Clear Sky Science · en
Navigating the complexities of ferroptosis in pancreatic ductal adenocarcinoma: roles, mechanisms and potential applications
Why this deadly cancer and a new form of cell death matter
Pancreatic ductal adenocarcinoma (PDAC) is one of the deadliest cancers; most patients are diagnosed late, treatments rarely work for long, and survival is measured in months rather than years. This review article explores a relatively recent discovery—ferroptosis, a kind of cell death powered by iron and fat damage—and asks whether we can turn it into a weapon against PDAC. For readers, it offers a window into how researchers are trying to outsmart an otherwise stubborn tumor by exploiting its own metabolic weaknesses.
Iron, fats, and rusting cancer cells
Ferroptosis is unlike the better-known forms of cell death such as apoptosis. Instead of neatly dismantling a cell, ferroptosis essentially makes the cell “rust from within.” Iron inside the cell fuels highly reactive molecules that attack certain fats in cell membranes, causing them to break apart. PDAC cells, which demand extra iron and energy to sustain rapid growth, are particularly vulnerable to this kind of damage. To survive, they rely on several built‑in defense systems that mop up these dangerous byproducts. The review describes four major protective circuits: a glutathione–GPX4 system that detoxifies damaged fats, an FSP1–CoQ and vitamin K system at the cell membrane, a DHODH–CoQ system in mitochondria, and a BH4‑based pathway that preserves delicate fats. Together these act like layered fire‑safety systems designed to keep ferroptosis at bay.
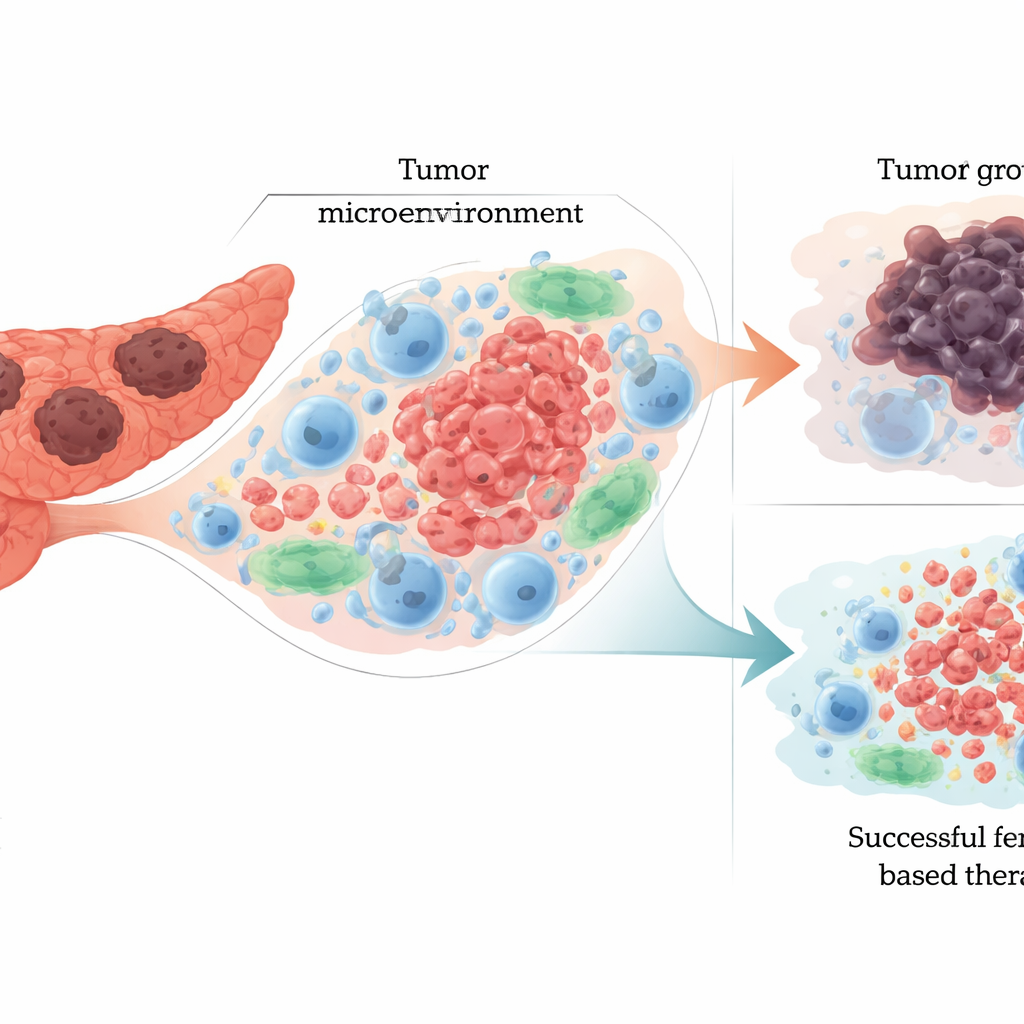
How pancreatic tumors tilt the balance
PDAC rewires basic metabolism—iron handling, fat use, and responses to oxidative stress—to favor growth and resist treatment. Tumor cells often import more iron, store it differently, and release it through processes like ferritin breakdown, which can both feed growth and prime cells for ferroptosis. They also change their fat composition: increasing certain polyunsaturated fats that are easy to oxidize, but simultaneously boosting enzymes and antioxidant systems that repair or shield membranes. Key cancer genes such as KRAS and p53, along with pathways like Keap1–Nrf2 and Hippo, help tune this balance, sometimes making cells more sensitive to ferroptosis and other times hardening their defenses. The article explains that these same pathways drive PDAC’s aggressiveness and drug resistance, suggesting that pushing them toward ferroptosis might selectively harm tumor cells.
Friend and foe: ferroptosis in the tumor neighborhood
Ferroptosis does not occur in isolation; it unfolds inside a crowded tumor microenvironment filled with immune cells, connective‑tissue cells, and blood vessels. When cancer cells undergo ferroptosis, they release iron, oxidized fats, and distress signals that can cut both ways. On the helpful side, these signals can wake up certain immune cells, prompting them to attack the tumor. On the harmful side, they can push other immune cells into a suppressive, tumor‑nurturing state or directly kill key defenders such as natural killer cells, B cells, and cytotoxic T cells. The review emphasizes this dual role: ferroptosis can shrink tumors by killing cancer cells, yet also remodel the surrounding ecosystem in ways that blunt immunity and encourage progression if not carefully controlled.

From biological insight to diagnosis and therapy
Because ferroptosis touches iron, fats, and stress responses, it leaves behind measurable molecular footprints. Researchers have begun to identify ferroptosis‑related genes, proteins, and RNA molecules in blood and tumor tissue that correlate with PDAC presence, stage, and patient outcomes. Elevated ferritin, altered iron‑regulating hormones, and specific gene signatures may help refine diagnosis and prognosis. On the treatment side, multiple experimental drugs and combinations aim to tip PDAC cells into ferroptosis—either by blocking their defenses (such as GPX4 or the cystine transporter system Xc−) or by overloading them with iron and reactive molecules. Some strategies pair ferroptosis inducers with standard chemotherapy like gemcitabine to overcome resistance, while others package these agents into nanoparticles and biomimetic vesicles to deliver them more precisely and reduce collateral damage.
Promise, risks, and the road ahead
The authors conclude that targeting ferroptosis could open a new front against PDAC by exploiting the very traits that make this cancer so difficult to treat. Yet they caution that ferroptosis is a double‑edged sword. The same mechanisms that kill tumor cells can injure healthy stem cells, liver and kidney tissue, and crucial immune cells, potentially causing serious side effects and even worsening outcomes if misapplied. Future progress will depend on mapping exactly how ferroptosis interacts with PDAC genetics, metabolism, and the immune microenvironment; developing delivery systems that focus its effects on tumors; and validating ferroptosis‑based biomarkers and drug combinations in carefully designed clinical trials. If these challenges can be met, ferroptosis may become a key component of more precise, effective, and personalized therapies for pancreatic cancer.
Citation: Xiao, Y., Wang, W., Wang, G. et al. Navigating the complexities of ferroptosis in pancreatic ductal adenocarcinoma: roles, mechanisms and potential applications. Cell Death Discov. 12, 117 (2026). https://doi.org/10.1038/s41420-026-02987-2
Keywords: pancreatic cancer, ferroptosis, tumor microenvironment, cancer metabolism, nanoparticle therapy