Clear Sky Science · en
Hepatocyte TonEBP promotes metabolic stress-induced hepatic fibroinflammation involving transcriptional activation of ELR⁺ CXC chemokines
Why this liver story matters
Many people with obesity or type 2 diabetes quietly develop fat in their liver, a condition that can progress to a dangerous stage marked by inflammation and scarring. This advanced form, known as metabolic dysfunction-associated steatohepatitis (MASH), can lead to cirrhosis, liver failure, or liver cancer. The study summarized here uncovers how ordinary liver cells themselves help turn harmless fat buildup into damaging inflammation, pointing to a new molecular switch that might be targeted to stop or slow this process.
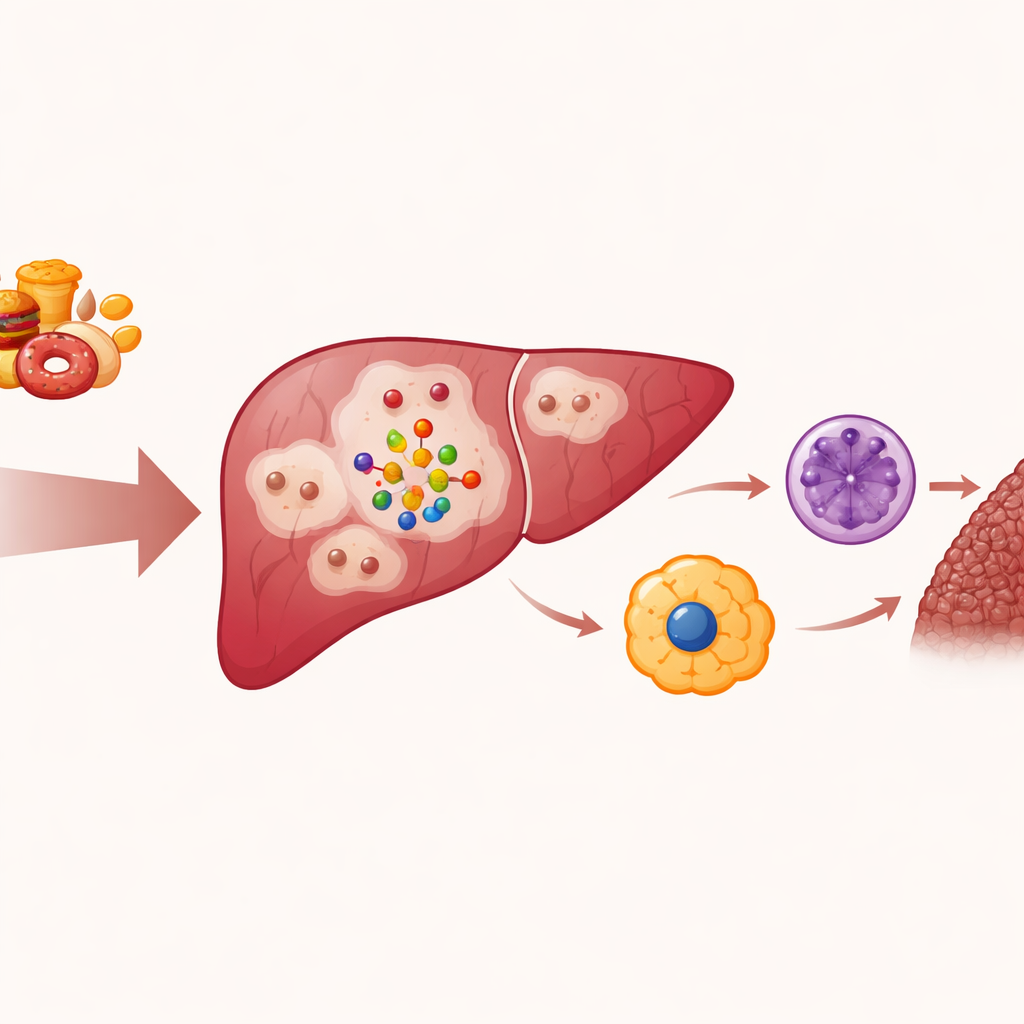
From fatty liver to scarred organ
Doctors have long seen that simple fatty liver does not always stay simple. In some people, it evolves into MASH, where liver tissue becomes inflamed and scarred. A key mystery has been how the strain of excess nutrients and fat inside liver cells turns into a call for immune cells that worsen damage. The authors focus on hepatocytes, the main working cells of the liver. Under metabolic overload, these cells do more than just store fat; they begin to send distress signals that summon waves of immune cells such as neutrophils and macrophages. These visitors release toxic molecules and help lay down scar tissue, driving the liver toward chronic disease.
A stress sensor inside liver cells
The researchers homed in on a protein called TonEBP, a stress-responsive switch inside cells that can tune many genes on or off. TonEBP was already known to respond to changes in salt balance and to be involved in inflammation and metabolic diseases in other tissues. What was unclear was its role inside hepatocytes during metabolic stress. To test this, the team engineered mice whose hepatocytes lacked TonEBP, while other cell types remained normal. They then exposed these mice and normal controls to several diets that mimic different flavors of human fatty liver disease: one very harsh, nutrient-deficient diet that rapidly causes fat, injury, and fibrosis, and another rich in fat, cholesterol, and sugar that more closely resembles a Western diet.
Blocking the switch calms the liver
Across these models, taking TonEBP out of hepatocytes consistently protected the liver. Even when fed harmful diets, these mice had less liver scarring, fewer signs of cell injury in the blood, and milder fat accumulation. Detailed gene analysis showed that, in normal mice, metabolic stress flips on large networks of genes linked to inflammation and fibrous tissue formation while damping down healthy metabolic pathways. In contrast, livers missing hepatocyte TonEBP looked more like those from healthy animals: inflammatory and scarring programs were muted, and metabolic functions were partly restored. Importantly, the numbers of neutrophils and macrophages entering the liver were markedly lower, suggesting that the early signals that normally recruit these cells were reduced.
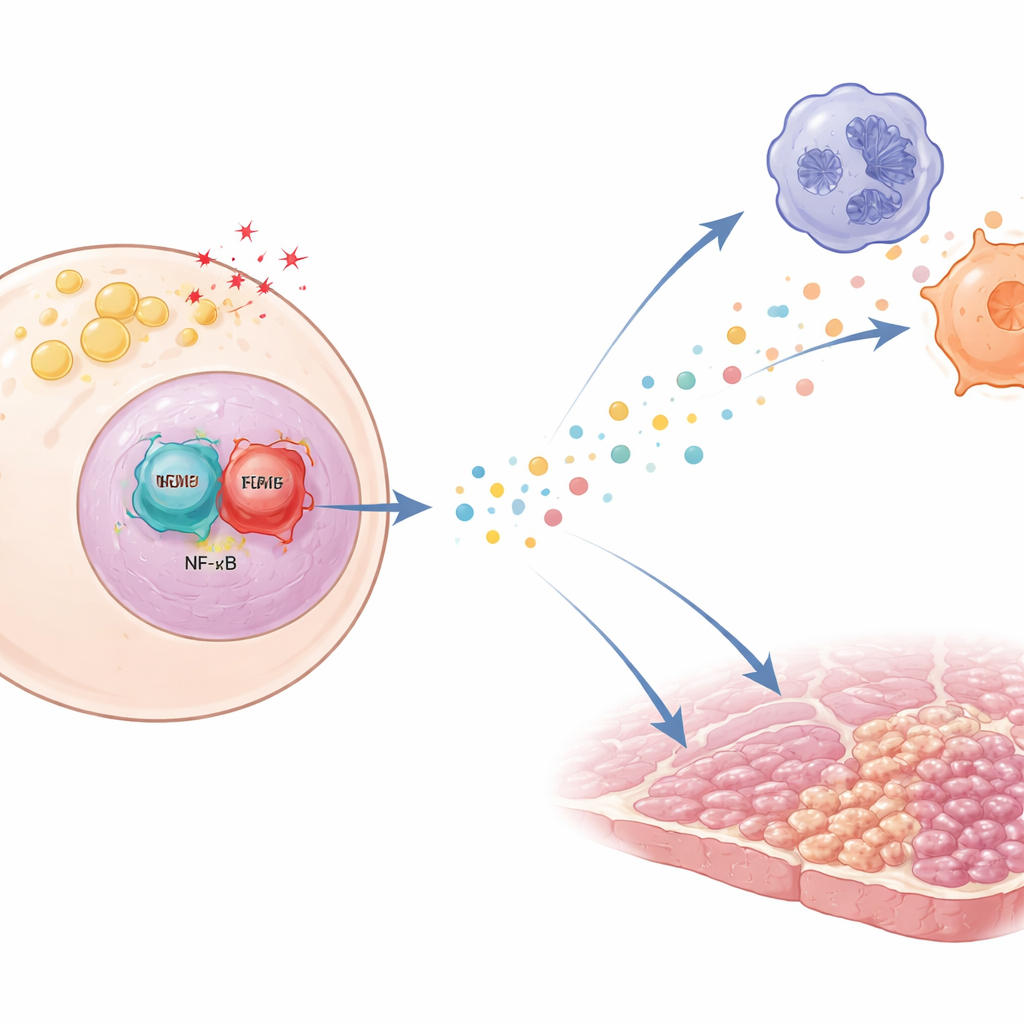
A chemical call that draws immune cells
To understand how TonEBP changes liver behavior at the molecular level, the team turned to human and mouse liver cells grown in dishes and stressed them with palmitate, a fatty acid that accumulates in diseased livers, as well as with inflammatory and oxidative stimuli. Under these stresses, hepatocytes sharply increased production of a group of small messenger proteins called ELR+ CXC chemokines, which are powerful beacons for neutrophils and can also attract macrophages. When TonEBP was lowered by genetic tools, both the gene activity and the release of these chemokines dropped, and immune-like cells became less willing to migrate toward the hepatocyte culture. Conversely, boosting TonEBP made the chemokine surge stronger.
The teamwork behind the signal
Digging further, the authors found that TonEBP does not act alone. It partners with another master regulator of inflammation, NF-κB. In stressed hepatocytes, TonEBP helps bring NF-κB to the stretches of DNA that control chemokine genes, turning them on more efficiently. When TonEBP is missing, or when a small molecule is used to disrupt its physical interaction with NF-κB, NF-κB can no longer bind these regions well, and chemokine production falls. This cooperation was seen both in cultured cells and in livers of diet-stressed mice, firmly linking the TonEBP–NF-κB duo to the chemokine wave that draws immune cells into the liver.
What this means for future treatments
For non-specialists, the bottom line is that the study reveals a concrete chain of events inside liver cells: metabolic overload activates a stress switch (TonEBP) that teams up with an inflammatory regulator (NF-κB) to crank out chemical signals that summon damaging immune cells and promote scarring. When this switch is removed or its partnership is disrupted, the liver is quieter, less inflamed, and less fibrotic, even under unhealthy dietary conditions. While directly blocking TonEBP everywhere in the body could have side effects, the work suggests that carefully targeted therapies—aimed at hepatocytes or at the TonEBP–NF-κB interaction itself—might one day help halt or slow the progression from simple fatty liver to serious, life-threatening liver disease.
Citation: Lee, J.H., Song, H., Yoo, E.J. et al. Hepatocyte TonEBP promotes metabolic stress-induced hepatic fibroinflammation involving transcriptional activation of ELR⁺ CXC chemokines. Cell Death Discov. 12, 116 (2026). https://doi.org/10.1038/s41420-026-02978-3
Keywords: fatty liver disease, liver inflammation, chemokines, TonEBP, metabolic stress