Clear Sky Science · en
APOA2-mediated endothelial mesenchymal transition and cancer lipid metabolism reprogramming confers antiangiogenic drug resistance through TGF-β
Why this discovery matters for patients
Doctors increasingly use drugs that starve liver tumors by cutting off their blood supply, yet many patients see their cancers stop responding after an initial benefit. This study asks a pressing question: why do some liver cancers become resistant to these blood-vessel-blocking treatments, and how might we outsmart them? The researchers uncover a hidden partnership between a blood fat protein and a powerful growth signal that lets tumors rewire both their blood vessels and their energy use, pointing to a different way to treat a hard-to-cure cancer.
A tumor that learns to live without its usual lifeline
Hepatocellular carcinoma, the most common form of liver cancer, depends heavily on new blood vessels to grow. Standard drugs for advanced disease are designed to block a key vessel-growth pathway centered on VEGF and its receptor VEGFR2. Yet in many patients, tumors keep thriving despite these medicines. By comparing tumor samples from patients who did or did not respond to the antiangiogenic drug apatinib, the authors found that one gene, APOA2, stood out. Tumors from resistant patients had markedly higher levels of APOA2, a protein best known for its role in transporting fats in the blood. This raised the possibility that APOA2 might help tumors evade vessel-targeting drugs.
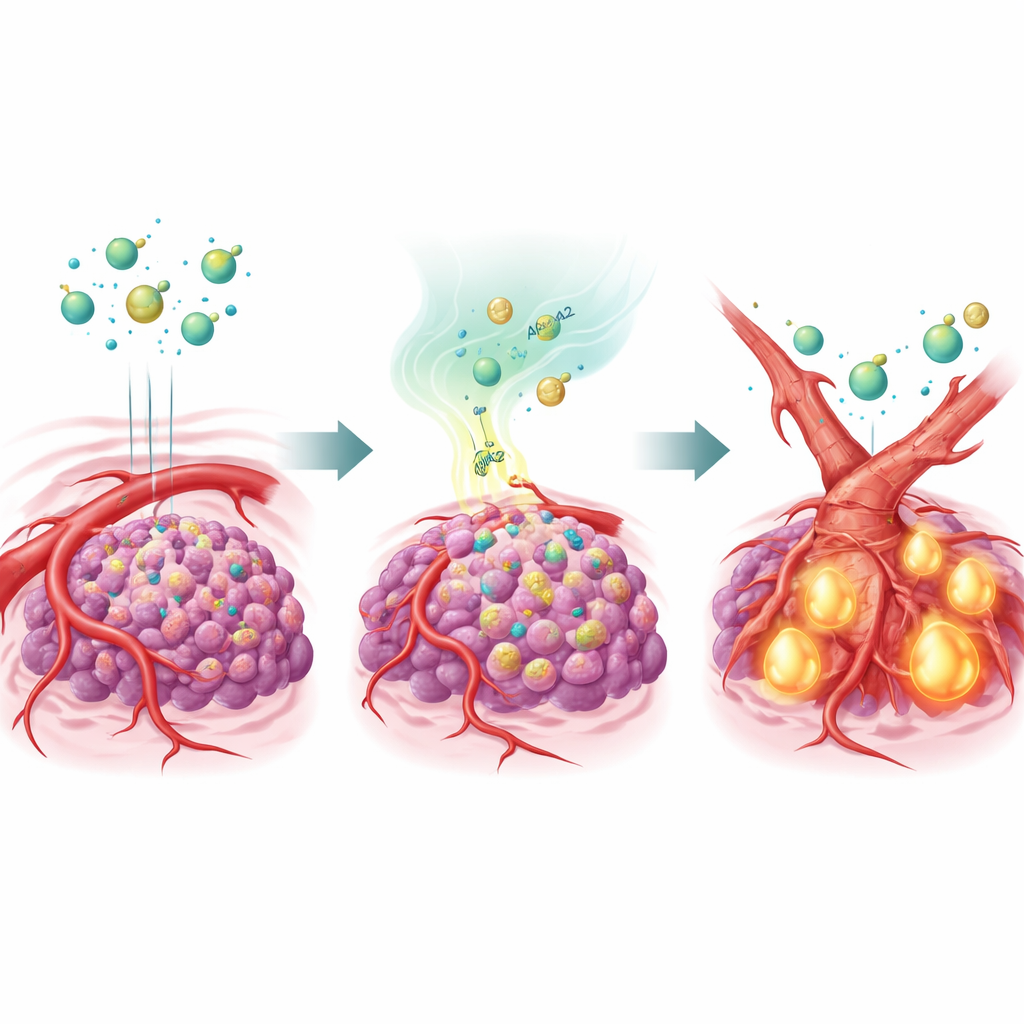
A fat-transport protein that reshapes blood vessels
To test this idea, the team engineered liver cancer cells to overproduce APOA2 and implanted them into mice. When treated with several different antiangiogenic drugs, these APOA2-rich tumors largely ignored therapy: their blood vessels persisted, and the cancers continued to grow. Detailed analysis showed that a master signal called TGF-β was strongly switched on in these tumors, and its levels were elevated in both the tumor environment and the animals’ blood. TGF-β acted on the cells lining blood vessels, pushing them to change identity in a process called endothelial-to-mesenchymal transition. As they shifted toward a more scar-like, mesenchymal state, these cells lost much of their VEGFR2, the very handle that antiangiogenic drugs are designed to grab. In effect, the vessels remodeled themselves into a form the drugs could no longer target.
A metabolic detour that keeps cancer cells fueled
The story did not end at the vessel wall. The same TGF-β signal driven by APOA2 also reprogrammed how cancer cells fuel themselves. Instead of relying mainly on sugar breakdown, APOA2-high cells began to pull in more free fatty acids from their surroundings and burn them in their mitochondria through fatty acid oxidation. Experiments measuring nutrient use and oxygen consumption showed these cells became more dependent on fat as an energy source and produced more cellular fuel (ATP). This shift allowed tumor cells to keep dividing rapidly and avoid cell death, even when blood supply was under pressure from antiangiogenic treatment.
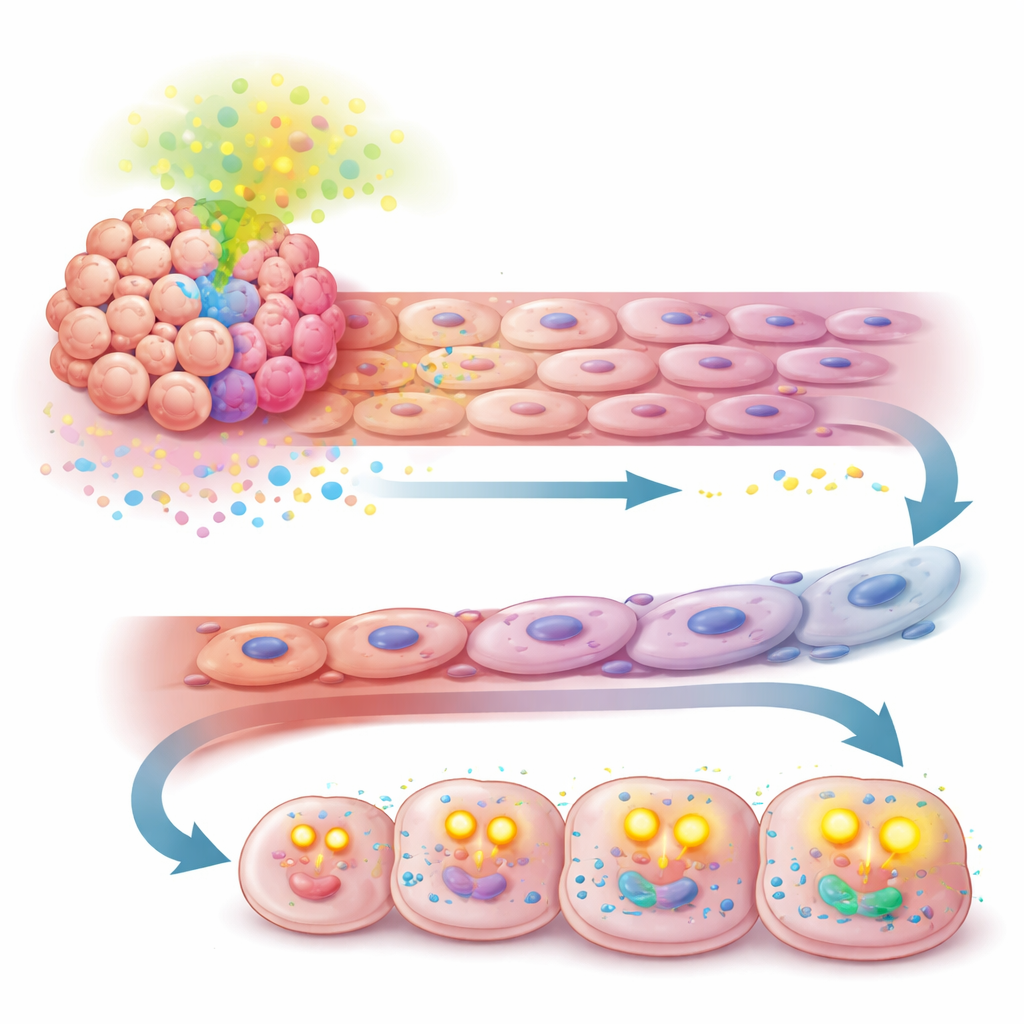
Turning a weakness into a new treatment target
Crucially, when the researchers blocked TGF-β, the advantages conferred by APOA2 unraveled. Inhibiting this signal in mouse models reduced the abnormal vessel changes, restored sensitivity to therapy, and slowed tumor growth. Even more promising, a dual antibody that simultaneously blocks TGF-β and the immune checkpoint PD-1 worked better than either approach alone in APOA2-high tumors. These results suggest that, in a subset of liver cancers marked by high APOA2, the disease has quietly switched its dependency from the usual VEGF pathway to a TGF-β–centered route that sustains both blood vessels and tumor metabolism.
What this means for future liver cancer care
For a layperson, the key message is that some liver tumors escape current blood-vessel drugs by rewiring both their plumbing and their power supply. A fat-associated protein, APOA2, cranks up the TGF-β signal, which in turn disguises blood vessels so existing drugs cannot see them and teaches cancer cells to live off fats instead of sugar. The study suggests that testing tumors for APOA2 could flag patients who are unlikely to benefit from standard antiangiogenic drugs but might respond to medicines that block TGF-β, especially when combined with modern immunotherapy. In short, by exposing how tumors adapt, this work points to a more tailored strategy that attacks both the vessels and the energy source these resilient cancers depend on.
Citation: Zhang, S., Fu, Z., Zhu, F. et al. APOA2-mediated endothelial mesenchymal transition and cancer lipid metabolism reprogramming confers antiangiogenic drug resistance through TGF-β. Cell Death Discov. 12, 119 (2026). https://doi.org/10.1038/s41420-026-02984-5
Keywords: hepatocellular carcinoma, antiangiogenic resistance, APOA2, TGF-beta signaling, cancer lipid metabolism