Clear Sky Science · en
The protective up-regulation of metallothionein-2A in intervertebral disc degeneration inhibits nucleus pulposus cell ferroptosis through activation of the PI3K/AKT/mTOR pathway
Why this back problem matters
Low back pain affects hundreds of millions of people worldwide and is a leading cause of disability. One major culprit is the slow breakdown of the cushions between our spinal bones, called intervertebral discs. This study asks a hopeful question: can the body’s own protective molecules help shield these discs from damage, and could boosting them point to new treatments for chronic back pain? 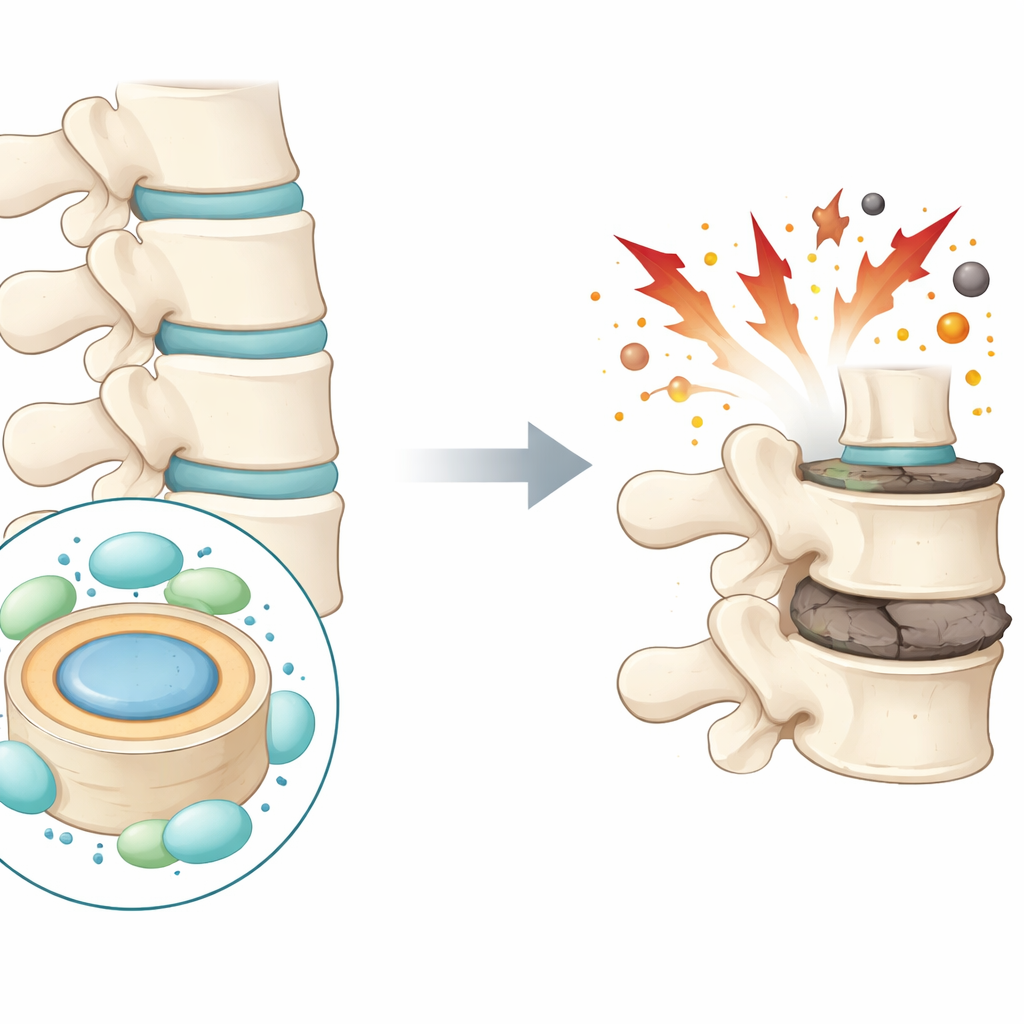
The cushions inside the spine
Each intervertebral disc has a soft, gel-like center called the nucleus pulposus, surrounded by a tougher ring. These structures act like shock absorbers for the spine. As discs degenerate, the cells in the central region die off and the supporting matrix of proteins and water wears away. The disc flattens and cracks, which can lead to pain and restricted movement. The authors focus on a specific type of disc cell death that has only recently attracted attention in many diseases: iron-driven, oxidation-related death known as ferroptosis. They suspect this process plays a key role in disc breakdown.
When iron and oxidation go wrong
Ferroptosis is triggered when free iron and reactive oxygen molecules build up inside cells, damaging fats in cell membranes and especially the small energy factories called mitochondria. The team analyzed single-cell genetic data from human disc tissue and found clear signs that ferroptosis-related genes are altered in degenerating discs. In particular, markers that normally protect cells from this kind of damage, such as the enzyme GPX4, were reduced in worn-out discs from both patients and rats. At the same time, markers that promote damage and inflammation were increased, painting a picture of cells under oxidative attack as discs degenerate.
A built-in metal‑binding protector
Among the many genes that changed in diseased discs, one stood out: metallothionein‑2A (MT2A), a small protein that binds metals like zinc and can mop up harmful reactive molecules. MT2A levels were strikingly higher in more severely degenerated human discs and in a rat model of disc injury. At first glance this seems paradoxical—why would a protective molecule rise in damaged tissue? The researchers hypothesized that the body is mounting a compensatory response, trying to counteract the surge in iron‑driven stress. In lab dishes, when disc cells were exposed to a chemical that mimics oxidative stress, MT2A levels climbed as cell health declined, reinforcing the idea that it is switched on as a defense.
Turning protection down and up
To test whether MT2A is truly protective rather than harmful, the team carefully dialed its levels down or up in human disc cells. When they blocked MT2A production, oxidative stress caused much more cell death, more iron buildup, stronger lipid damage, and severely injured mitochondria. When they boosted MT2A instead, or treated cells with a known ferroptosis blocker, many of these problems were eased: iron and reactive molecules dropped, protective antioxidants recovered, and mitochondria looked healthier. These changes were also reflected in better preservation of the cells’ supporting matrix proteins, which are crucial for keeping discs plump and functional. 
A key signaling route inside the cell
Diving deeper, the scientists looked at how MT2A sends protective signals inside cells. Gene sequencing and protein measurements pointed to the PI3K/AKT/mTOR pathway, a well-known internal control system that influences growth, metabolism, and survival. Lowering MT2A dampened the activity of this pathway, while raising MT2A switched it on. When the researchers used drugs to block different steps in this pathway, the benefits of MT2A overexpression vanished: ferroptosis markers rose again, oxidative damage returned, and disc cells lost more of their structural proteins. This suggests that MT2A shields disc cells largely by activating this pro-survival signaling route, which in turn restrains ferroptosis.
Proof of concept in animals
Finally, the team asked whether boosting MT2A could actually slow disc degeneration in a living organism. In rats, they created disc injury using a needle puncture and then delivered a virus designed to increase MT2A right inside the disc. Weeks later, imaging and tissue analysis showed that discs with extra MT2A kept more of their height, had clearer internal structure, and showed less loss of key matrix proteins compared to untreated injured discs. Markers of ferroptosis were also reduced, supporting the idea that MT2A helps protect the disc cushion from collapsing under oxidative and iron stress.
What this means for future back pain care
Taken together, the work suggests that MT2A is not just a bystander but a built-in safety factor that ramps up as discs begin to fail, trying to defend cells against damaging iron-driven oxidation. By activating an internal survival pathway, MT2A limits a specific form of cell death, preserves the disc’s structure, and slows degeneration—at least in animal models and cell cultures. For patients, this points to MT2A and its signaling partners as promising targets for drugs or gene-based therapies aimed at protecting or rejuvenating spinal discs, potentially offering more than just pain relief and surgery in the long run.
Citation: Cai, H., Zheng, Hl., Chen, Qz. et al. The protective up-regulation of metallothionein-2A in intervertebral disc degeneration inhibits nucleus pulposus cell ferroptosis through activation of the PI3K/AKT/mTOR pathway. Cell Death Discov. 12, 111 (2026). https://doi.org/10.1038/s41420-026-02972-9
Keywords: low back pain, intervertebral disc degeneration, ferroptosis, metallothionein-2A, PI3K AKT mTOR pathway