Clear Sky Science · en
Multi-omics reveals heterogeneity and functional populations of oligodendrocyte progenitor cells induced by human neural stem cells
Why protecting brain insulation matters
Our brain’s wiring depends on a fatty coating called myelin, which wraps around nerve fibers like insulation on electrical cables. When myelin is damaged, as in multiple sclerosis and other disorders, signals slow down or fail, causing problems with movement, vision, and thinking. This study explores a special group of human cells that can rebuild myelin and asks a practical question: which cells are best suited for future cell therapies to repair this crucial insulation?
From starter brain cells to myelin makers
The researchers began with human neural stem cells—versatile starter cells taken from fetal brain tissue that are already committed to becoming cells of the nervous system. They coaxed these stem cells in the lab to turn into oligodendrocyte progenitor cells, or hOPCs, which are the direct precursors of the myelin-forming cells. Under the microscope, the cells changed from simple round shapes into more complex forms with multiple branches, and they switched on hallmark proteins that mark steps along the path toward mature myelin-producing cells. This provided a reliable, relatively safe source of human myelin-building precursors for detailed analysis.
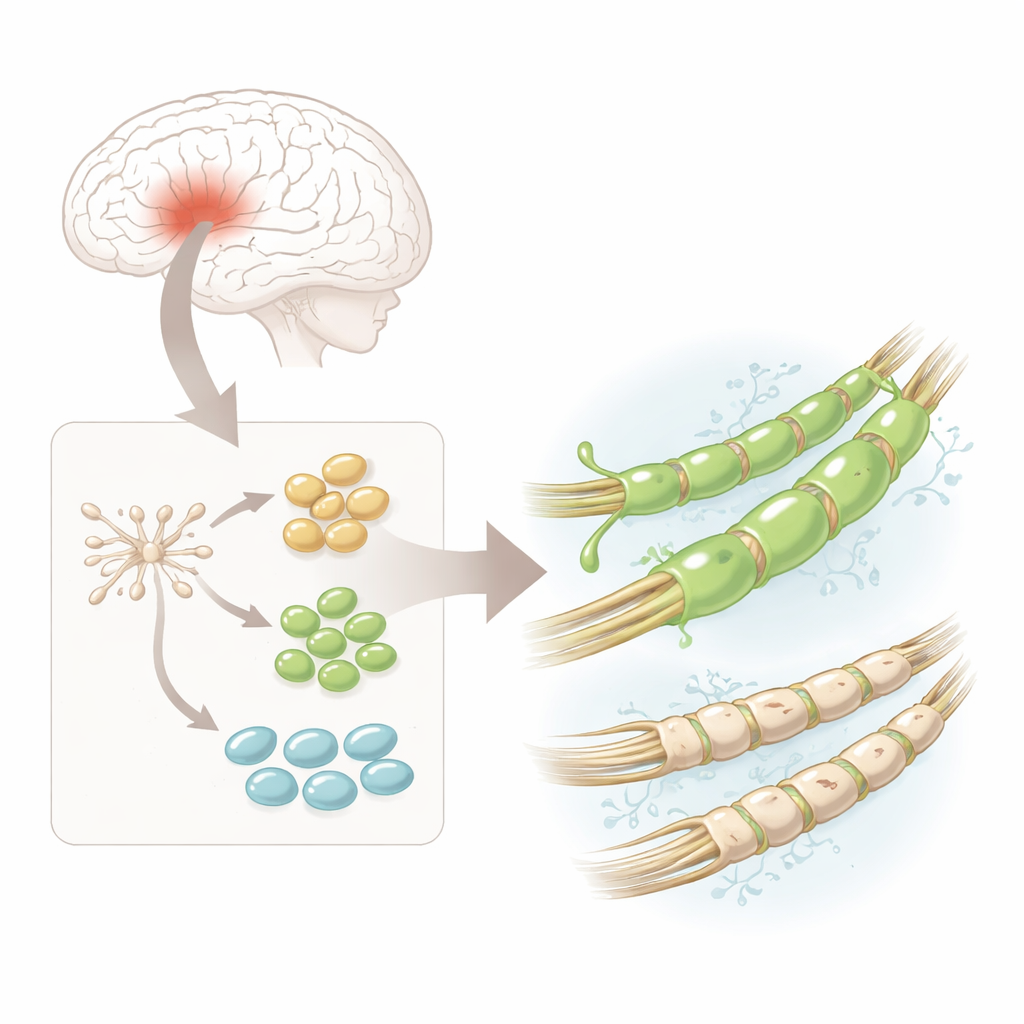
One cell type, many hidden varieties
Using single-cell RNA sequencing—an approach that reads out which genes are active in thousands of individual cells—the team discovered that the hOPCs were not all the same. Instead, they grouped into three main stages: early “pre-progenitors,” more committed progenitors, and cells approaching full oligodendrocyte identity. Across all of these stages, one gene stood out: PDGFRA, which encodes a cell-surface receptor. A highly sensitive imaging method called RNA-Scope confirmed that PDGFRA’s message and protein were more abundant than other key markers at every stage, suggesting that this receptor might define particularly capable myelin-building cells.
Sorting out the most powerful repair cells
To test this idea, the scientists separated hOPCs into two groups based on whether they carried the PDGFR-α receptor on their surface. They then compared PDGFR-α–positive cells, PDGFR-α–negative cells, and unsorted cells in a series of functional tests. When transplanted into “shiverer” mice—which cannot form normal myelin—the PDGFR-α–positive cells produced denser, more compact myelin around nerve fibers than the other groups. These cells also migrated farther and multiplied more rapidly in laboratory assays. In other words, the PDGFR-α–positive population was the most vigorous at moving to where it was needed, expanding in number, and rebuilding insulation.
Inside signals that drive growth and repair
Digging deeper, the team compared gene activity between PDGFR-α–positive and –negative cells. They found that the positive cells switched on networks of genes involved in glial cell growth and myelin formation, as well as several key signaling routes inside the cell. Two pathways in particular stood out: PI3K–AKT–mTOR, long linked to cell growth and myelin production, and TGF-β signaling, which influences how young brain cells choose their fates. The data suggested that activation of PDGFR-α feeds into PI3K–AKT–mTOR, which in turn boosts TGF-β–related signals, together nudging cells toward becoming effective myelin makers.
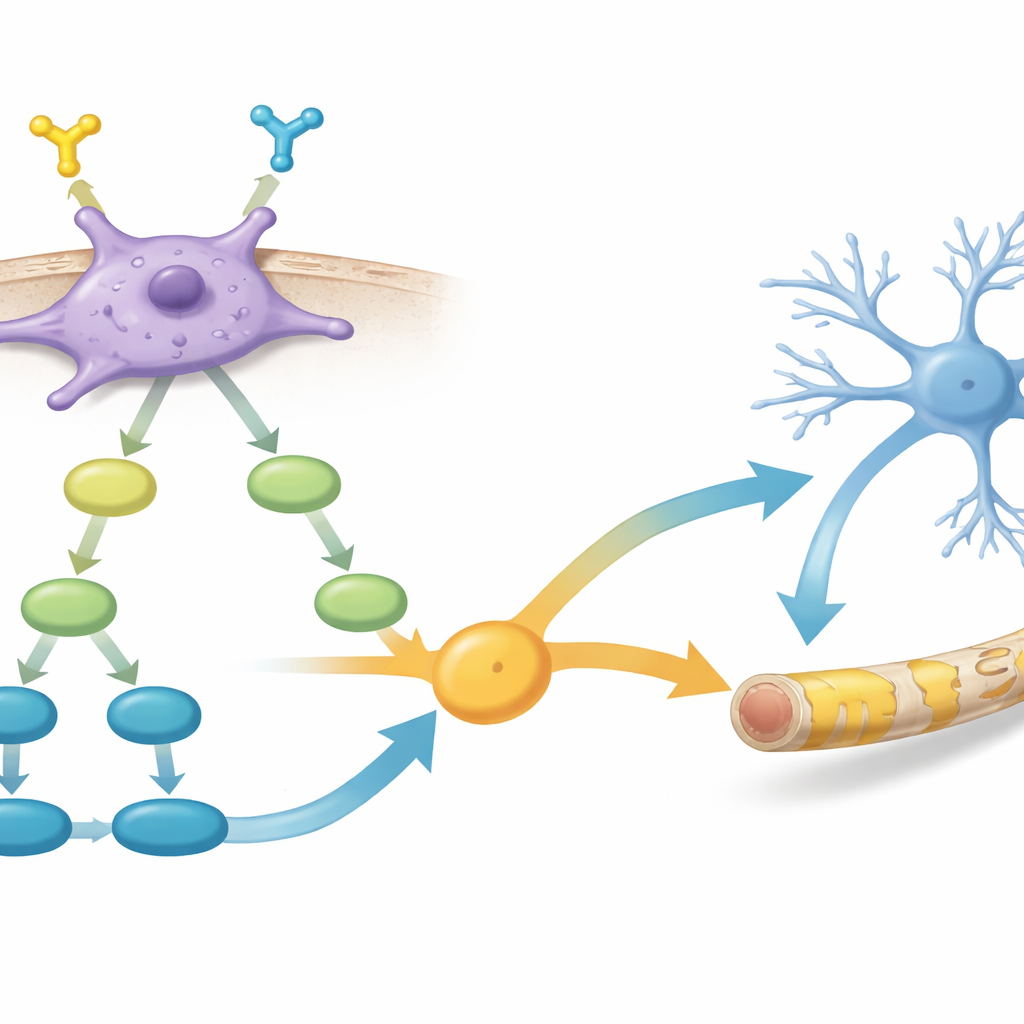
Boosting myelin repair with a chemical helper
The researchers then asked whether they could further enhance these promising cells. They treated PDGFR-α–positive hOPCs with a small molecule that activates the TGF-β pathway and examined both gene activity and behavior. After treatment, these cells showed higher levels of multiple myelin-related genes and produced thicker, more complete myelin sheaths when transplanted into shiverer mice, compared with untreated PDGFR-α–positive cells. This supports the idea that carefully tuning internal signaling in already well-chosen progenitor cells can further improve their performance as living myelin repair tools.
What this means for future treatments
For non-specialists, the core message is that not all myelin-building precursor cells are equal. By combining powerful gene-reading and imaging tools, this study identifies a standout subgroup—PDGFR-α–positive hOPCs—that migrate better, divide more, and rebuild myelin more effectively than their peers. It also highlights internal signal routes that can be chemically nudged to make these cells even more potent. Together, these insights lay a roadmap for designing safer, more efficient cell therapies to restore the brain’s insulation in diseases where myelin is lost.
Citation: Ye, D., Zhou, H., Qu, S. et al. Multi-omics reveals heterogeneity and functional populations of oligodendrocyte progenitor cells induced by human neural stem cells. Cell Death Discov. 12, 112 (2026). https://doi.org/10.1038/s41420-026-02971-w
Keywords: myelin repair, oligodendrocyte progenitor cells, neural stem cells, cell therapy, single-cell RNA sequencing