Clear Sky Science · en
Deciphering the STAT3-PXN positive feedback loop in GBM, IDH-wildtype: transcriptional regulation and inhibition of YB-1 ubiquitination
Why this brain cancer study matters
Glioblastoma is one of the most aggressive brain cancers, and even with surgery, radiation, and chemotherapy, most patients live little more than a year. This study digs into the inner wiring of glioblastoma cells to find out why they grow so fast and resist the standard drug temozolomide. By uncovering a self‑reinforcing molecular loop that keeps these tumors alive and dangerous, the researchers point to new weak spots that future treatments could exploit.
A deadly tumor with few good options
The work focuses on a form of glioblastoma known as IDH‑wildtype, which is now the main category of this cancer in adults. Patients with this diagnosis typically survive only 12 to 21 months, and nearly all tumors become resistant to temozolomide over time. To search for new treatment targets, the team used large cancer databases and survival data to look for genes that are especially active in these tumors and linked to poor outcomes. One gene stood out: paxillin, abbreviated PXN, a protein that helps cells attach to their surroundings and sense growth signals.
Spotlighting a key troublemaker
Using patient samples and brain tumor cell lines, the researchers confirmed that PXN levels are much higher in glioblastoma tissue than in nearby normal brain and in non‑cancerous brain cells. Tumors with more PXN were associated with worse patient prognosis. When the team used genetic tools to dial PXN down in glioblastoma cells, the cancer cells grew more slowly, formed fewer colonies, and were less able to move and invade in lab dishes. In mice, tumors lacking PXN grew poorly and the animals lived longer. The reverse was also true: forcing cells to make extra PXN boosted growth, spread, and tumor‑forming ability in animals, underscoring PXN as a driver of malignancy rather than a passive bystander.
A self‑reinforcing growth loop inside tumor cells
The study then asked what controls PXN and how it, in turn, talks to other signaling pathways. The team homed in on STAT3, a well‑known switch that relays growth and survival cues in many cancers. Data from patient tumors showed that STAT3 activity tracks closely with PXN levels. Blocking STAT3, either with a genetic approach or a chemical inhibitor, lowered PXN and reduced activity from the PXN gene’s on–off switch. A binding assay confirmed that STAT3 physically occupies PXN’s control region in DNA, directly turning it on. Surprisingly, the influence also ran the other way: when PXN was reduced, the activated form of STAT3 dropped, even though total STAT3 did not change. The researchers found that PXN can move into the cell nucleus and latch onto the control region of another gene, SRC, which encodes a protein that turns STAT3 on. By boosting SRC production, PXN raises STAT3 activity, which then further raises PXN—a classic positive feedback loop that keeps growth signals humming.
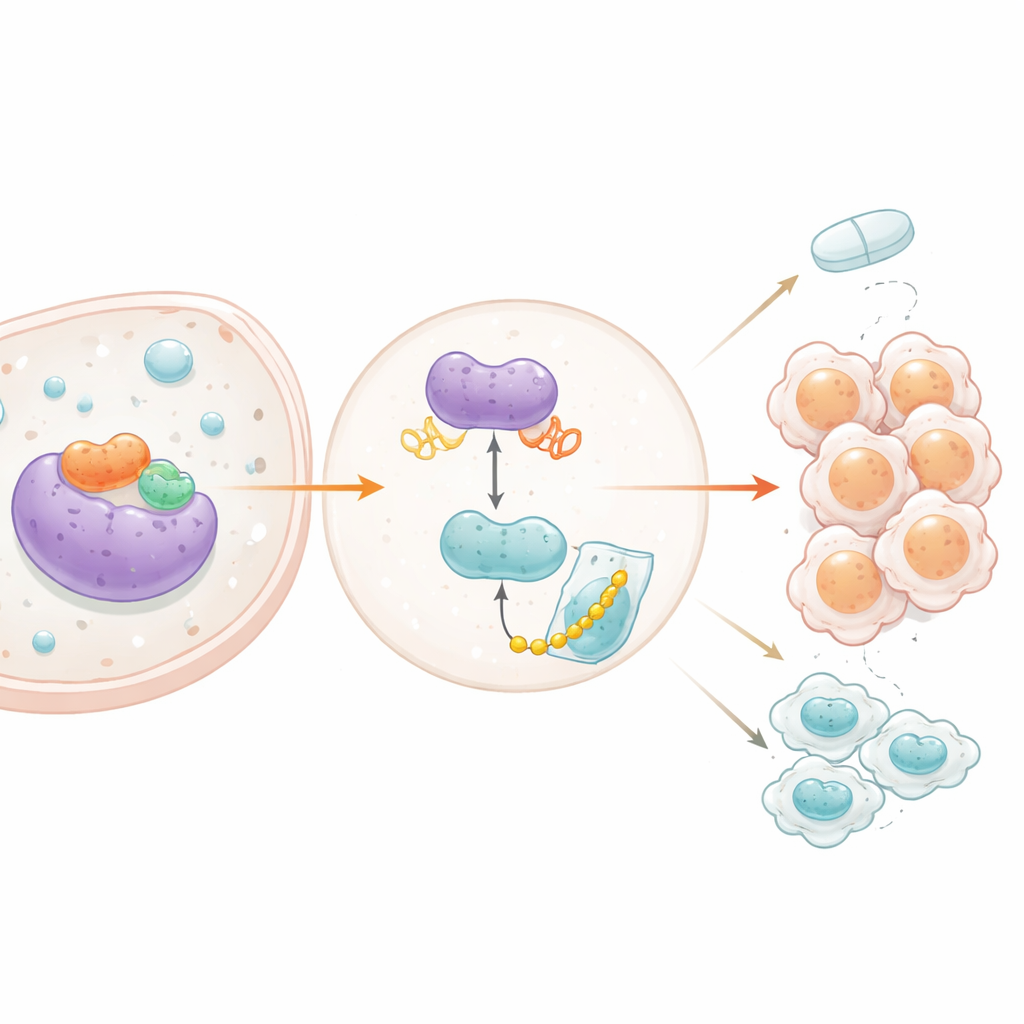
Protecting a powerful helper protein
Beyond this loop, PXN also stabilizes another influential molecule called YB‑1, known to promote brain tumor growth and therapy resistance. PXN physically binds YB‑1, and when PXN was silenced, YB‑1 protein levels fell even though its RNA levels stayed the same, hinting at a change in protein lifespan rather than gene output. Follow‑up experiments showed that, without PXN, YB‑1 is broken down more quickly by the cell’s protein disposal machinery. Blocking this disposal system rescued YB‑1 levels, and cells lacking PXN showed more of the small tags that mark YB‑1 for destruction. In other words, PXN acts like a shield, preventing YB‑1 from being tagged and shredded. When the team globally profiled genes controlled by YB‑1, they found that it sustains multiple cancer‑promoting pathways and supports genes tied to resistance against temozolomide, including the well‑known marker CD44.
New angles for future treatments
Taken together, the findings paint PXN as a central amplifier in glioblastoma: it sits at the crossroads of a STAT3–SRC feedback loop that keeps growth signals switched on and protects YB‑1 from breakdown so that many cancer pathways and drug‑resistance programs remain active. For patients, this means PXN and its partners are attractive targets for new therapies, especially in combination with temozolomide. While turning such molecular insights into safe brain‑penetrating drugs will be challenging, this work maps out a concrete network of weak spots that future treatments and smart drug‑delivery systems could try to hit.
Citation: Li, X., Guo, H., Liu, Z. et al. Deciphering the STAT3-PXN positive feedback loop in GBM, IDH-wildtype: transcriptional regulation and inhibition of YB-1 ubiquitination. Cell Death Discov. 12, 168 (2026). https://doi.org/10.1038/s41420-026-03035-9
Keywords: glioblastoma, paxillin, STAT3 signaling, YB-1, temozolomide resistance