Clear Sky Science · en
SREBP1 knockdown triggers ferroptosis by suppressing the Nrf2-XCT/GPX4 axis in ovarian cancer
Turning Cancer’s Fat Factory Against Itself
Ovarian cancer is often discovered late and can be very hard to treat. This study explores a surprising weakness in these tumors: their addiction to making fats and oils. The researchers show that shutting down a key "fat factory" switch in cancer cells can both slow their growth and flip on a special kind of cell death, while also making it easier for the immune system to attack the tumor.
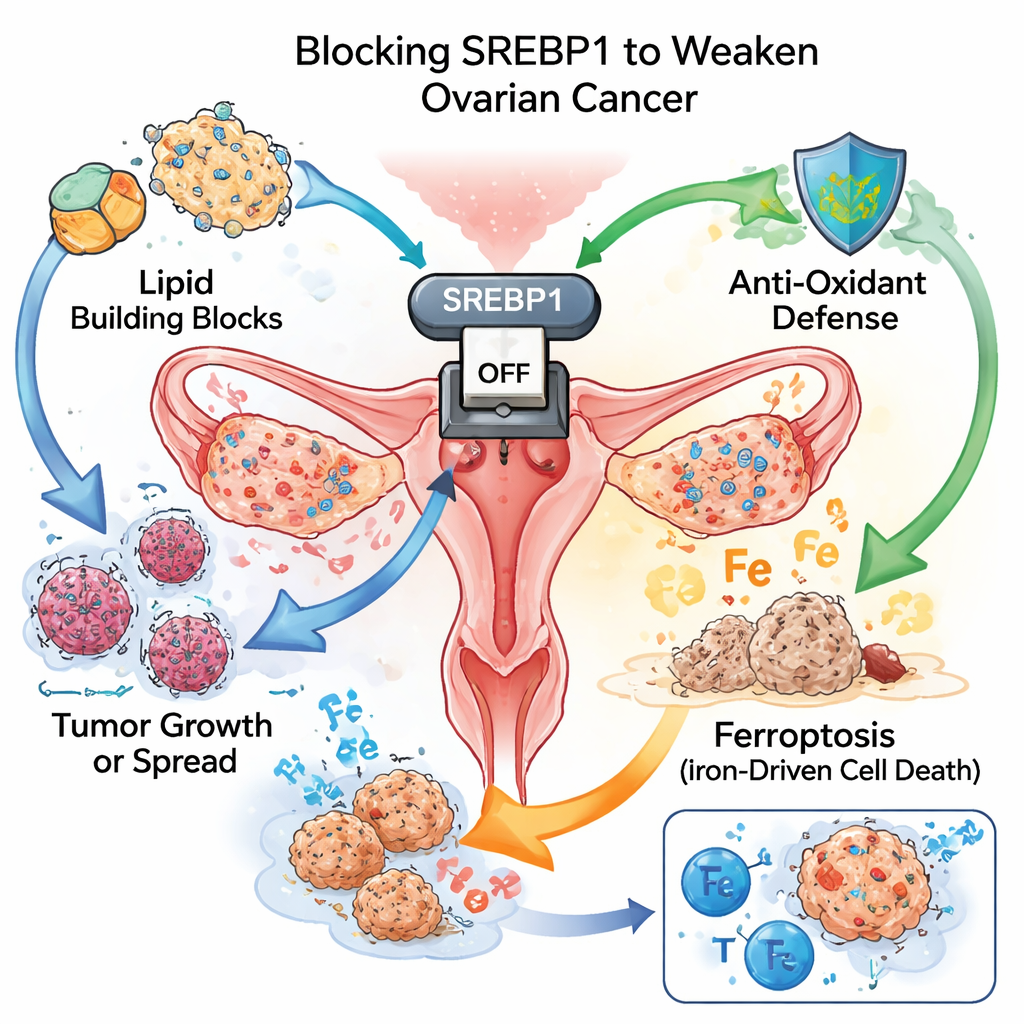
A Master Switch for Tumor Fuel
Like fast‑growing weeds, cancer cells need huge amounts of building blocks and energy. One way they meet this demand is by rewiring how they make and use fats. The team focused on SREBP1, a protein that acts as a master switch for fat production. In tissue samples from more than a hundred patients, ovarian tumors had much higher SREBP1 levels than nearby normal tissue. Women whose tumors showed stronger SREBP1 staining tended to have more aggressive disease and shorter survival, marking this protein as both a driver of progression and a warning sign for poor outcome.
Slowing Growth and Spread by Cutting Off Fat
In ovarian cancer cell lines grown in the lab, turning down SREBP1 sharply reduced the cells’ ability to multiply and form colonies. The cells became stuck at specific stages of the cell cycle, meaning they could no longer divide efficiently. The researchers also saw fewer signs of movement and invasion: scratch and Transwell tests showed that cells with less SREBP1 were less able to migrate, and key markers of a shape‑shifting process called EMT shifted back toward a more “normal” state. At the same time, measurements of triglycerides and cholesterol dropped, and staining showed fewer lipid droplets inside the cells, confirming that their internal fat‑making machinery had been dialed down.
Triggering an Iron‑Fueled Cell Demise
The most striking effect of blocking SREBP1 was the activation of ferroptosis, a recently recognized form of cell death driven by iron and the rusting, or peroxidation, of cell membranes. Only a ferroptosis blocker, and not inhibitors of other death pathways, could rescue SREBP1‑silenced cells, pointing to ferroptosis as the main cause of their demise. Chemical tests showed more damage products from oxidized fats, less antioxidant glutathione, and higher levels of reactive lipid molecules. Two proteins that normally shield cells from ferroptosis, called xCT and GPX4, were strongly reduced when SREBP1 was knocked down, removing a crucial protective buffer against this destructive process.
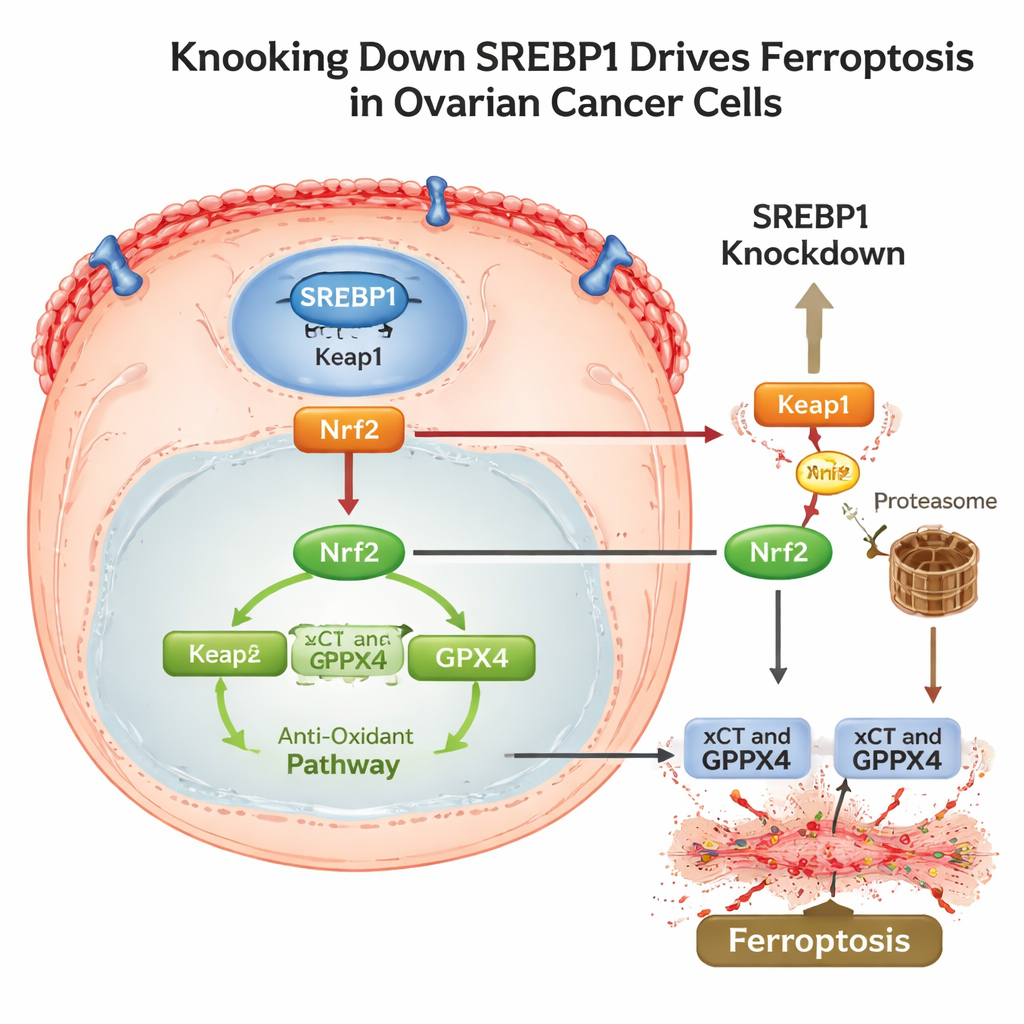
Disarming the Cell’s Antioxidant Shield and Evasion Tricks
To understand how SREBP1 connects to this death program, the scientists traced a pathway through another major stress‑response protein, Nrf2. Under normal conditions, Nrf2 helps cells survive by switching on antioxidant genes, including xCT and GPX4. The study found that lowering SREBP1 increased levels of Keap1, a protein that tags Nrf2 for disposal, leading to more Nrf2 breakdown and less of it reaching the nucleus. As Nrf2 fell, its downstream defenses weakened and ferroptosis followed. Importantly, SREBP1‑rich tumors also showed higher levels of PD‑L1, a surface protein that helps cancer cells hide from attacking T cells. When SREBP1 was silenced in mouse tumors, the cancers grew more slowly, showed more signs of lipid damage, less Nrf2 and GPX4, and lower PD‑L1, alongside increased immune activity.
Why This Matters for Future Treatments
In plain terms, ovarian tumors seem to use SREBP1 as a double‑edged survival tool: it feeds their growth by boosting fat production and at the same time arms them with an antioxidant shield and immune disguise. This work shows that turning off SREBP1 can starve the tumor of lipids, strip away its protection against iron‑driven damage, and reduce its ability to dodge immune attack. That makes SREBP1 an attractive target for new drugs and suggests that combining SREBP1 blockers with treatments that trigger ferroptosis or unleash the immune system could offer patients more effective, longer‑lasting control of their disease.
Citation: Nie, R., Zhou, H., Chen, L. et al. SREBP1 knockdown triggers ferroptosis by suppressing the Nrf2-XCT/GPX4 axis in ovarian cancer. Cell Death Discov. 12, 101 (2026). https://doi.org/10.1038/s41420-026-02964-9
Keywords: ovarian cancer, lipid metabolism, ferroptosis, SREBP1, tumor immunity