Clear Sky Science · en
Absolute dynamic and relative static: the relationship of glycolysis and OXPHOS in cancer development
Why Cancer’s Energy Choices Matter
Cancer cells are like extreme endurance athletes: they must constantly fuel themselves to grow, spread, and evade treatment. This review explains how tumor cells tap into two main energy systems—one fast and one efficient—and, more importantly, how they mix and switch between them. Understanding these shifting energy strategies offers a new way to think about diagnosing and treating cancer over the entire course of the disease.
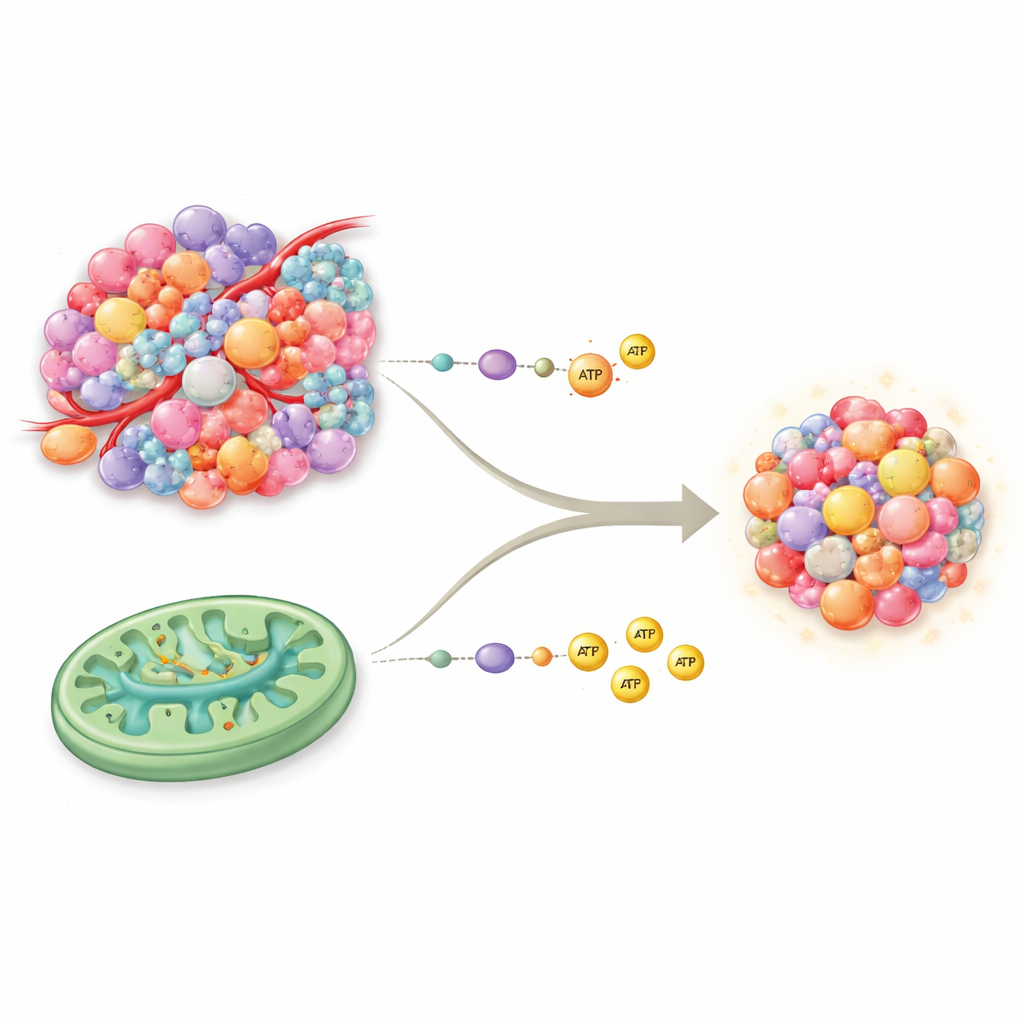
Two Main Ways Cells Make Fuel
Our cells typically make energy through two core routes. Glycolysis is the quick, “cash-in-hand” system that breaks down sugar rapidly but yields relatively little energy per unit. Oxidative phosphorylation, or OXPHOS, runs inside mitochondria and works more like a high-efficiency power plant, producing much more energy but demanding good oxygen and nutrient supply. For decades, cancer was thought to rely almost exclusively on glycolysis, even when oxygen was present—a pattern known as the Warburg effect. New evidence overturns this simple view: many tumors have fully functioning mitochondria and can use OXPHOS and the tricarboxylic acid (TCA) cycle alongside glycolysis, giving them a richer and more flexible energy toolkit than previously appreciated.
Many Cell Types, Many Fuel Choices
A tumor is not a uniform mass. It is a small ecosystem containing cancer cells, support cells, blood vessels, and immune cells, each with distinct fuel needs. Some cancer cells live close to blood vessels and enjoy ample oxygen and nutrients; others are buried deep in poorly supplied regions. As a result, cells within the same tumor can favor glycolysis, rely mainly on OXPHOS, or use both at once. This metabolic variety—called heterogeneity—arises from differences in blood flow, nutrient levels, cell type, and signals from the surrounding environment. On top of that, tumors often display metabolic symbiosis: glycolytic cells export lactate as a waste-like product, which neighboring OXPHOS-reliant cells import and burn as a valuable fuel. Similar lactate sharing occurs between cancer cells and support cells such as fibroblasts, blood vessel cells, and immune-suppressive cells, collectively helping the tumor thrive and resist therapy.
Forever Changing: How Tumor Energy Shifts Over Time
Tumor metabolism is not only diverse at any given moment; it is also highly dynamic. As tumors grow, they encounter changing conditions such as low oxygen, acid buildup, lack of nutrients, and variations in tissue stiffness. Under harsh, low-oxygen conditions, many cancer cells shift toward glycolysis. Under acid build-up or sugar starvation, they may swing back toward heavier use of OXPHOS. The same flexibility appears during key biological milestones. Rapidly dividing cells lean on glycolysis to generate building blocks for new DNA, fats, and membranes. Cells that are resting or circulating in the blood often depend more on OXPHOS. During invasion and metastasis, cancer cells adjust their fuel choice repeatedly: some steps favor glycolysis, others favor OXPHOS, and metastatic cells that colonize new organs tune their metabolism to match the “soil” of the destination tissue.
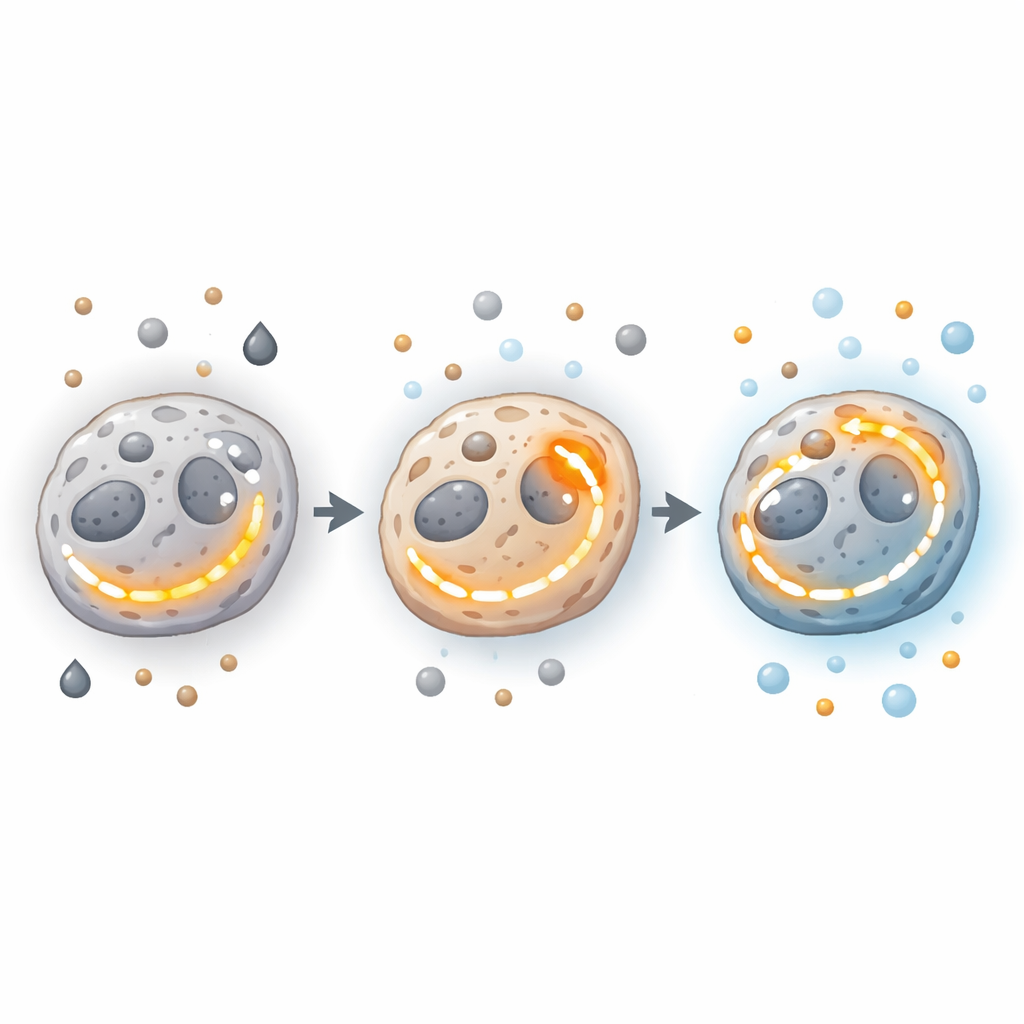
Genes, Enzymes, and Power Plants Behind the Switch
Deep in the machinery of the cell, genes and enzymes steer these energy decisions. Oncogenes and tumor suppressors can push cells toward glycolysis or OXPHOS, or enable them to ramp up both. Mutations or changes in key TCA enzymes, such as those that handle citrate, succinate, or isocitrate, can tilt the balance, sometimes driving cancer-promoting signals like blood vessel growth and invasion. Enzymes that sit at crucial crossroads—such as pyruvate kinase M2, which helps decide whether sugar-derived carbon becomes energy or building material—tie glycolysis and mitochondrial function together. Meanwhile, differences in mitochondrial number, structure, and performance across tumor regions further widen the spectrum of possible energy states.
Rethinking Treatment Through a Moving Energy Target
Because tumor cells can shift between glycolysis and OXPHOS, simply blocking one pathway is often not enough; the cancer may reroute its fuel use and survive. The authors argue that energy metabolism in cancer is only briefly stable and is fundamentally, and constantly, changing. They propose viewing tumors through an “energy metabolism spectrum” that must be measured over time, not just once. In practice, this could mean tracking a tumor’s fuel preferences during different disease stages and pairing inhibitors of glycolysis and OXPHOS in tailored combinations. Such dynamic, metabolism-guided treatment strategies might better cut off the tumor’s power supply, limit its ability to adapt, and improve long-term control of the disease.
Citation: Bao, X., Hou, B., Guo, Z. et al. Absolute dynamic and relative static: the relationship of glycolysis and OXPHOS in cancer development. Cell Death Discov. 12, 136 (2026). https://doi.org/10.1038/s41420-026-02992-5
Keywords: cancer metabolism, glycolysis, oxidative phosphorylation, tumor microenvironment, metabolic plasticity