Clear Sky Science · en
KCTD1 stabilizes c-Myc to upregulate PD-L1 and suppress anti-tumor immunity in hepatocellular carcinoma
Why this matters for future cancer care
Many people living with liver cancer do not benefit from today’s most exciting immunotherapies, which are designed to unleash the body’s own T cells against tumors. This study explores why a major form of liver cancer, hepatocellular carcinoma, can still hide from immune attack even during treatment—and identifies a new molecular switch, called KCTD1, that could make existing immunotherapy drugs work better.
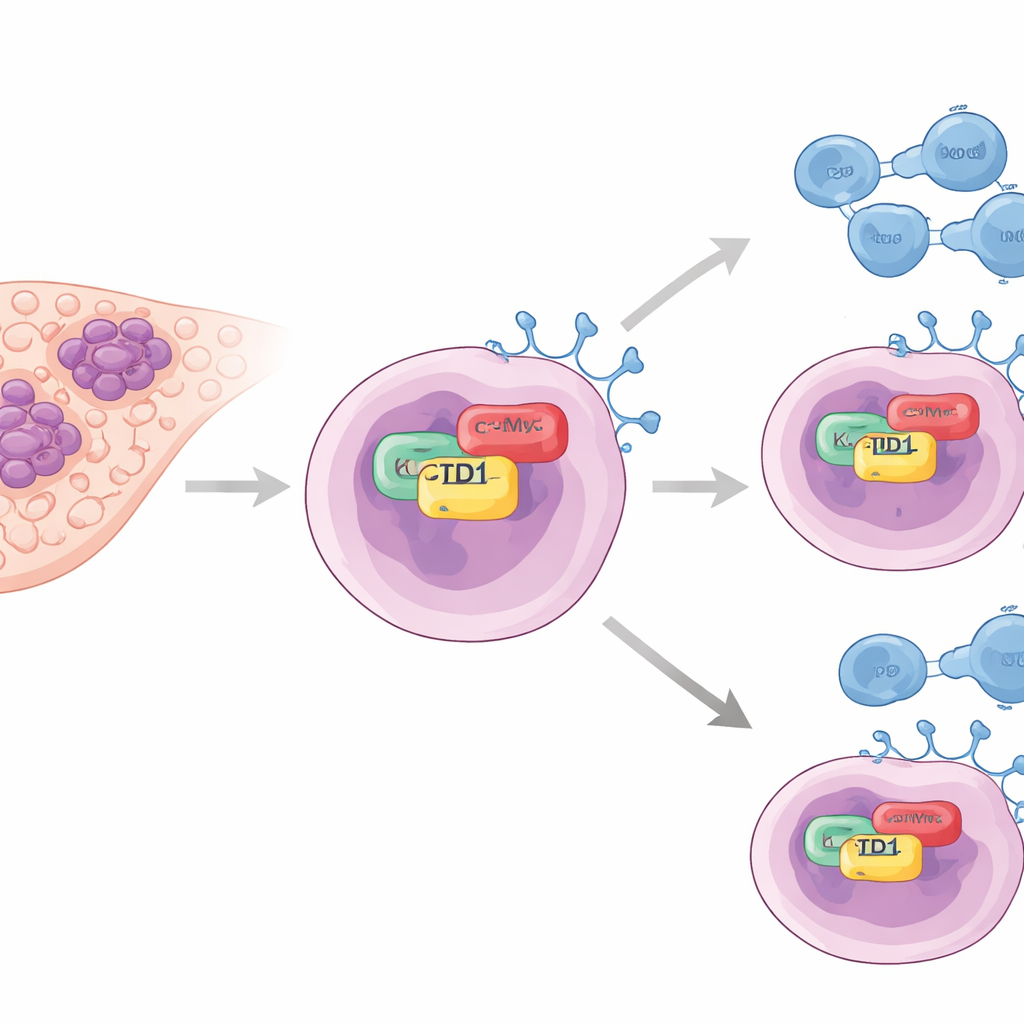
A hidden helper of tumor “off switches”
The authors focused on a protein named KCTD1, previously linked to liver tumor growth but not well studied in immune escape. In tumor samples from patients, they found that KCTD1 levels were markedly higher in cancerous liver tissue than in normal liver. Importantly, tumors rich in KCTD1 also had high levels of PD-L1, a surface protein on cancer cells that binds to PD-1 on T cells and effectively tells the immune system to stand down. Patients whose tumors had lower KCTD1 tended to live longer, suggesting that this protein is connected to both disease severity and the strength of the tumor’s immune shield.
Connecting the molecular dots inside cancer cells
To understand how KCTD1 boosts PD-L1, the team turned to liver cancer cell lines grown in the lab. When they reduced KCTD1, PD-L1 protein levels dropped and the remaining PD-L1 broke down more quickly, indicating that KCTD1 helps stabilize PD-L1 rather than simply turning on its gene. Using biochemical pull-down experiments and fluorescent microscopy, they showed that KCTD1 physically binds another key cancer driver, the oncoprotein c-Myc, inside the cell nucleus. This interaction occurs through specific contact regions on both proteins and leads to more stable c-Myc, which in turn enhances PD-L1 production. When c-Myc alone was reduced, PD-L1 fell even if KCTD1 was abundant, placing c-Myc as the crucial middle link in a KCTD1–c-Myc–PD-L1 chain.
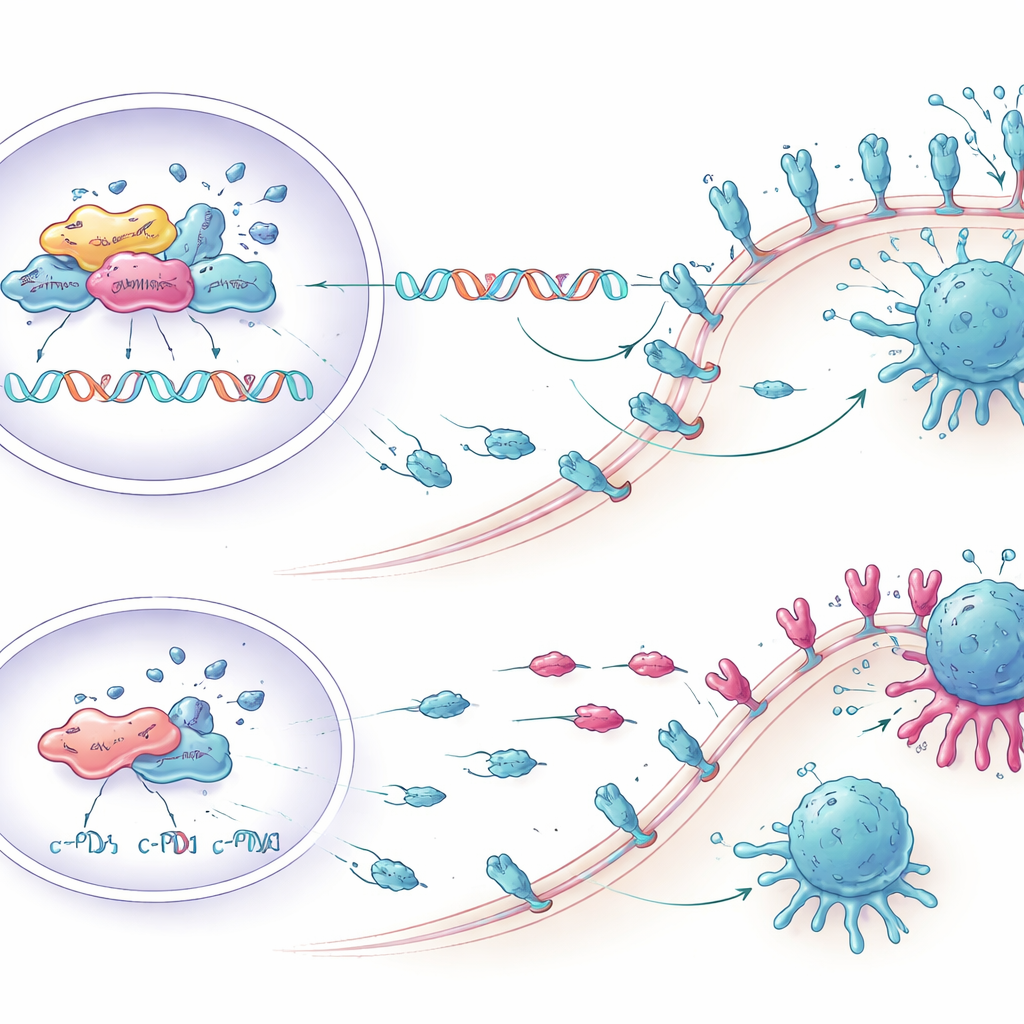
Reawakening T cells in the lab
The researchers then asked whether lowering KCTD1 could actually strengthen immune attack. They co-cultured human liver cancer cells with immune cells taken from blood donors. Tumor cells engineered to have less KCTD1, and therefore less PD-L1, triggered stronger responses from CD8 T cells—the immune system’s main killers of cancer cells. More of these T cells produced inflammatory molecules such as TNF-α and interferon-gamma, showed higher markers of growth and activation, and bore fewer signs of exhaustion. As a result, more tumor cells underwent programmed cell death when KCTD1 was suppressed, demonstrating that the molecular changes translated into real gains in immune killing.
Putting the mechanism to the test in mice
To see if the same pattern holds in living organisms, the team implanted mouse liver cancer cells lacking KCTD1 directly into the livers of mice. Tumors with reduced KCTD1 formed fewer and smaller nodules, showed slower cell division, and had more cancer cells undergoing cell death. These tumors also expressed less c-Myc and PD-L1. Removing CD8 T cells from the mice erased much of this benefit, indicating that the tumor slowdown depended heavily on T cell activity. Finally, when the researchers combined KCTD1 knockdown with an anti–PD-1 antibody—an existing immune checkpoint drug—the mice had the smallest tumor burden and the highest levels of CD4 and CD8 T cells infiltrating their tumors.
What this could mean for patients
Taken together, the work reveals KCTD1 as a central coordinator that stabilizes c-Myc, boosts PD-L1 on liver cancer cells, and blunts CD8 T cell attack. Disrupting this pathway makes tumors both more vulnerable to the immune system and more responsive to PD-1–blocking drugs in mice. For patients, this suggests that drugs aimed at KCTD1 or its contact surface with c-Myc could, in the future, be paired with current immunotherapies to help more people with liver cancer benefit from durable immune control of their disease.
Citation: Zhong, D., Long, S., Dai, Y. et al. KCTD1 stabilizes c-Myc to upregulate PD-L1 and suppress anti-tumor immunity in hepatocellular carcinoma. Cell Death Discov. 12, 129 (2026). https://doi.org/10.1038/s41420-026-02975-6
Keywords: hepatocellular carcinoma, tumor immunotherapy, PD-1 PD-L1 pathway, c-Myc signaling, T cell anti-tumor immunity