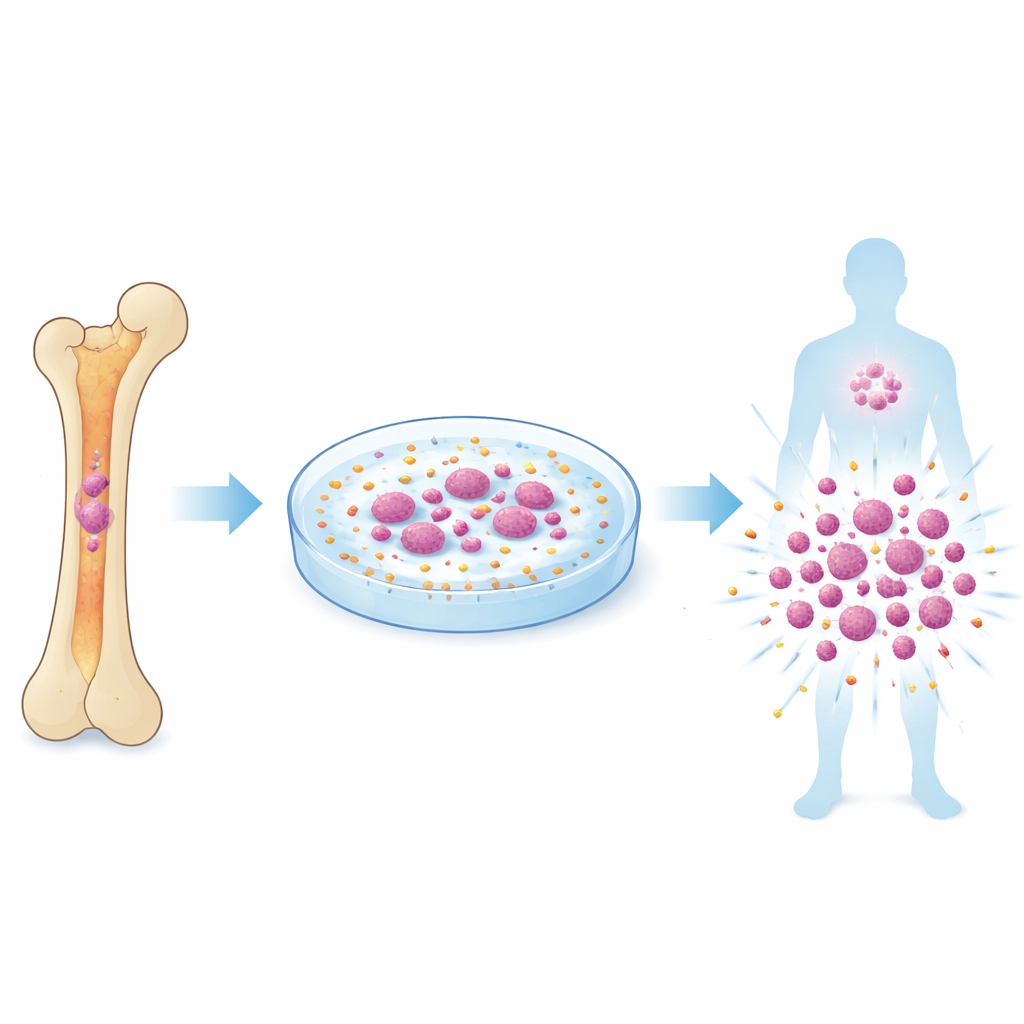
Harnessing Tiny Beads to Tap the Sensor
To provide just the right mechanical nudge, the team turned to microscopic polymer spheres, essentially smooth plastic beads, that could bump against stem cells in culture. They tested beads of different sizes and stiffness and discovered that 500-nanometer polystyrene spheres—called PS500—were remarkably effective. When mouse bone marrow cells were grown together with these beads, the number of stem cells increased dramatically, and these cells maintained strong long-term repopulating ability in serial transplants across multiple generations of mice. Careful imaging and mechanical measurements showed that the beads repeatedly indented the stem cell membrane with tiny forces, enough to open Piezo1 channels in short bursts without overwhelming the cells.
From Mechanical Touch to Growth Signals
The brief opening of Piezo1 channels triggered small pulses of calcium entry into the stem cells. This, in turn, switched on internal pathways that led the cells to produce certain signaling proteins, notably the growth factor interleukin-6. Interleukin-6 then fed back on the stem cells and activated a protein called Stat3 in their nuclei, which is known to support stem cell survival and self-renewal. When the researchers blocked this Stat3 pathway, the beneficial effect of the beads vanished, confirming that this chain of events—mechanical touch, calcium pulse, interleukin-6 release, and Stat3 activation—was crucial for expansion. Importantly, after only a short period of exposure to the beads, the signaling stayed active and stem cell numbers continued to rise, underscoring that constant stimulation was not necessary.
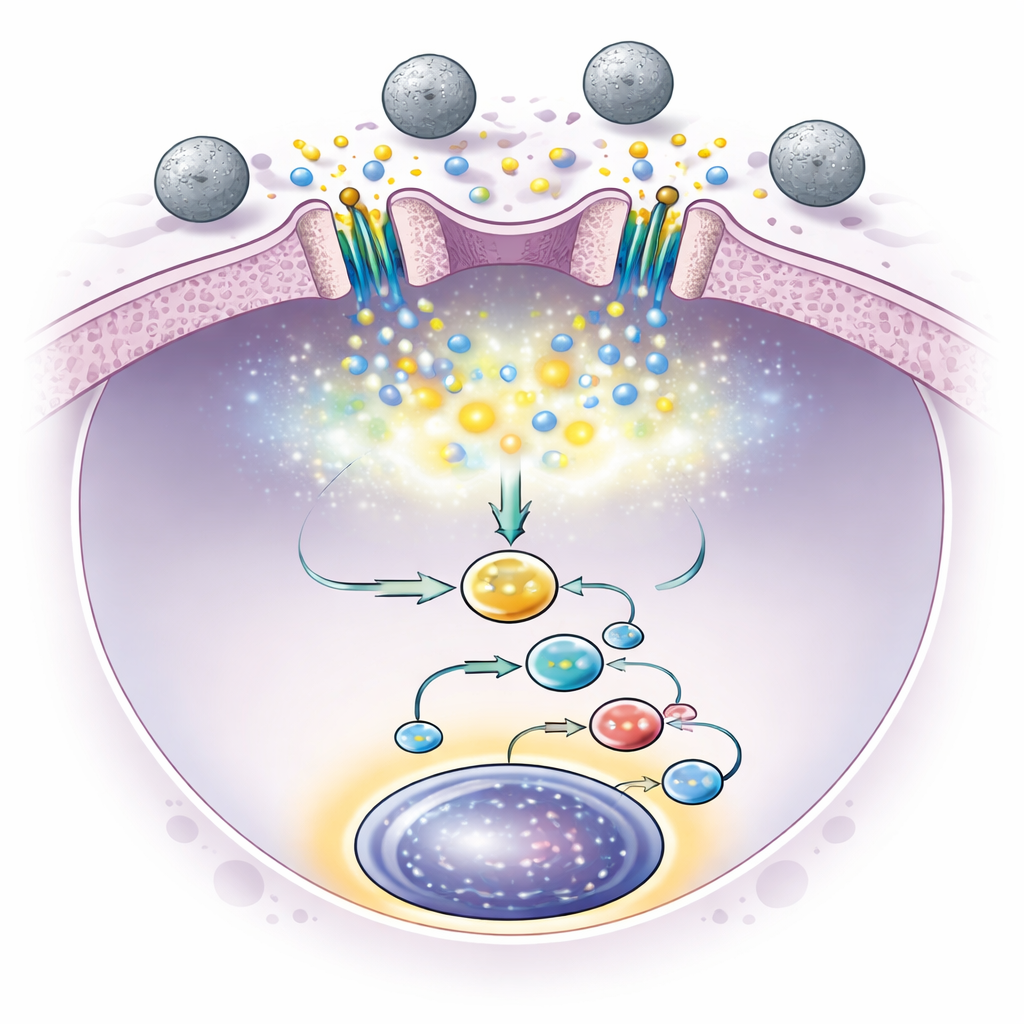
Promise for Human Transplants
The team next applied the same approach to human blood stem cells collected from umbilical cord blood, a common but often limited source for transplantation. With PS500 beads, these human cells expanded several times more than with leading small-molecule methods alone, yet preserved their ability to permanently repopulate the blood of immunodeficient mice. Safety studies in mice suggested that the beads do not enter the stem cells, can be efficiently removed by simple centrifugation or filtration, and do not cause obvious organ damage or blood abnormalities at tested doses. This positions the bead-based system as a practical and potentially scalable technology for blood banks and transplant centers.
What This Could Mean for Patients
In essence, the study shows that a gentle mechanical cue—delivered by drifting nanoscale beads—can coax blood stem cells to multiply while preserving their potency, by briefly activating a built-in pressure sensor rather than overwhelming it. If translated to the clinic, this approach could make it easier to generate enough high-quality stem cells from a small cord blood unit or a partial donor, expanding access to curative transplants for many more people. It also highlights how paying attention not just to the chemical “soup” around cells but also to the physical sensations they experience can unlock new ways to control cell fate.
Citation: Wang, Q., Zeng, X., Yang, H. et al. Transient mechanical activation of the Piezo1 channel facilitates ex vivo expansion of hematopoietic stem cells. Cell Res 36, 272–285 (2026). https://doi.org/10.1038/s41422-025-01209-1
Keywords: hematopoietic stem cells, mechanosensitive ion channels, Piezo1, polystyrene microspheres, stem cell transplantation