Clear Sky Science · en
Chronobiology of neurotropic viruses: rhythmic viral entry and arrhythmic host clocks
Why timing matters for brain infections
Many dangerous viruses, including rabies and herpes, have a knack for invading the brain and nerves. This study asks a surprisingly simple question with big implications: does the time of day change how easily these “neurotropic” viruses can get into our nervous system, and can the viruses in turn knock our internal clocks off balance? Understanding this two‑way conversation between body clocks and viruses could open new ways to time vaccines, antivirals, and even shift work to reduce risk.
Daily body clocks and viral doorways
Almost every cell in the body carries a built‑in 24‑hour timing system, often called the circadian clock. It is driven by a set of core clock proteins, including BMAL1 and REV-ERBα, that turn thousands of genes on and off in rhythmic waves. The authors focused on the “door handles” viruses use to enter cells – receptor proteins on the cell surface – for dozens of brain‑targeting or brain‑invading viruses. By examining brain‑like organoids grown from stem cells, nerve‑derived cell lines, and mouse tissues, they found that most of these viral receptors rise and fall over the day. These rhythms closely tracked the activity of clock components, suggesting that our daily timing system quietly controls how many viral entry points are available at any given hour.
A cell‑cycle factor becomes a clock messenger
Digging deeper, the researchers asked how a protein usually known for controlling cell division, called E2F8, fits into this timing network. They discovered that E2F8 itself is under circadian control and behaves like a relay between the clock and at least one key rabies receptor, p75NTR. When the clock protein REV-ERBα is active, it suppresses E2F8. In turn, low E2F8 releases its brake on the p75NTR gene, allowing more of this receptor to be produced and making cells more welcoming to rabies virus. Strikingly, E2F8 also feeds back on the clock: it can bind directly to the control regions of core clock genes such as REV-ERBα and PER2 and dampen their activity. This creates a feedback loop in which a cell‑cycle regulator helps sculpt daily rhythms and, through them, the vulnerability of nerve cells to infection.
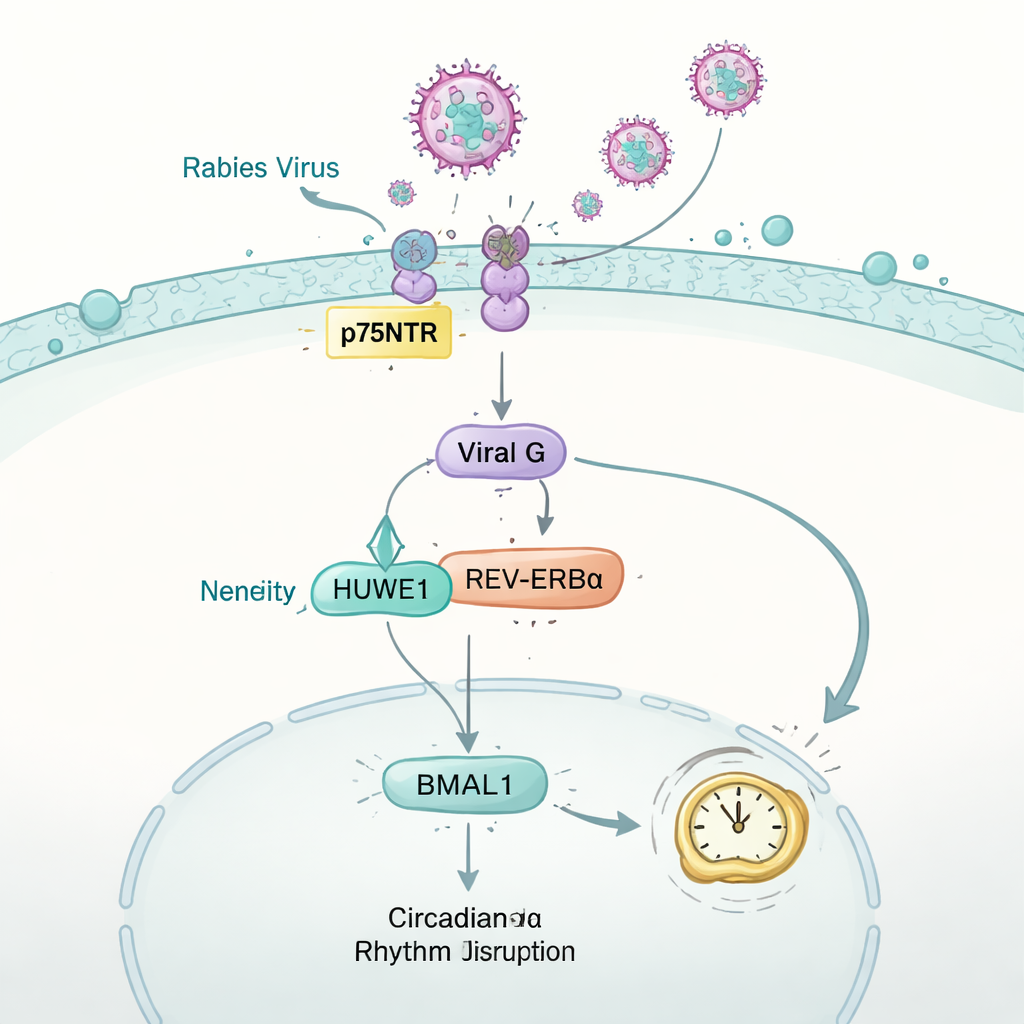
How rabies virus pushes the clock off track
The study then turned the question around: can a virus push back on the clock? Using mice infected with rabies virus, the team monitored running activity, metabolism, and breathing chemistry. Despite normal food intake early in infection, the animals gradually lost the regular day‑night patterns that healthy mice show. In the brain and several other organs, levels of the crucial clock protein BMAL1 dropped sharply. In nerve‑like cells, this effect could be traced to the rabies glycoprotein “G,” a surface protein that normally helps the virus fuse with cells. G hijacked a host enzyme named HUWE1, which usually helps tag another clock protein, REV-ERBα, for disposal. By competing for HUWE1, the viral G protein stabilized REV-ERBα, leading to extra suppression of BMAL1 and, ultimately, disturbance of the clockwork that keeps physiology on a 24‑hour schedule.
Timing, jet lag, and infection severity
To test whether these molecular changes matter for the whole organism, the team infected mice with rabies at different times of day, under regular light‑dark cycles, under chronic “jet lag” conditions, and in animals genetically lacking REV-ERBα. Mice infected at the beginning of their active (night) phase, when the rabies receptor p75NTR peaks, lost weight faster, had higher viral loads in multiple brain regions, more brain inflammation, and died sooner than mice infected at the start of their rest phase. Mice exposed to repeated phase shifts mimicking rotating shift work lost normal clock rhythms in the brain, showed persistently higher p75NTR levels, had weaker baseline antiviral signals, and suffered worse disease. In contrast, mice without REV-ERBα expressed less p75NTR, carried lower viral loads, and survived longer after infection, indicating that this clock component is a critical gatekeeper for rabies severity.
What this means for people
In plain terms, the study shows that our internal clocks do not just set sleep times; they also open and close molecular doorways that brain‑invading viruses use, and those same viruses can hit back by scrambling the clock. A single factor, REV-ERBα, sits at a key junction: together with E2F8 it shapes both receptor levels and daily timing, while rabies virus manipulates it to its own advantage. These insights suggest practical paths forward. Vaccines or antiviral drugs might work better at certain times of day, when receptors are low or immune responses are strong. People with chronically disrupted rhythms – such as shift workers or frequent travelers – might face higher risks from some neurotropic infections. Targeting REV-ERBα or related pathways could help tip the balance in favor of the host, making infections less severe and improving outcomes.
Citation: Zeng, S., Zhang, Q., Yang, X. et al. Chronobiology of neurotropic viruses: rhythmic viral entry and arrhythmic host clocks. Cell Discov 12, 11 (2026). https://doi.org/10.1038/s41421-026-00867-8
Keywords: circadian clock, neurotropic viruses, rabies, viral receptors, chronotherapy