Clear Sky Science · en
The mitochondrial translocation of phosphorylated EZH2 promotes PARP inhibitor resistance in BRCA1-deficient epithelial ovarian cancer
Why Some Ovarian Cancers Outsmart Targeted Drugs
Targeted drugs called PARP inhibitors have transformed treatment for many women whose ovarian tumors carry defects in the BRCA1 gene. Yet, after an initial response, cancers often return, now able to shrug off the very medicines that once held them in check. This study uncovers a surprising escape route deep inside the cell’s power plants, offering a new way to predict and potentially reverse this dangerous drug resistance.
When a Precision Drug Stops Working
BRCA1-deficient epithelial ovarian cancer relies on faulty DNA repair, which makes these tumors especially vulnerable to PARP inhibitors. By further crippling DNA repair, PARP inhibitors push cancer cells toward self-destruction. However, many patients eventually relapse with tumors that no longer respond. Known resistance mechanisms, such as mutations that restore BRCA1 function, explain only a fraction of cases. The authors set out to find new drivers of resistance using patient-derived organoids—three-dimensional mini-tumors grown from patients’ own cancer cells that closely mimic the original disease.
Finding a Hidden Culprit in Drug-Resistant Tumors
Screening a panel of epigenetic drugs on PARP inhibitor–resistant organoids, the researchers identified one standout: an inhibitor of EZH2, a protein best known for controlling which genes are switched on or off. Blocking EZH2’s enzyme activity resensitized resistant organoids and cell lines to PARP inhibitors, and tumors with reduced EZH2 grew more slowly in mice receiving these drugs. Surprisingly, classic gene-silencing marks controlled by EZH2 did not change between sensitive and resistant cells, suggesting that EZH2 was driving resistance through a different, nontraditional role.
A Detour from the Nucleus to the Cell’s Power Plants
Diving deeper, the team discovered that in resistant cells, EZH2 becomes chemically modified on a specific building block (tyrosine 728) by a kinase protein called YES1. This modification changes EZH2’s behavior. Instead of staying in the nucleus to regulate DNA, the modified EZH2 accumulates in the cytoplasm and then relocates to mitochondria, the energy-producing structures often called the cell’s power plants. Resistant cells showed more interconnected, elongated mitochondria—signs of increased fusion—which are known to help cells survive stress and avoid programmed cell death. Inhibiting YES1 or EZH2 reversed this fusion, promoted mitochondrial fragmentation, and restored the cancer cells’ tendency to die when treated with PARP inhibitors.
A Molecular Relay That Protects Cancer Cells
Inside mitochondria, EZH2 takes on an unexpected task: it directly modifies another protein, MYO19, which helps control how mitochondria move and divide. The authors found that phosphorylated EZH2 adds small chemical tags to MYO19 at a specific site, weakening MYO19’s ability to promote mitochondrial fission. The result is a shift toward fused, networked mitochondria that are more resistant to damage and less likely to trigger cell death. When MYO19 was altered so it could no longer be modified in this way, mitochondria stayed more fragmented, the cell-death machinery was reactivated, and PARP inhibitors became effective again in previously resistant cells.
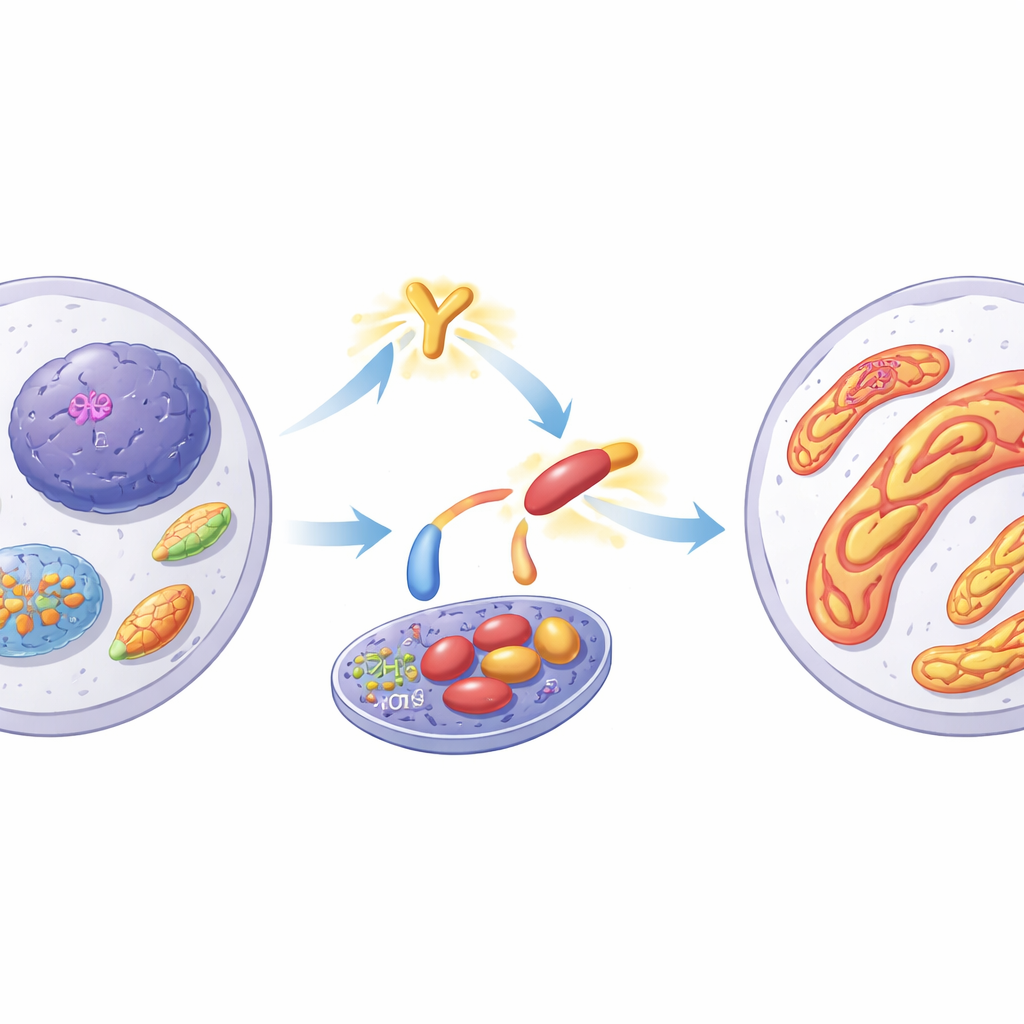
How the Drug Itself Fuels Resistance
Strikingly, exposure to PARP inhibitors helped set this entire escape route in motion. In initially sensitive cells, PARP inhibitor treatment activated YES1, which in turn stabilized EZH2 by preventing its breakdown through the cell’s protein disposal system. This led to higher levels of EZH2 and more of it in its phosphorylated, mitochondria-bound form. Over time, this feedback loop could convert a PARP inhibitor–sensitive tumor into a resistant one, even without changes in the BRCA1 gene itself.
Turning a Weakness Back into a Strength
To a layperson, the takeaway is that some ovarian cancers learn to survive PARP inhibitors not just by fixing their DNA, but by rewiring their energy factories to resist self-destruction. The study reveals a key relay—YES1 activating EZH2, EZH2 modifying MYO19, and mitochondria shifting toward a survival mode—that allows BRCA1-deficient tumors to outlast treatment. By blocking YES1 or EZH2, the researchers were able to break this relay, push mitochondria back into a more fragile state, and make resistant tumors vulnerable again in organoids, cell cultures, and mouse models. These findings suggest that testing tumors for the phosphorylated form of EZH2 and combining PARP inhibitors with existing YES1 or EZH2 drugs could offer a promising new strategy to extend the benefits of precision therapy for women with high-risk ovarian cancer.
Citation: Hu, L., Ma, X., Cai, X. et al. The mitochondrial translocation of phosphorylated EZH2 promotes PARP inhibitor resistance in BRCA1-deficient epithelial ovarian cancer. Cell Discov 12, 17 (2026). https://doi.org/10.1038/s41421-026-00880-x
Keywords: ovarian cancer, PARP inhibitor resistance, BRCA1 deficiency, mitochondrial dynamics, EZH2