Clear Sky Science · en
Molecular insights into Silodosin’s anti-cancer effects: a promising repurposing strategy for breast cancer
Why an old drug is getting new attention
Breast cancer remains one of the most common and deadly cancers in women, and many patients eventually run out of effective treatment options. This study explores whether silodosin, a pill already used safely for urinary problems in men, could be repurposed to fight breast cancer. Because it is an approved, affordable drug with a known safety record, showing that it can slow or kill breast cancer cells could speed up the path to new therapies, especially for people with aggressive disease.
A familiar medicine in a new role
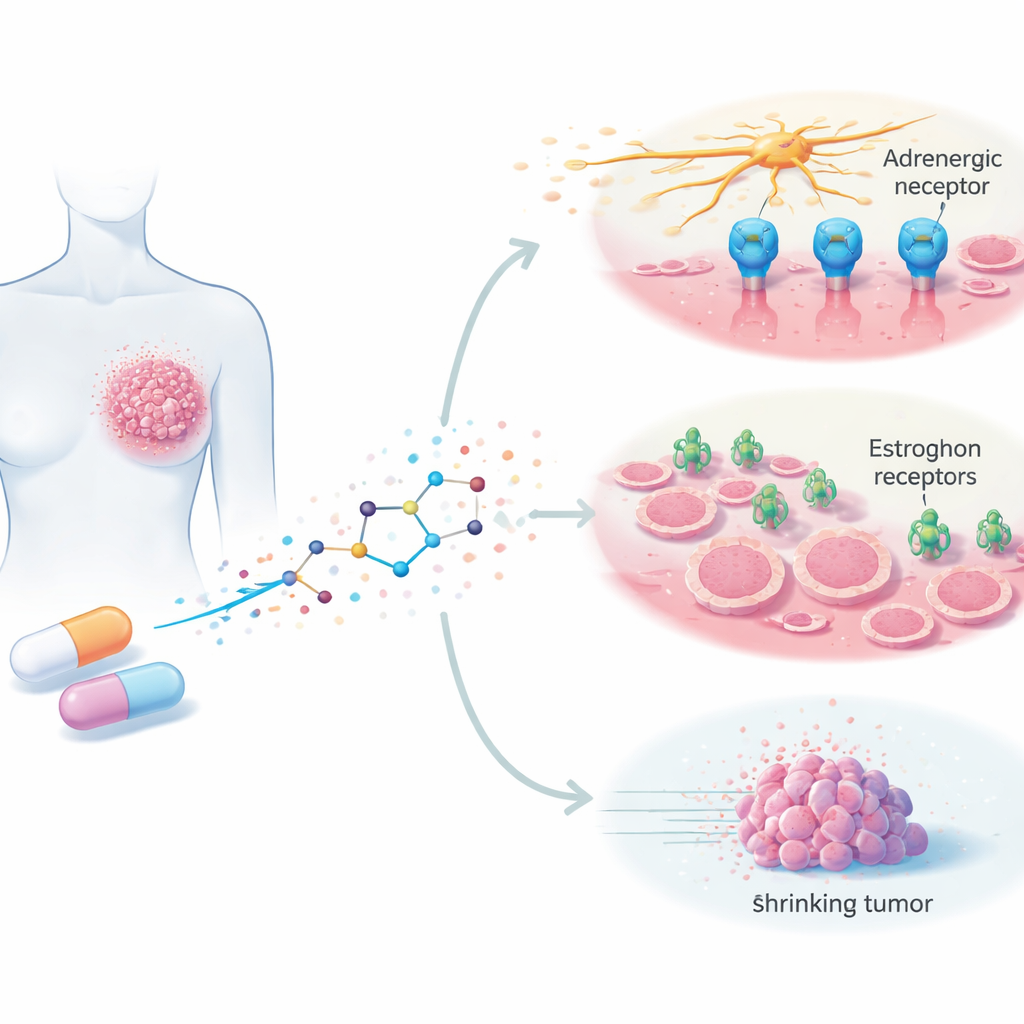
Silodosin is currently prescribed to ease urinary symptoms caused by an enlarged prostate by blocking certain nerve signals called adrenergic signals. These signals do more than control muscles; growing evidence suggests they can also help tumors grow, spread, and form new blood vessels. The authors wondered whether silodosin could interfere with these harmful signals in breast cancer cells as well. They tested the drug on several human breast cancer cell lines grown in the lab, representing both hormone-sensitive tumors and hard-to-treat triple-negative tumors, and compared its effects with those on healthy breast cells.
Slowing growth and triggering cell death
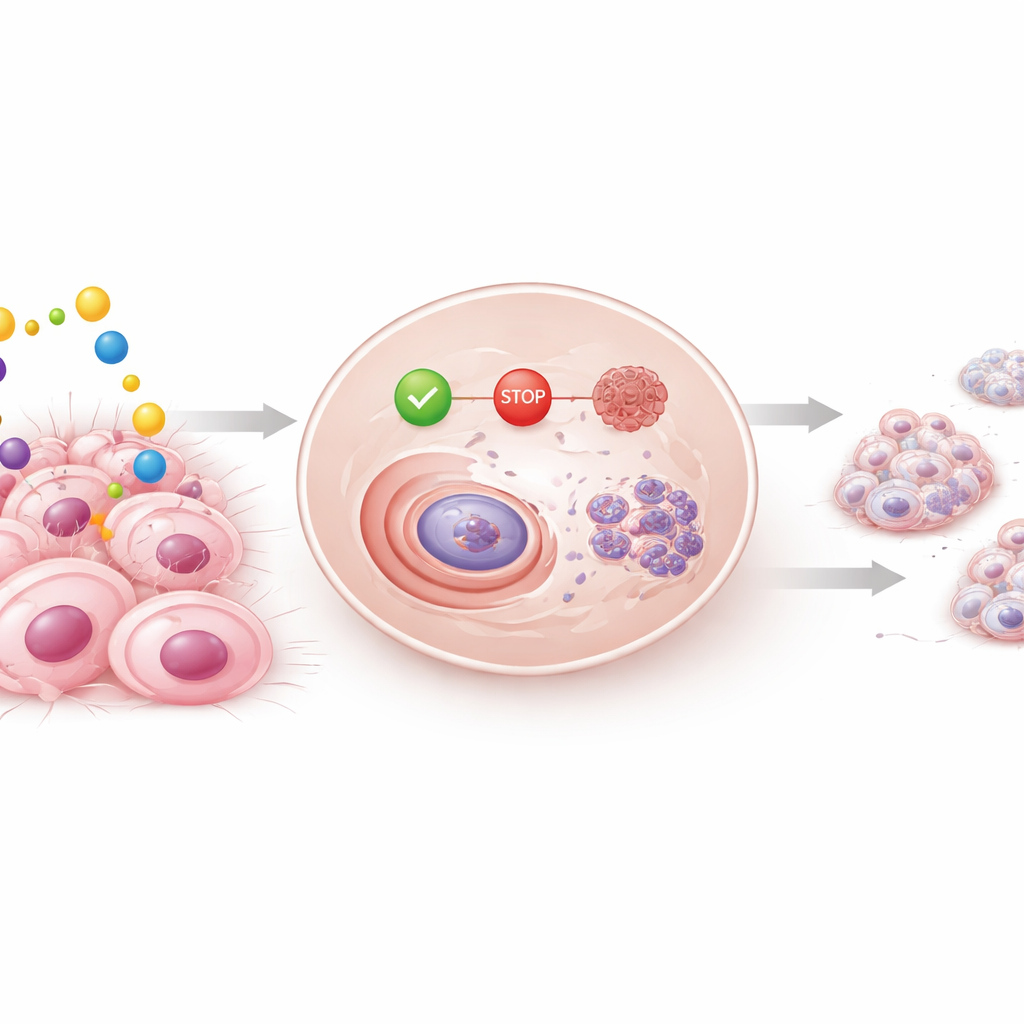
When breast cancer cells were exposed to silodosin, their growth slowed in a time- and dose-dependent way. Hormone-sensitive cells were especially affected, but even aggressive triple-negative cells showed reduced ability to survive and form colonies over time, while normal breast cells were largely spared. Further experiments revealed what was happening inside the cells: silodosin stalled them in an early phase of the cell cycle, the sequence of steps cells must complete to divide. Key "go" proteins that push cells toward division decreased, while "stop" proteins and the well-known guardian protein p53 increased. The drug also pushed cancer cells toward programmed cell death, as shown by growing numbers of cells with fragmented DNA.
Blocking movement and 3D tumor-like growth
Cancer becomes life-threatening when cells migrate and establish new tumors elsewhere. Using a scratch-like wound in a sheet of cells, the researchers found that silodosin slowed how quickly both hormone-sensitive and triple-negative breast cancer cells could move in to close the gap. The team also turned to three-dimensional spheroids, tiny ball-shaped cell clusters that better mimic real tumors than flat cell layers. Silodosin markedly reduced the formation and size of these spheroids. At the same time, it lowered levels of E-cadherin, a molecule that helps cells stick together and also participates in growth and survival signaling, suggesting that disrupting cell–cell contacts may be one way the drug interferes with tumor organization and spread.
A surprising second target inside cancer cells
Silodosin was designed to block adrenergic receptors, but the data did not fit a simple "on one target only" story. Some breast cancer cells that responded strongly to the drug had little or no detectable adrenergic receptor of the type silodosin is supposed to hit, and cells with more of this receptor were not always more sensitive. To search for other targets, the authors used computer modeling to see whether silodosin might also fit into estrogen receptors, the molecular switches that drive many breast cancers and are the main targets of drugs like tamoxifen. Their simulations showed that silodosin can latch onto both major forms of the estrogen receptor with meaningful strength, interacting with the same key contact points as tamoxifen. This suggests silodosin may act as an estrogen receptor modulator in addition to blocking nerve-related signals.
What this could mean for patients
Taken together, the experiments point to silodosin as a multi-purpose attacker of breast cancer cells: it slows their growth, pushes them toward self-destruction, hampers their movement, and prevents them from organizing into tumor-like spheroids, while likely acting on both adrenergic and estrogen-driven pathways. The effective doses in cell dishes are higher than those reached in patients at standard urology doses, so more work in animals and combination treatments will be needed to judge real-world potential. Still, because silodosin is already approved and generally well tolerated, this study lays the groundwork for repurposing it as part of new treatment strategies, particularly for patients with few options such as those with triple-negative breast cancer.
Citation: Pellegrino, M., Occhiuzzi, M.A., Marra, M. et al. Molecular insights into Silodosin’s anti-cancer effects: a promising repurposing strategy for breast cancer. Cell Death Discov. 12, 128 (2026). https://doi.org/10.1038/s41420-026-02973-8
Keywords: breast cancer, drug repurposing, silodosin, estrogen receptor, triple-negative breast cancer