Clear Sky Science · en
Palmitic acid activates c-Myc via dual palmitoylation-dependent pathways to promote colon cancer
Why Fat in Our Diet Matters for Colon Health
Colon cancer is one of the most common cancers worldwide, and many studies link high-fat diets to a greater risk of developing it. Yet how exactly certain fats in our food talk to cancer-driving genes inside colon cells has been unclear. This paper uncovers a direct connection between palmitic acid—a major fat in meat, dairy, and many processed foods—and a powerful cancer-promoting gene called c-Myc. The work shows how this common dietary fat can stoke inflammation, rewire cell metabolism, and lock colon tumors into a harmful dependency on palmitic acid.
From Everyday Fat to Chronic Gut Inflammation
Palmitic acid is a saturated fat abundant in high-fat diets. The researchers fed mice different diets: normal chow, palmitic-acid-enriched diets, or a classical high-fat diet, and then triggered colitis, a type of colon inflammation. Mice on palmitic-acid-rich or high-fat diets developed more severe gut inflammation than those on a normal diet. Their colons were shorter, more damaged under the microscope, and filled with immune cells, signs of aggressive inflammatory injury. Gene activity patterns in these inflamed tissues looked similar to those seen in a well-known inflammation-driven colon cancer model, suggesting that palmitic acid pushes the gut environment toward a cancer-favorable state.
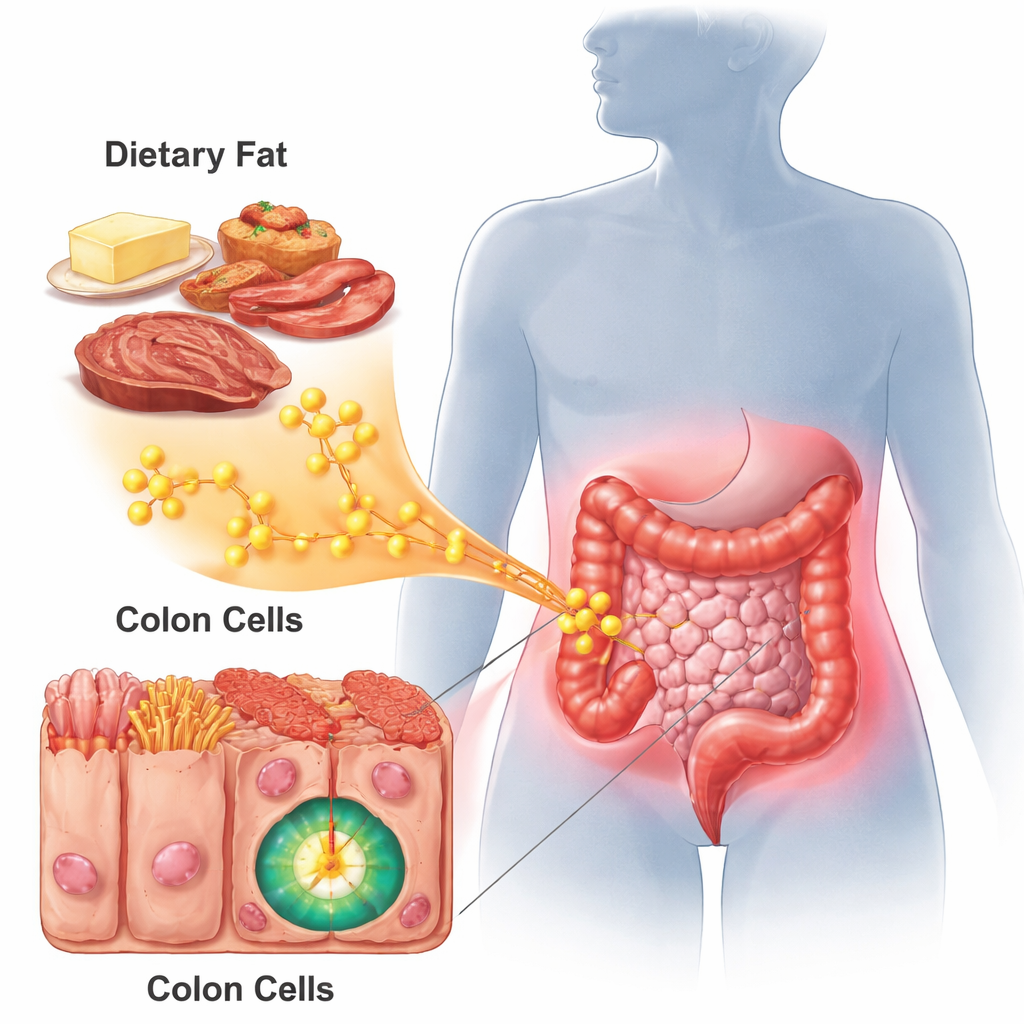
A Fat-Sensing Switch That Supercharges a Cancer Gene
The team focused on c-Myc, a master control gene that drives cell growth, survival, and stem-like behavior in many cancers. In both mouse tissues and human colon cancer samples, c-Myc’s activity—not its amount—was abnormally high. By comparing gene profiles from palmitic-acid-fed mice, early colon tumors, and advanced cancers, the scientists homed in on one critical helper protein: ZDHHC9. This enzyme attaches fatty acid chains to other proteins in a process called palmitoylation. They found that an inflammatory molecule, IL-1β, rises during colitis and boosts ZDHHC9 levels, which in turn directly modifies c-Myc at a specific site. This fatty “tag” strengthens c-Myc’s partnership with its binding partner MAX, making the c-Myc complex more active in turning on growth and stemness genes, even without increasing c-Myc’s own gene expression.
How Cancer Cells Become Addicted to Palmitic Acid
As tumors progress, the relationship between c-Myc and palmitic acid flips from a one-way push to a self-reinforcing loop. The study shows that c-Myc directly switches on FATP2, a transporter that imports long-chain fatty acids like palmitic acid into colon cancer cells. Tumors with higher c-Myc activity had more FATP2 and stronger signatures of fatty acid metabolism. In lab dishes and mouse tumor grafts, extra palmitic acid accelerated cancer cell growth and increased palmitoylation of c-Myc, further amplifying its activity. This creates a vicious cycle: inflammation and diet raise palmitic acid, which activates c-Myc; c-Myc then increases FATP2, pulling in even more palmitic acid and keeping the “growth engine” revved.
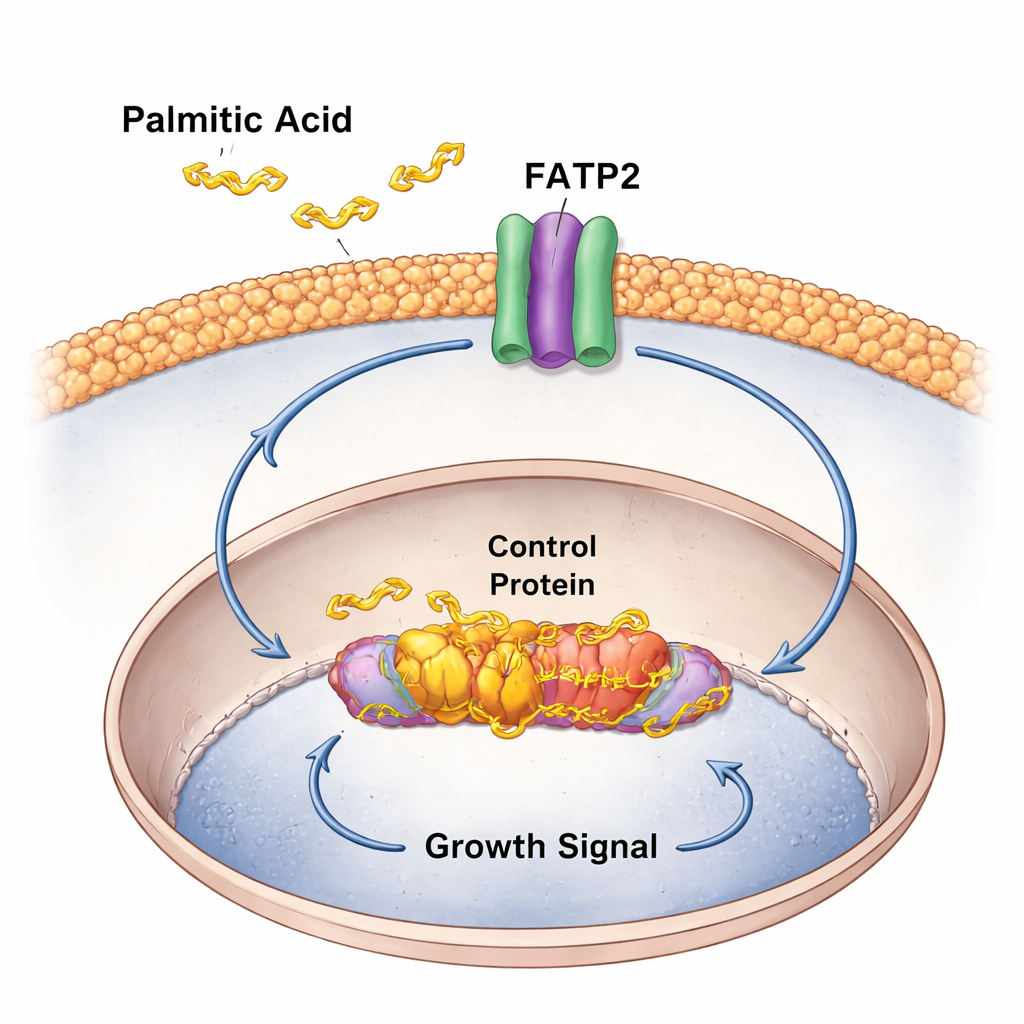
Turning a Metabolic Weakness into a Treatment Opportunity
Because colon tumors in this model become dependent on palmitic acid and on the ZDHHC9–FATP2–c-Myc axis, the authors tested whether breaking this circuit could slow cancer. Using genetic tools to switch off ZDHHC9 or FATP2 in tumor cells dramatically reduced tumor growth in mice fed palmitic-acid-rich diets. Combining both knockdowns was even more effective. They then used drug-like inhibitors: 2BP, which broadly blocks palmitoylation enzymes, and Lipofermata, which blocks FATP2. Each treatment reduced tumor size and the activity of c-Myc target genes, and together they shrank tumors by over 80 percent, without obvious toxicity in the mice. This suggests that targeting how cancer cells handle palmitic acid could indirectly tame an otherwise “undruggable” gene like c-Myc.
What This Means for Diet and Future Therapies
In plain terms, this study reveals that a common dietary fat can act as a signal that flips c-Myc into a higher gear, first by worsening gut inflammation and then by wiring mature tumors to crave more palmitic acid. Rather than just being fuel, palmitic acid becomes part of a communication network between diet, the immune system, and cancer-driving genes. The work suggests two main takeaways: limiting palmitic-acid-heavy foods may help reduce colon cancer risk, especially in people with chronic gut inflammation, and new treatments that block ZDHHC9 or FATP2 could exploit the tumor’s addiction to this fat to slow or stop disease.
Citation: Du, W., Zhang, J., Wang, Y. et al. Palmitic acid activates c-Myc via dual palmitoylation-dependent pathways to promote colon cancer. Cell Discov 12, 12 (2026). https://doi.org/10.1038/s41421-026-00869-6
Keywords: colon cancer, high-fat diet, palmitic acid, c-Myc, metabolic addiction