Clear Sky Science · en
PDHA1 enhances resistance to ferroptosis in anoikis-resistant prostate cancer by upregulating AIFM2
Why some prostate cancer cells refuse to die
When cancer cells break away from a tumor and enter the bloodstream, most should die before they ever reach a new organ. Yet a dangerous minority survive, travel, and seed metastases that are often lethal. This study asks a key question: what lets certain prostate cancer cells resist a form of cell death that normally strikes once they lose contact with their home tissue? By uncovering a hidden survival circuit inside these cells, the research points to new ways to block metastasis at its source.
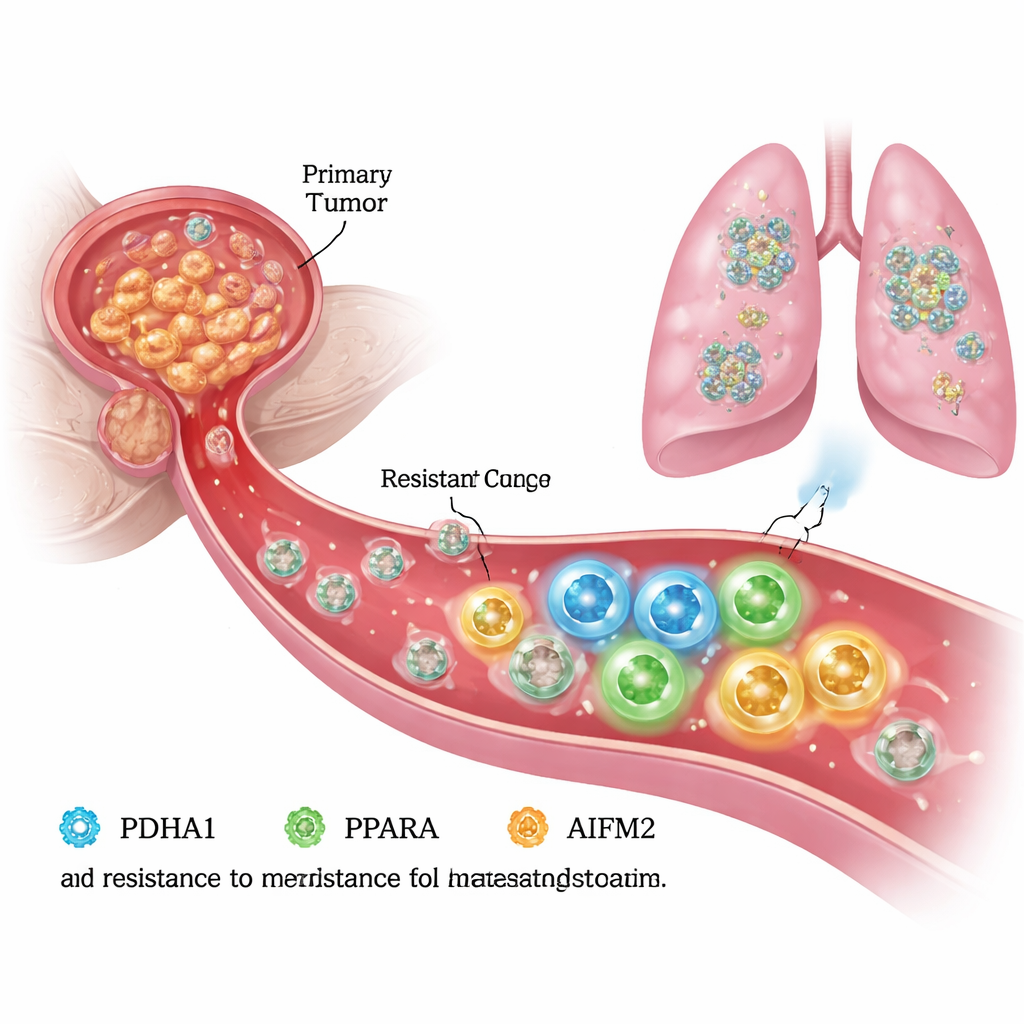
Escaping death in the bloodstream
To spread, prostate cancer cells must endure life without the support of the surrounding tissue, known as the extracellular matrix. Under these “detached” conditions, healthy cells undergo a programmed death called anoikis, and many cancer cells are also vulnerable to a related iron-driven death process called ferroptosis. The authors created anoikis-resistant prostate cancer cells in the lab and compared them with their original “parental” cells. They found that the resistant cells not only migrated and invaded more aggressively, but also survived better under detachment, suggesting that they had rewired their internal machinery to avoid ferroptosis and keep growing.
A metabolic switch with a second job
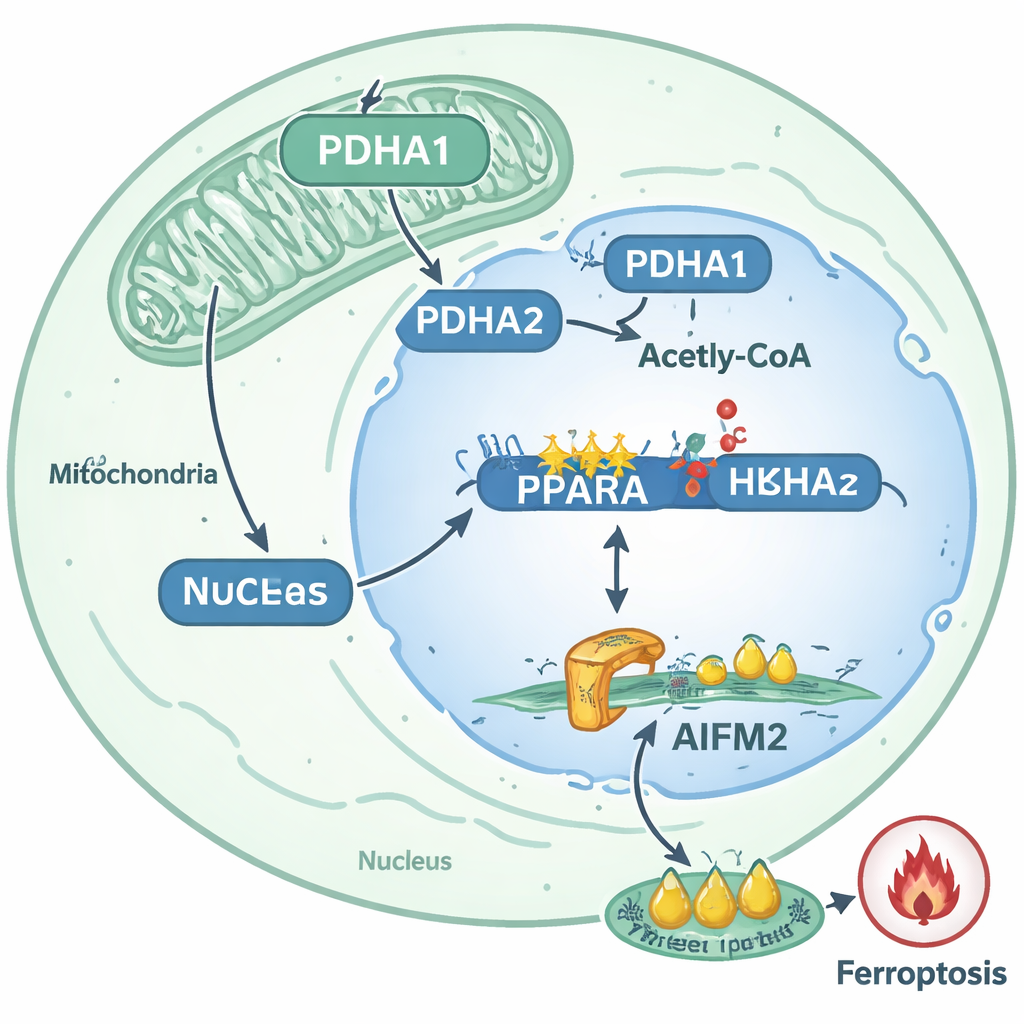
Digging deeper, the team focused on a metabolic enzyme called PDHA1, best known for helping cells turn sugar-derived pyruvate into acetyl-CoA in mitochondria, fueling energy production. In the anoikis-resistant cells, PDHA1 levels and activity were markedly higher, and patient samples showed more PDHA1 in primary tumors from men whose prostate cancer had spread to lymph nodes or distant sites. When the researchers reduced PDHA1, resistant cells lost much of their ability to move, invade, and survive. Surprisingly, a substantial portion of PDHA1 in these cells was no longer confined to mitochondria; it had relocated into the nucleus, where DNA is packaged with histones that can be chemically modified to turn genes on or off.
Rewriting gene activity to block ferroptosis
Inside the nucleus, PDHA1 appeared to fuel the local production of acetyl-CoA, a key building block for histone acetylation. The authors showed that nuclear PDHA1 boosted a specific mark, H3K9 acetylation, at the control region of a gene called PPARA. This change acted like loosening the chromatin “spools,” making it easier to switch PPARA on. PPARA, in turn, served as a master switch that increased the production of another protein, AIFM2, by binding to its promoter and raising its transcription. Rather than directly adjusting the AIFM2 gene, PDHA1 thus worked through PPARA, establishing a multi-step pathway from altered metabolism to altered gene regulation.
Building a shield against iron-driven damage
AIFM2 is known as an internal brake on ferroptosis: it helps protect fats in cell membranes from destructive oxidation. In the resistant prostate cancer cells, PDHA1 upregulation raised AIFM2 levels, while PDHA1 knockdown lowered them. Functional tests showed that reducing PDHA1 intensified the hallmarks of ferroptosis—more reactive oxygen species, more lipid damage, lower mitochondrial membrane potential, and characteristic mitochondrial shrinkage—especially when cells were exposed to the ferroptosis-inducing drug erastin. Overexpressing AIFM2 reversed these effects, rescuing cell survival in dishes and restoring lung metastasis in mice, even when PDHA1 was suppressed. This confirmed that AIFM2 is a critical downstream effector of PDHA1-driven ferroptosis resistance.
From molecular circuit to treatment ideas
By mapping these events, the study outlines a clear route by which detached prostate cancer cells become harder to kill: PDHA1 moves into the nucleus, increases histone acetylation at the PPARA gene, boosts PPARA activity, and thereby raises AIFM2 levels to block ferroptosis. This pathway is strongly linked to metastatic behavior in patient tumors and in animal models. For non-specialists, the takeaway is that certain cancer cells repurpose a standard metabolic enzyme into an epigenetic tool that rewires gene activity and builds a biochemical shield against a powerful form of cell death. The authors suggest that drugs targeting PDHA1, PPARA, or AIFM2—or therapies that deliberately trigger ferroptosis—could one day be combined to strip metastatic prostate cancer cells of this survival advantage and make them far more vulnerable during their most vulnerable journey: the leap from one organ to another.
Citation: Cong, Y., Chen, K., Ju, Y. et al. PDHA1 enhances resistance to ferroptosis in anoikis-resistant prostate cancer by upregulating AIFM2. Cell Death Discov. 12, 105 (2026). https://doi.org/10.1038/s41420-026-02958-7
Keywords: prostate cancer metastasis, ferroptosis, anoikis resistance, PDHA1, AIFM2