Clear Sky Science · en
Dynamic changes of the immune microenvironment in ovarian cancer following neoadjuvant chemotherapy
Why this research matters for women with ovarian cancer
Advanced ovarian cancer is often treated with powerful chemotherapy before or after surgery, yet many women still see their disease return and stop responding to drugs. This study asks a pressing question: how does chemotherapy quietly re‑shape the body’s own defences around the tumour, and can we tweak that response to keep treatment working longer? By looking at individual cells and testing new drug combinations in mice, the researchers uncover a hidden role for fatty hormone‑like molecules called prostaglandins in helping tumours evade the immune system after chemotherapy.
A shifting battlefield around the tumour
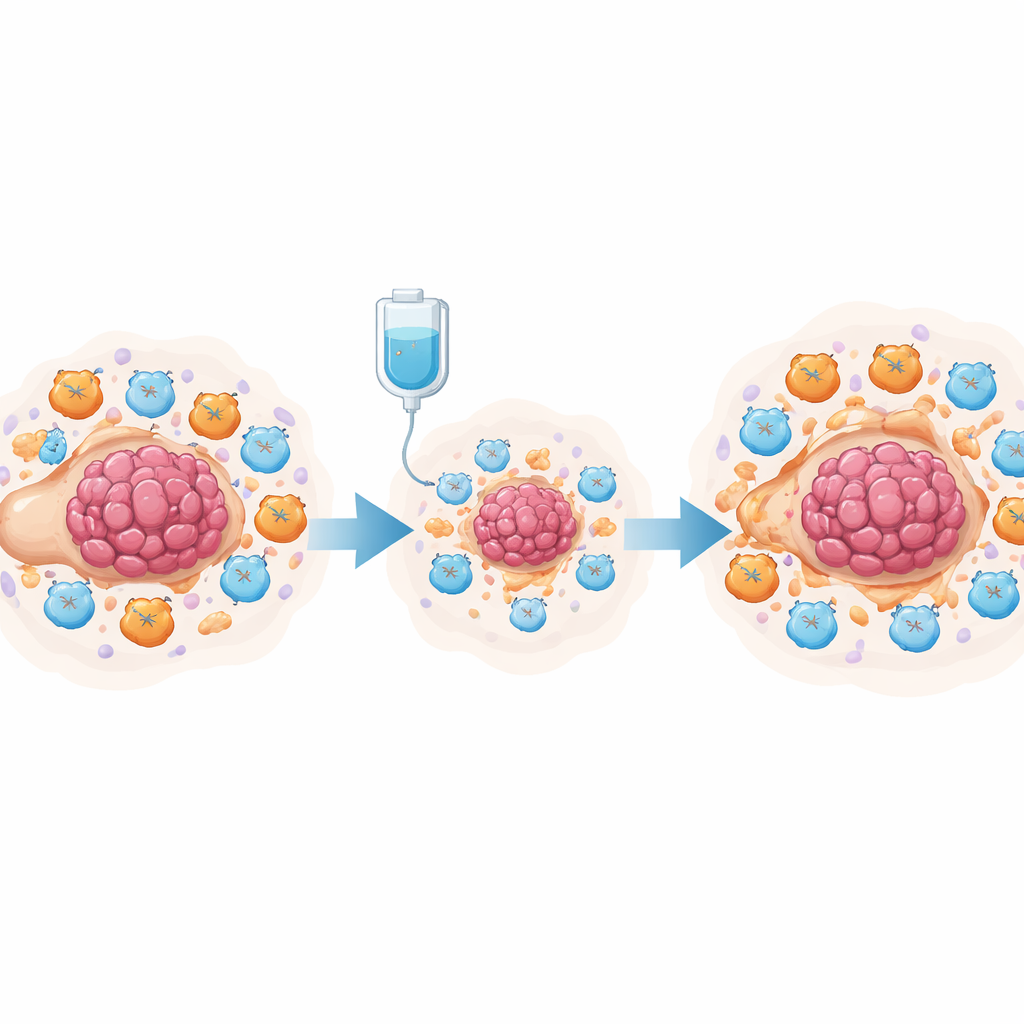
Ovarian tumours do not grow in isolation: they sit in a crowded “neighbourhood” of immune cells, connective tissue, and fluid in the abdomen. Before treatment, this neighbourhood is already tilted in favour of the cancer, with few aggressive killer T cells and many cells that dampen immune attack. The team analysed previously published single‑cell RNA sequencing data from women with high‑grade serous ovarian cancer, comparing samples taken before and after neoadjuvant chemotherapy (chemotherapy given before surgery). They found that chemotherapy did not simply kill tumour cells; it also temporarily stirred up immune activity, increasing signals linked to T‑cell stimulation and inflammation while reducing some regulatory T cells that usually suppress attack.
When helpful changes turn harmful
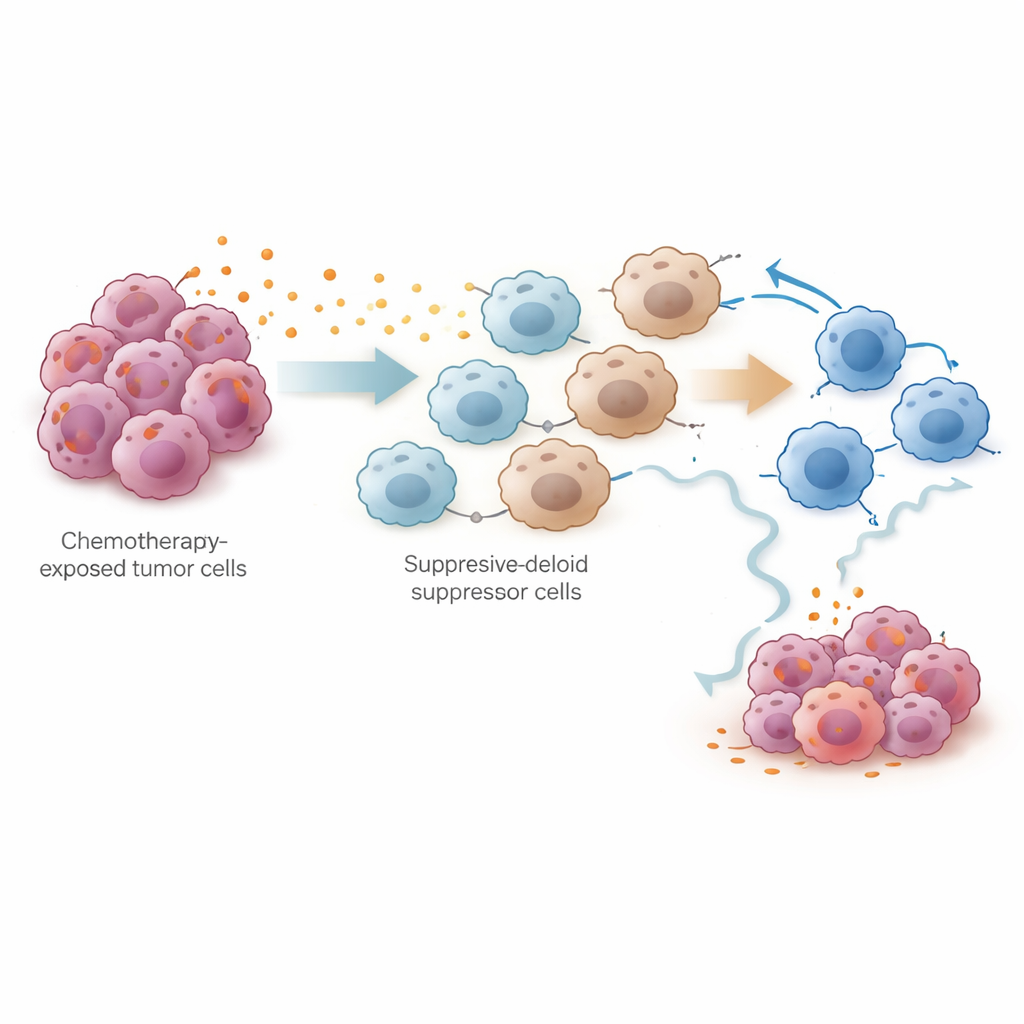
Despite this brief boost, most patients still relapse and become resistant to platinum drugs such as cisplatin. To understand why, the researchers probed deeper into how different cell types talk to each other after treatment. They discovered that chemotherapy strongly increased genes involved in making prostaglandins in tumour‑associated macrophages and fibroblasts, and boosted communication between these cells and T cells. Using advanced mathematical tools, they showed that the overall environment shifted toward a pro‑inflammatory yet ultimately suppressive state, with rising levels of chemokines that attract myeloid cells and enhanced pathways that can exhaust T cells over time. This suggested that prostaglandins might act as a molecular switch, turning an initially helpful immune surge into a long‑lasting brake on anti‑tumour immunity.
Chemotherapy, prostaglandins, and silenced T cells
To test this idea experimentally, the team used a mouse model of ovarian cancer treated with cisplatin. Single‑cell analysis of tens of thousands of cells from the abdominal cavity showed that cisplatin reduced tumour cells and increased total T cells, but also markedly expanded myeloid‑derived suppressor cells (MDSCs) and exhausted killer T cells. Both tumour cells and these suppressor cells produced high levels of prostaglandin‑making enzymes. In co‑culture dishes, cisplatin‑treated tumour cells drove bone‑marrow cells to adopt an MDSC‑like state and weakened the activity and growth of CD8+ killer T cells. The researchers traced this effect to the NF‑κB signalling pathway in tumour cells, which switched on the PTGES gene, a key step in producing prostaglandin E2. Blocking this pathway reduced PTGES levels and prostaglandin output.
Turning the tide with combination therapy
The most practical question was whether blocking prostaglandins could make chemotherapy work better. In mice, the scientists used genetic deletion of Ptges in tumour cells, antibodies that neutralise prostaglandin E2, or small‑molecule drugs that inhibit prostaglandin E synthase. All of these approaches lowered prostaglandin levels, reduced the build‑up of MDSCs, and restored the killing ability and stem‑like resilience of CD8+ T cells. When combined with cisplatin, or with the standard cisplatin‑paclitaxel regimen, prostaglandin inhibitors slowed tumour growth and spread more effectively than chemotherapy alone. The data indicate that prostaglandin‑driven immune suppression is not a side note but a central reason why tumours escape after an initial response.
A new angle on keeping treatment effective
In everyday terms, this work suggests that chemotherapy changes the tumour’s surroundings in two phases: first it weakens the cancer and briefly rallies the immune system, but then it triggers a wave of prostaglandins that call in suppressor cells and tire out the body’s natural tumour‑killing forces. By identifying PTGES‑driven prostaglandin production as a key culprit, the study points to a tangible strategy: pairing standard platinum chemotherapy with drugs that block prostaglandin pathways may help maintain a strong immune attack and delay or prevent relapse in ovarian cancer. While clinical trials are still needed, this combination approach offers a hopeful new direction for extending the benefit of existing treatments.
Citation: Wu, M., Lv, F., Jin, Y. et al. Dynamic changes of the immune microenvironment in ovarian cancer following neoadjuvant chemotherapy. Cell Death Discov. 12, 130 (2026). https://doi.org/10.1038/s41420-026-03070-6
Keywords: ovarian cancer, tumor microenvironment, chemotherapy resistance, prostaglandins, immunotherapy