Clear Sky Science · en
ATP6V1B2 alleviates hepatic steatosis by promoting lysosomal acidification in hepatocytes
Why a fatty liver matters
Many people carry extra fat in their liver without realizing it. This condition, now called metabolic dysfunction-associated steatotic liver disease (MASLD), affects roughly a quarter of the world’s population and can quietly progress to scarring, liver failure, and other serious illnesses. Yet doctors still lack good ways to predict who will worsen and few treatments directly target the root cellular problems. This study uncovers how a little-known cellular “acid pump” helps liver cells dispose of excess fat and suggests a new direction for both diagnosis and therapy.
A hidden helper inside liver cells
Inside every liver cell are tiny recycling centers called lysosomes. They rely on an internal acid bath to break down worn-out parts and surplus fat. The acid is maintained by a molecular pump known as V-ATPase, built from several protein pieces. The authors focused on one crucial piece, ATP6V1B2, which is especially important in keeping lysosomes acidic. They asked whether changes in this protein might help explain why fat builds up in MASLD and whether its levels in the blood could signal disease.
Clues from patients and animal models
When the researchers examined public genetic databases and blood samples from patients, they found that ATP6V1B2 levels were consistently lower in people with MASLD and even lower in those with cirrhosis, a severe form of liver damage. Lower levels of ATP6V1B2 in the liver were linked with more inflammation, worse scarring, and higher markers of liver injury in the blood. In mouse models fed fatty or nutrient-deficient diets—standard ways to mimic MASLD—the liver’s supply of ATP6V1B2 also dropped. Together, these findings suggest that loss of this protein is not just a side effect but may be tied closely to disease progression.
What goes wrong inside stressed liver cells
To see what happens when ATP6V1B2 is disturbed, the team turned to cultured liver cells overloaded with fatty acids, mimicking a “lipotoxic” environment. When they artificially reduced ATP6V1B2, fat droplets in the cells swelled, harmful oxygen byproducts surged, and the cells’ energy factories (mitochondria) began to fail. Stress signals inside the cell spiked, and a key fat-building enzyme called fatty acid synthase (FASN) rose sharply. In contrast, when ATP6V1B2 was boosted, cells handled the fat load much better: lipid droplets shrank, oxidative stress eased, and mitochondria recovered. These experiments show that ATP6V1B2 acts as a protective brake on fat buildup and cell damage.
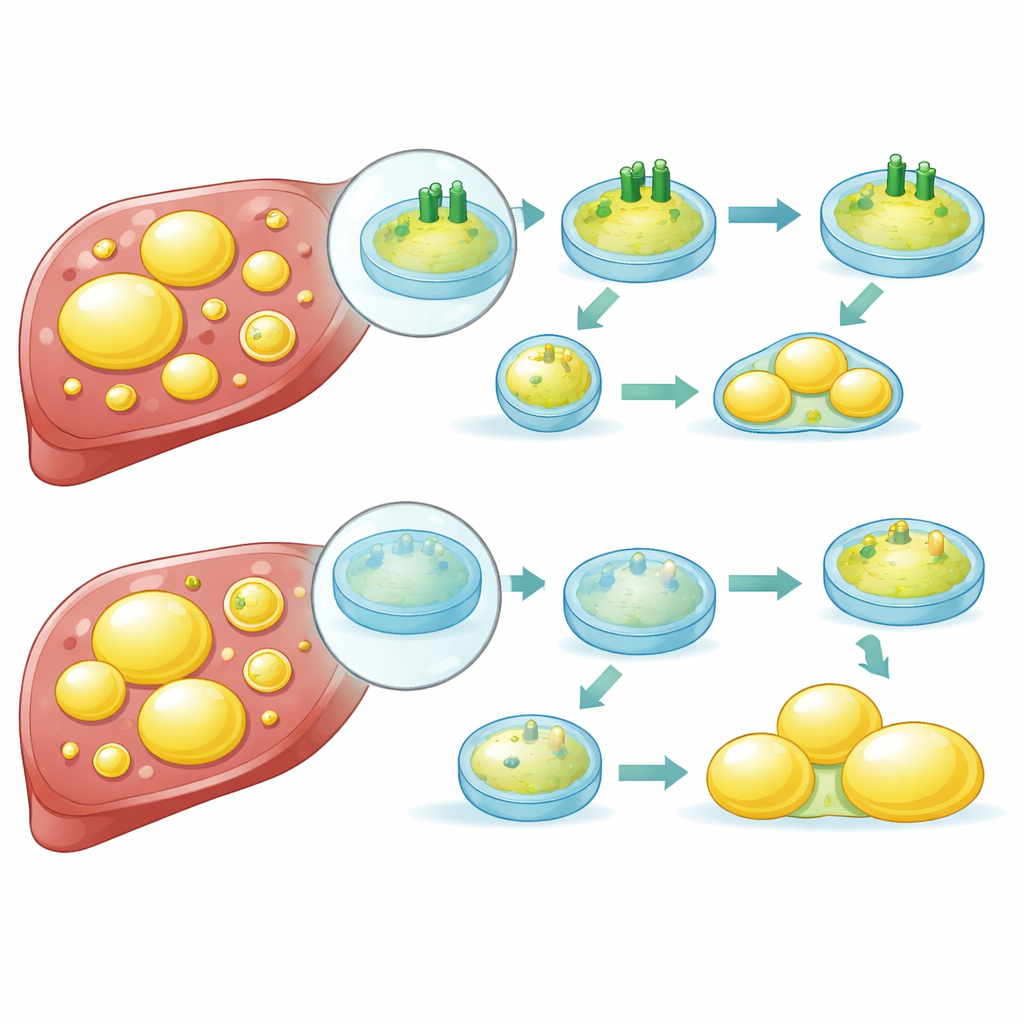
How broken recycling drives fat buildup
Digging deeper, the researchers discovered that ATP6V1B2 controls how acidic and active lysosomes remain. When the protein was reduced, lysosomes lost acidity, their ability to chew up cargo dropped, and the flow of material through the cell’s recycling system (autophagy) became blocked. Under normal conditions, FASN can be sent to lysosomes for destruction. With healthy ATP6V1B2, this pathway works efficiently, keeping FASN levels in check and limiting new fat production. But when ATP6V1B2 is scarce, lysosomes cannot degrade FASN properly. The enzyme accumulates, the cell ramps up fat synthesis, and droplets of fat continue to grow. The study also suggests that stress pathways inside the cell further increase FASN production when ATP6V1B2 is low, creating a vicious cycle.
What this means for future care
For non-specialists, the message is straightforward: a single protein that helps lysosomes stay acidic can strongly influence whether liver cells store or clear fat. Lower ATP6V1B2 levels, seen in both patients and models of MASLD, weaken the liver’s internal recycling system, allowing fat and damage to build up. Measuring this protein in blood may help doctors gauge disease severity, and strategies that restore lysosomal acidity or enhance ATP6V1B2 activity could form the basis of new treatments. By shining light on this “acid pump” and its control over fat-handling enzymes, the study points toward lysosome-focused therapies as a promising way to halt or reverse fatty liver disease.
Citation: Xu, R., Yang, F., Zhang, Z. et al. ATP6V1B2 alleviates hepatic steatosis by promoting lysosomal acidification in hepatocytes. Cell Death Discov. 12, 170 (2026). https://doi.org/10.1038/s41420-026-03052-8
Keywords: fatty liver disease, lysosomes, autophagy, lipid metabolism, liver inflammation