Clear Sky Science · en
Bilirubin directly activates RIPK3 to induce non-classical necroptosis
When a Helpful Molecule Turns Harmful
Bilirubin is best known as the yellow pigment behind newborn jaundice and the familiar color of bruises. At low levels, it actually helps protect our cells from damage. But when bilirubin builds up in the body and seeps into the brain, it can cause lasting injury, especially in newborns and people with severe liver disease. This study uncovers a surprising way that excess bilirubin can directly flip a molecular “kill switch” inside brain cells, driving a destructive form of cell death and offering a fresh target for future therapies.
From Blood Pigment to Brain Threat
Bilirubin is produced when our bodies recycle red blood cells. Normally, the liver modifies and removes it, keeping levels low and relatively safe. In certain conditions—such as neonatal jaundice, severe infections, or liver failure—unprocessed bilirubin can accumulate. Because it dissolves easily in fats, this “free” form can cross the blood–brain barrier, especially when that barrier is weakened by illness or inflammation. Once inside the brain, bilirubin is known to be toxic, but the exact steps by which it damages nerve cells have remained unclear.
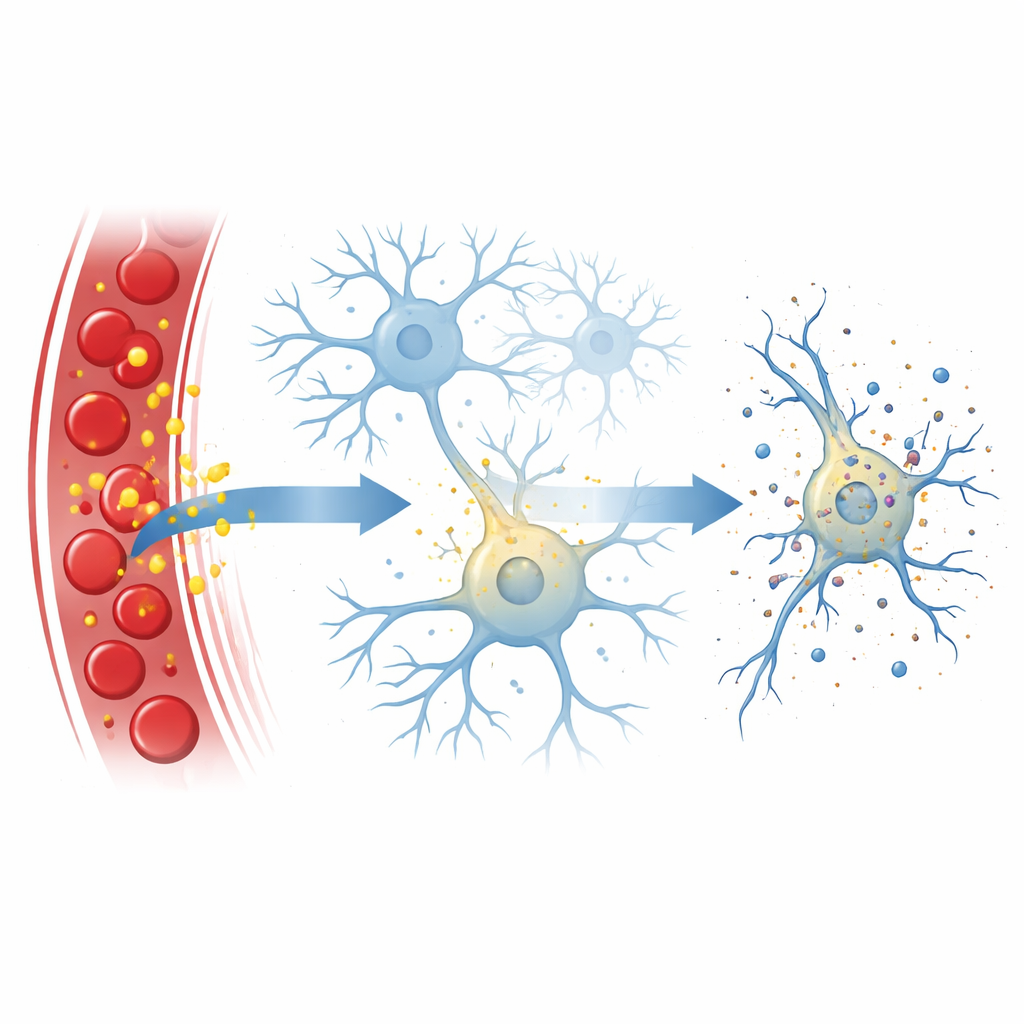
A Controlled Cell Explosion
The researchers focused on necroptosis, a regulated form of cell death that looks like a tiny explosion: the cell swells, its outer membrane ruptures, and its contents spill out, often triggering inflammation. This process is normally driven by a chain of proteins, with one called RIPK3 acting as a central hub and another, MLKL, punching holes in the cell’s membrane. In standard textbook pathways, RIPK3 is switched on by partner proteins that recognize danger signals such as inflammatory molecules or viral genetic material. Here, the team found that bilirubin can bypass these usual partners and still drive necroptosis in nerve cells.
Bilirubin Grabs the Cell’s Kill Switch
Using cultured neurons, brain slices, and mouse models, the scientists showed that bilirubin selectively activates RIPK3 and its downstream partner MLKL in nerve cells, while leaving the usual upstream proteins largely unchanged. Genetic experiments revealed that when RIPK3 or MLKL were removed, bilirubin could no longer efficiently kill neurons. Biochemical tests went further: bilirubin physically bound to RIPK3 at two specific spots within its active core, encouraged RIPK3 molecules to cluster together, and boosted their kinase activity—the chemical function that turns the death pathway on. This direct grip on RIPK3 did not depend on the protein’s typical interaction surface (the RHIM domain) or on other adapter proteins, marking out a non-classical route into necroptosis.
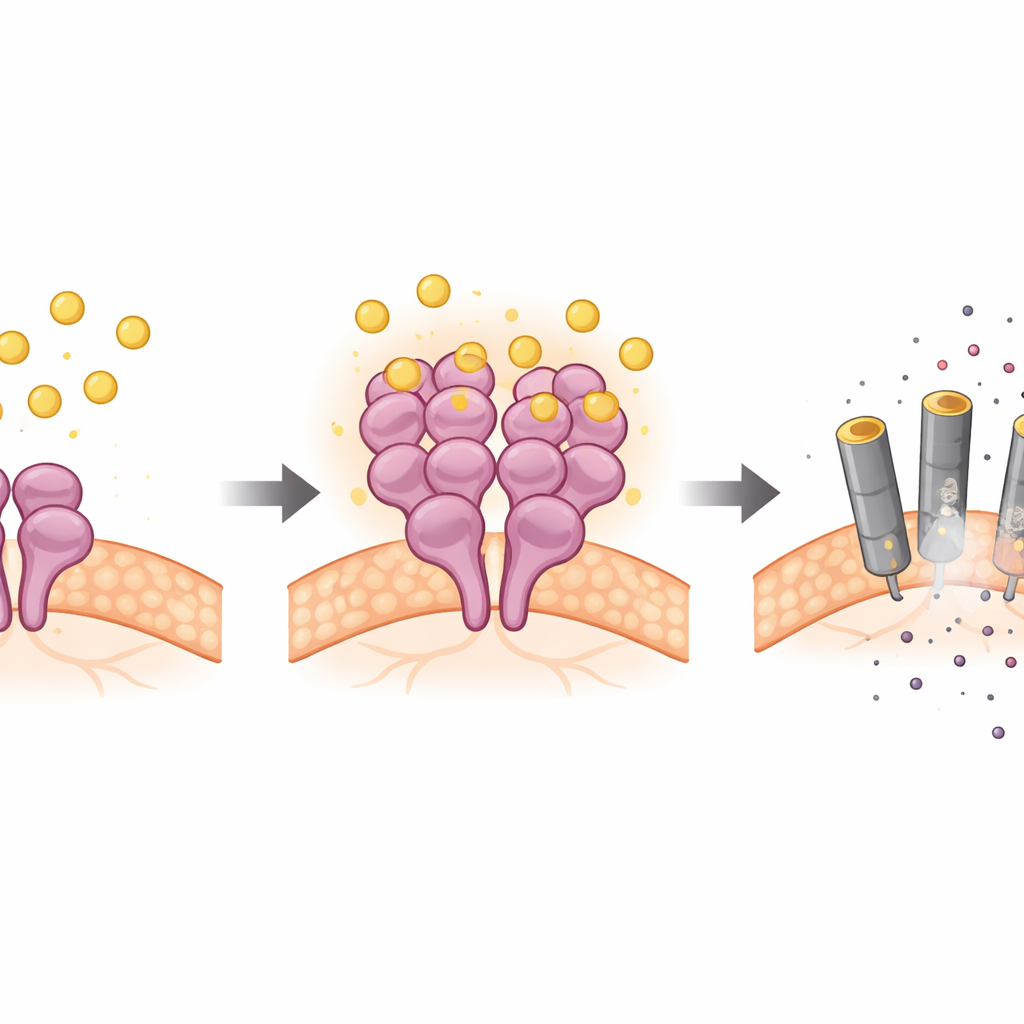
Evidence from the Living Brain
To see whether this mechanism matters in whole animals, the team raised bilirubin levels in mice either by injecting it into the brain or by inducing liver injury and inflammation so that bilirubin naturally entered the brain. In normal mice, this led to strong RIPK3 and MLKL activation in vulnerable brain regions, increased markers of cell death, and visible loss of healthy neurons. Mice engineered to lack RIPK3 were protected: their neurons showed much less damage, fewer death signals, and reduced inflammatory responses, even though bilirubin levels were just as high. Importantly, a processed, water-soluble form of bilirubin that is usually considered harmless neither activated RIPK3 nor caused similar brain injury, underscoring that the unmodified, fat-soluble form is the real culprit.
What This Means for Patients
This work reveals that excess unconjugated bilirubin can directly flip on RIPK3, triggering a controlled but destructive rupture of brain cells through necroptosis. Rather than acting only as a general poison or source of oxidative stress, bilirubin behaves like a small-molecule activator of a specific death program in neurons and brain immune cells. For patients with severe jaundice or liver failure, this suggests that blocking RIPK3 or MLKL—especially with drugs that can cross into the brain—might one day help limit neurological damage. In simple terms, the study shows how a normally helpful pigment can become a precise molecular assassin, and it points to new ways of disarming it without disrupting its everyday protective roles.
Citation: Xue, Q., Ma, X., Chen, Z. et al. Bilirubin directly activates RIPK3 to induce non-classical necroptosis. Cell Discov 12, 21 (2026). https://doi.org/10.1038/s41421-026-00876-7
Keywords: bilirubin neurotoxicity, necroptosis, RIPK3, liver failure and brain injury, cell death pathways