Clear Sky Science · en
Exosomal S100A9 promotes lung metastasis of adenoid cystic carcinoma via activating cancer-associated fibroblasts
How a Silent Salivary Cancer Finds Its Way to the Lungs
Adenoid cystic carcinoma is a slow-growing but stubborn cancer that often starts in the salivary glands and later appears in the lungs, sometimes years after the first tumor is removed. For patients and families, this delayed spread is frightening and difficult to predict or prevent. This study unpacks how cells in these tumors secretly prepare distant lung tissue for cancer invasion, revealing a hidden conversation between tumor cells and the surrounding support cells that may finally offer new points for treatment.
The Cancer That Lingers and Returns
Adenoid cystic carcinoma (ACC) is a cancer of the glands in the head and neck, especially the salivary glands. Surgery and radiation can often control the main tumor, yet many patients eventually develop lung metastases, which are the leading cause of death. Current drugs for widespread ACC are limited, in part because scientists do not fully understand how cells from the original tumor manage to settle and grow in the lungs. The authors of this paper set out to identify the key signals that allow ACC cells to reshape their surroundings and create a welcoming “soil” for future lung tumors.
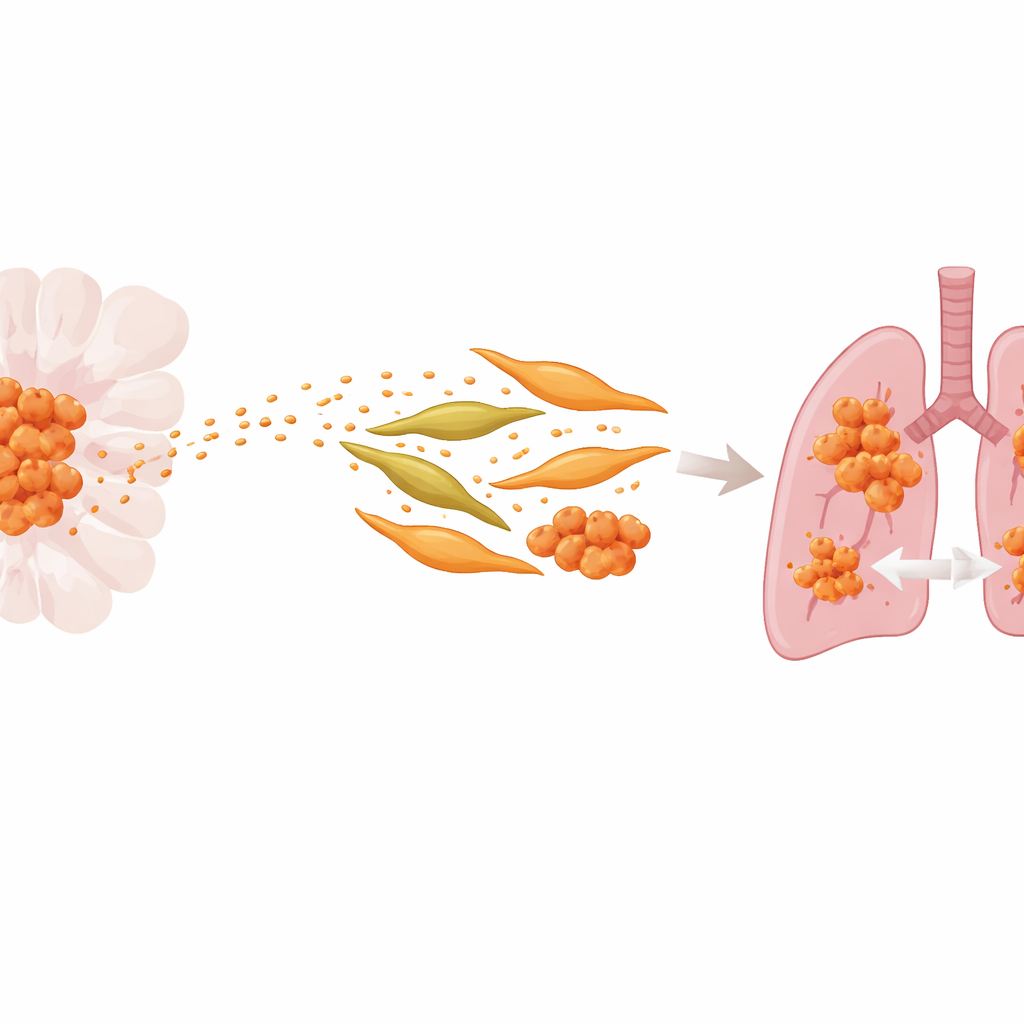
Tiny Packages with a Big Impact
The researchers focused on exosomes, tiny membrane-bound bubbles that cancer cells release into the bloodstream. These vesicles carry proteins and genetic material that can reprogram other cells at a distance. By comparing a standard ACC cell line with a version specially selected for strong lung-spreading ability, the team found that both the aggressive cells and their exosomes were enriched in a protein called S100A9, which is linked to inflammation. Patients whose tumors and blood exosomes contained more S100A9 were more likely to have lung metastases and shorter periods without disease returning, suggesting that this protein helps drive the cancer’s spread.
Rewiring the Tumor’s Support Crew
Using single-cell RNA sequencing, the authors built a detailed map of all the cell types inside ACC tumors and nearby normal glands. They discovered intense communication between the tumor’s epithelial cells and nearby fibroblasts, the structural cells that produce connective tissue. In cancer, these fibroblasts can be “recruited” and turned into cancer-associated fibroblasts (CAFs), which stiffen tissue, fuel inflammation, and help cancer cells move. The study showed that S100A9-rich exosomes from aggressive ACC cells could turn normal fibroblasts into activated CAFs: they became more contractile, more mobile, and began producing enzymes and inflammatory molecules that remodel tissue and promote tumor growth.
From Local Signals to Lung Colonies
The team then asked whether these exosome-activated fibroblasts actually make it easier for ACC to colonize the lungs. In mouse models, injecting S100A9-loaded exosomes into the bloodstream primed the lungs: fibroblasts there switched on activation markers, and lung tissue began to express factors associated with new blood vessel growth and matrix breakdown. When ACC cells were later introduced, mice pretreated with S100A9 exosomes developed more and larger lung metastases. Further analyses showed that this process depended on a chain of inflammatory signals centered on the messenger IL-17, along with TNF and NF-κB, which together amplified fibroblast activation and reshaped the lung microenvironment.
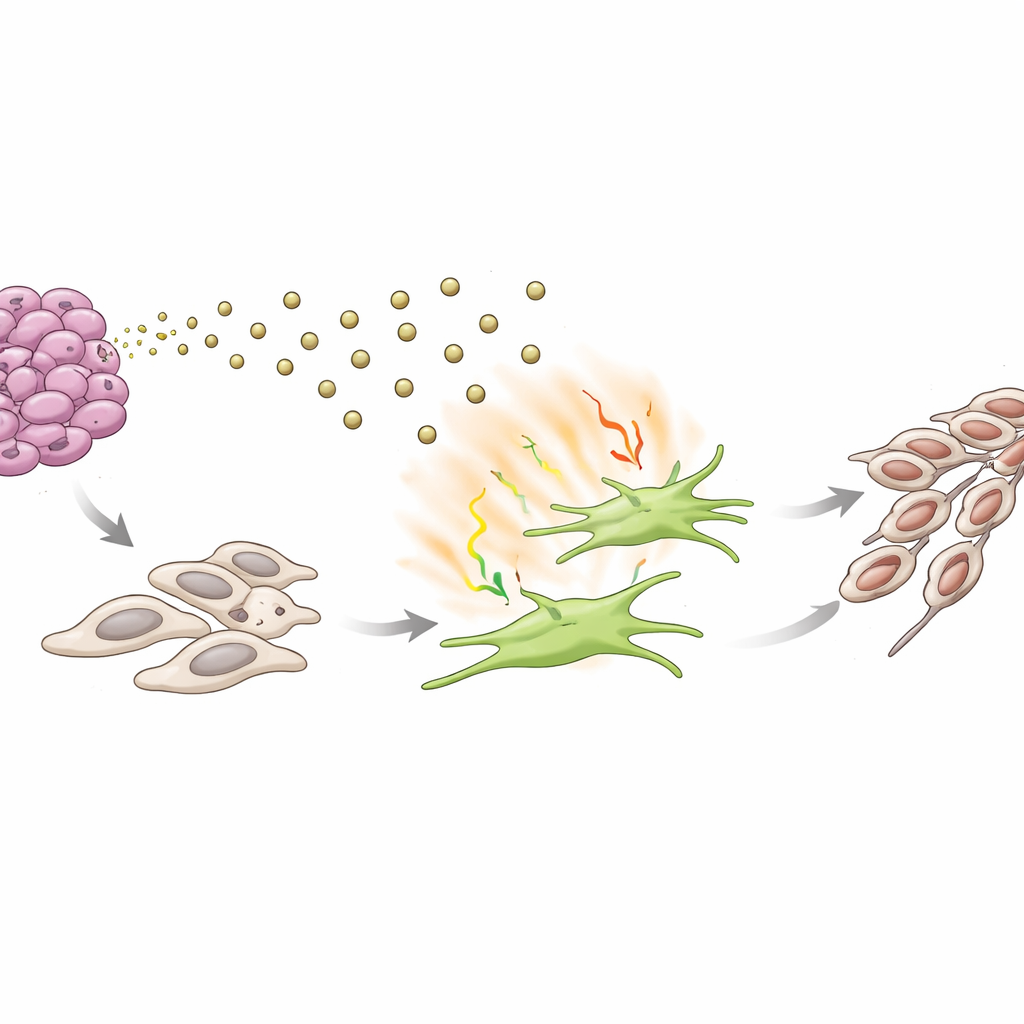
A Vicious Circle That Drives Cancer Shape-Shifting
In laboratory co-culture systems, fibroblasts that had been “educated” by S100A9-rich exosomes, or by this IL-17–driven signaling pathway, made nearby ACC cells more aggressive. The cancer cells became better at moving, invading through barriers, growing into colonies, and adopting a more mobile, spindle-like form associated with an epithelial–mesenchymal transition, a key step toward metastasis. Blocking IL-17 with a neutralizing antibody reversed many of these changes, while adding purified IL-17 alone pushed ACC cells toward this more invasive state. Together, these findings outline a self-reinforcing loop: tumor cells send S100A9 in exosomes to activate fibroblasts; activated fibroblasts release IL-17 and other signals that, in turn, make tumor cells more prone to spread.
Breaking the Hidden Communication Loop
To a layperson, the message is that ACC does not spread to the lungs by chance. Instead, the original tumor secretly trains support cells and distant tissues to help it grow elsewhere, using tiny protein-filled parcels as messengers. This work identifies S100A9 and the downstream IL-17 signaling route as central hubs in that process. By targeting this communication axis—either blocking S100A9, dampening IL-17 signaling, or disrupting the activation of fibroblasts—future therapies may be able to interrupt the “set-up phase” of lung metastasis, giving clinicians new ways to keep this quiet but dangerous cancer from returning far from its starting point.
Citation: Chen, Cw., Zhang, Sr., Yan, Ym. et al. Exosomal S100A9 promotes lung metastasis of adenoid cystic carcinoma via activating cancer-associated fibroblasts. Cell Death Discov. 12, 120 (2026). https://doi.org/10.1038/s41420-026-02991-6
Keywords: adenoid cystic carcinoma, exosomes, cancer-associated fibroblasts, lung metastasis, S100A9 IL-17 axis