Clear Sky Science · en
Inosine promotes erythrocyte metabolic reprogramming and restores oxygen release for rejuvenation via 2,3-BPG-PNP axis
Why This Matters for Healthy Aging
As people grow older, their organs often don’t get quite enough oxygen, a quiet stress that contributes to heart disease, memory loss, weak muscles, and other age‑related problems. This study asks a simple but powerful question: what if part of the problem starts inside the red blood cells that carry oxygen, and what if a common natural molecule called inosine could help restore their vigor?
How Red Blood Cells Change With Age
The researchers followed more than 300 adults ranging from 20 to 85 years old and carefully measured how well their red blood cells released oxygen. They found a steady decline across the lifespan: older people’s red blood cells held onto oxygen more tightly and released less of it to tissues. This change went hand in hand with warning signs in other organs, including higher blood pressure and subtle declines in kidney and liver function. In mice, the same pattern appeared: older animals had red blood cells that released less oxygen and showed more oxidative stress, a chemical wear‑and‑tear linked to aging.
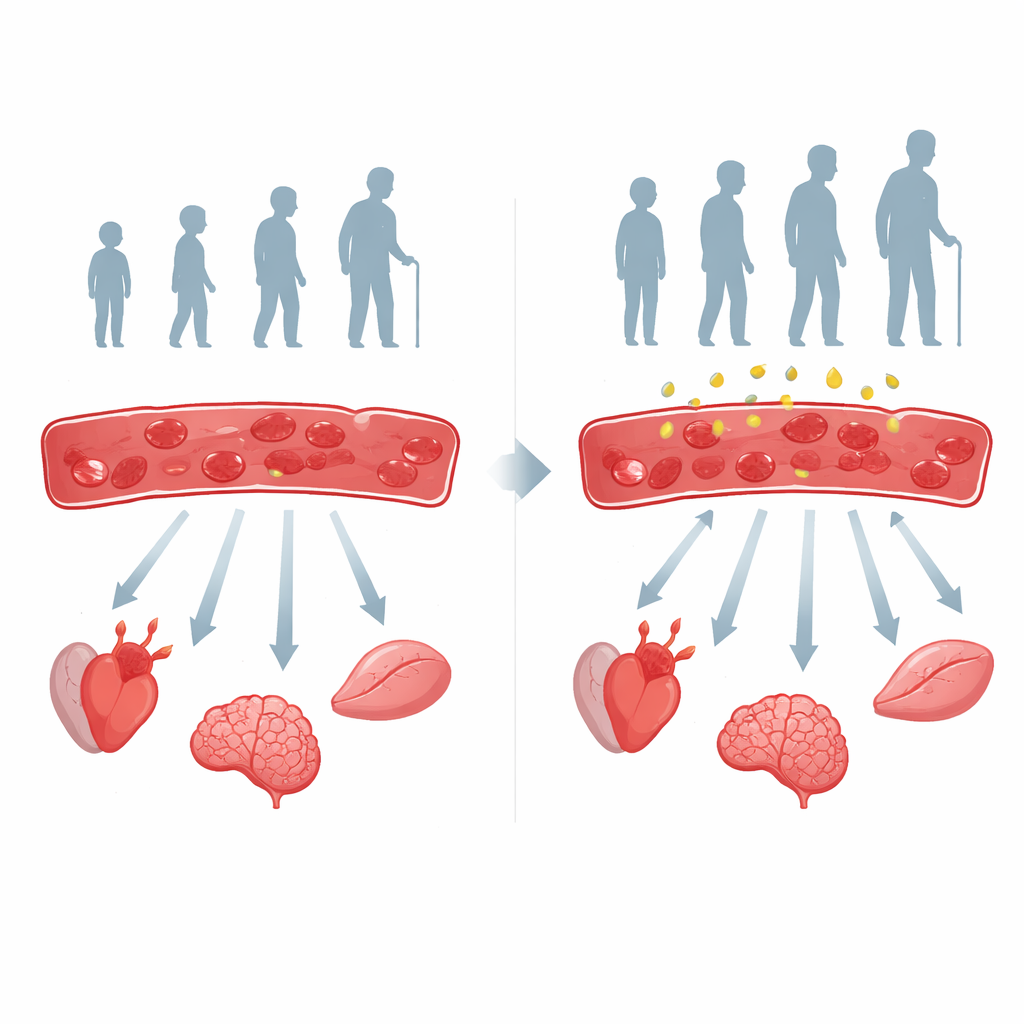
The Hidden Fuel Switch Inside Blood Cells
Red blood cells lack nuclei and mitochondria, so they rely on a streamlined internal chemistry to make energy and fine‑tune oxygen release. A small molecule called 2,3‑BPG normally helps hemoglobin let go of oxygen where it is needed. The team found that 2,3‑BPG levels fall with age because an enzyme that makes it, BPGM, becomes less active. In both humans and mice, lower 2,3‑BPG tracked closely with poorer oxygen release. When the scientists engineered mice whose red blood cells could not make 2,3‑BPG, the animals developed earlier tissue hypoxia, more oxidative stress, weaker muscles, worse memory, and poorer control of blood sugar—essentially an accelerated aging picture driven just by red blood cell changes.
Inosine as a Backup Energy Source
Metabolic profiling revealed that aged red blood cells don’t just slow down; they rewire. As glucose handling falters, they increasingly burn inosine, a natural compound derived from nucleotides. In older people and mice, the enzyme PNP, which breaks down inosine into a sugar fragment called ribose‑1‑phosphate, became more active. Using labeled inosine, the researchers showed that this ribose fragment flows into key internal pathways that support energy production and, importantly, help rebuild 2,3‑BPG. In test‑tube experiments, bathing human or mouse red blood cells in inosine quickly boosted their ability to release oxygen and lowered damaging reactive oxygen species—provided that the cells could take up inosine through a transporter called ENT1 and digest it with PNP.
Genetic Tests and Animal Trials
To confirm the importance of this backup fuel system, the team created mice whose red blood cells lacked ENT1, the entry door for inosine. These animals could not benefit from inosine, had poorer oxygen release, higher oxidative stress, and showed faster declines in cognition, muscle strength, and glucose control as they aged. The scientists then moved to a preclinical test: they gave middle‑aged mice daily inosine injections for a month. Treated animals showed red blood cells that released more oxygen and produced fewer harmful oxidants. The mice ran better on a rotating rod, gripped more strongly, performed better in memory tests, and had less evidence of low‑oxygen damage in the heart, kidneys, muscles, and key brain regions involved in learning and memory.
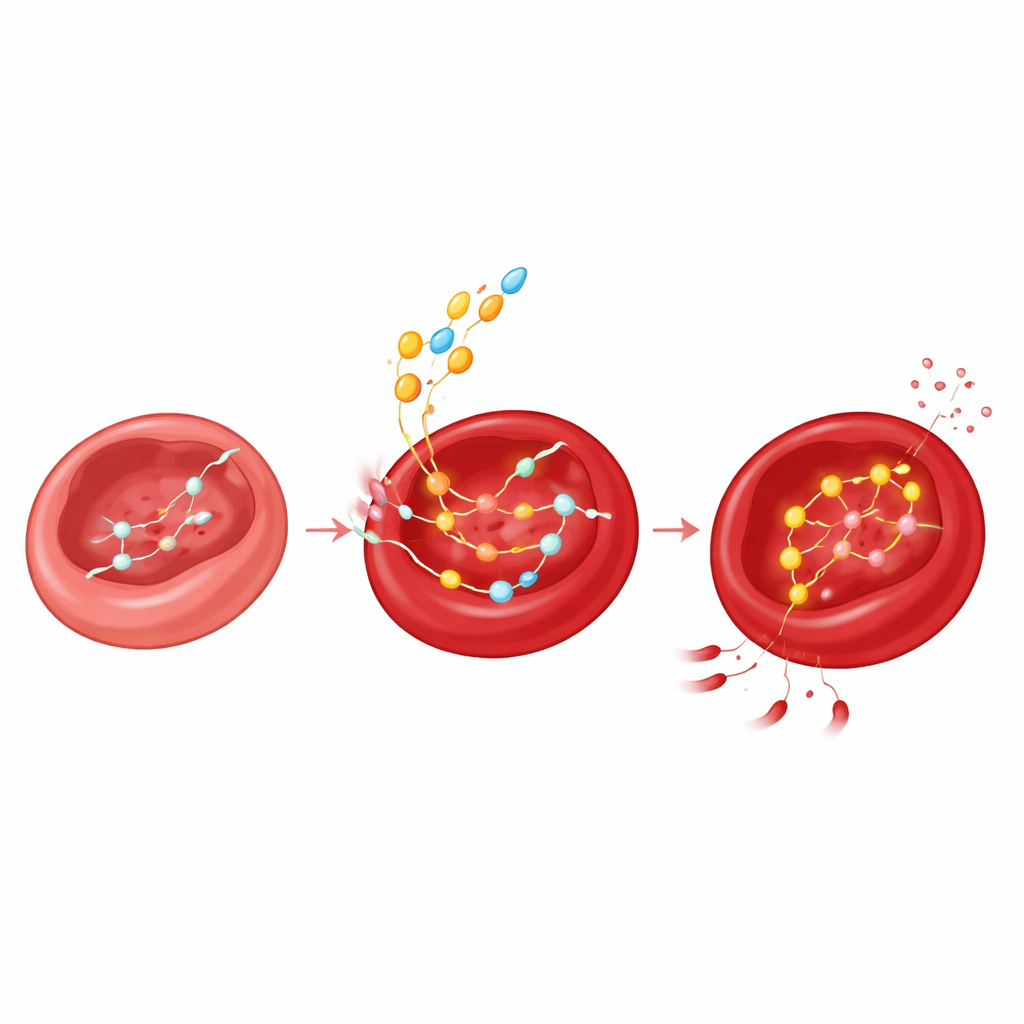
A Molecular Brake That Loosens With Age
The study also uncovers an elegant feedback loop. Using molecular modeling, enzyme assays, and targeted mutations, the authors show that 2,3‑BPG itself binds to PNP and blocks its activity by competing with phosphate at specific contact points on the enzyme. In youth, plentiful 2,3‑BPG therefore keeps inosine breakdown in check. As 2,3‑BPG levels fall with age, this brake releases: PNP becomes more active, inosine is consumed more rapidly, and its ribose fragment is diverted into energy‑producing pathways to compensate for sluggish glucose metabolism. In other words, the same molecule that helps hemoglobin release oxygen also quietly governs how red blood cells tap into an emergency fuel reserve.
What This Means for Aging and Rejuvenation
Taken together, the work proposes that aging is not only about failing organs but also about aging red blood cells that deliver less oxygen. A drop in BPGM activity and 2,3‑BPG marks a new “hallmark of aging,” while the inosine–PNP–ENT1 axis serves as a built‑in rescue system that partially restores energy flow and oxygen release. By supplying extra inosine from outside, at least in mice, the researchers could strengthen this rescue system, improve red blood cell performance, ease tissue hypoxia, and blunt declines in strength and memory. Although much remains to be tested in humans, the study raises the intriguing possibility that fine‑tuning red blood cell metabolism—perhaps with inosine or related strategies—could become a new route to healthier aging.
Citation: Liu, W., Yang, Z., Chen, C. et al. Inosine promotes erythrocyte metabolic reprogramming and restores oxygen release for rejuvenation via 2,3-BPG-PNP axis. Cell Discov 12, 19 (2026). https://doi.org/10.1038/s41421-026-00877-6
Keywords: red blood cells, aging, oxygen delivery, inosine, metabolism