Clear Sky Science · en
Osteocytic Lipocalin-2 regulates bone formation locally through iron-dependent ferroptosis and Wnt suppression
When Bone Cells Quietly Shape Whole-Body Health
Most of us think of bones as rigid scaffolding, but they are alive with cells that constantly rebuild and repair our skeleton. This study reveals how one little protein made by bone cells, called lipocalin-2, can quietly weaken bones from within by disturbing how cells handle iron and energy. Understanding this hidden process could point the way to new treatments for osteoporosis and age-related bone loss.
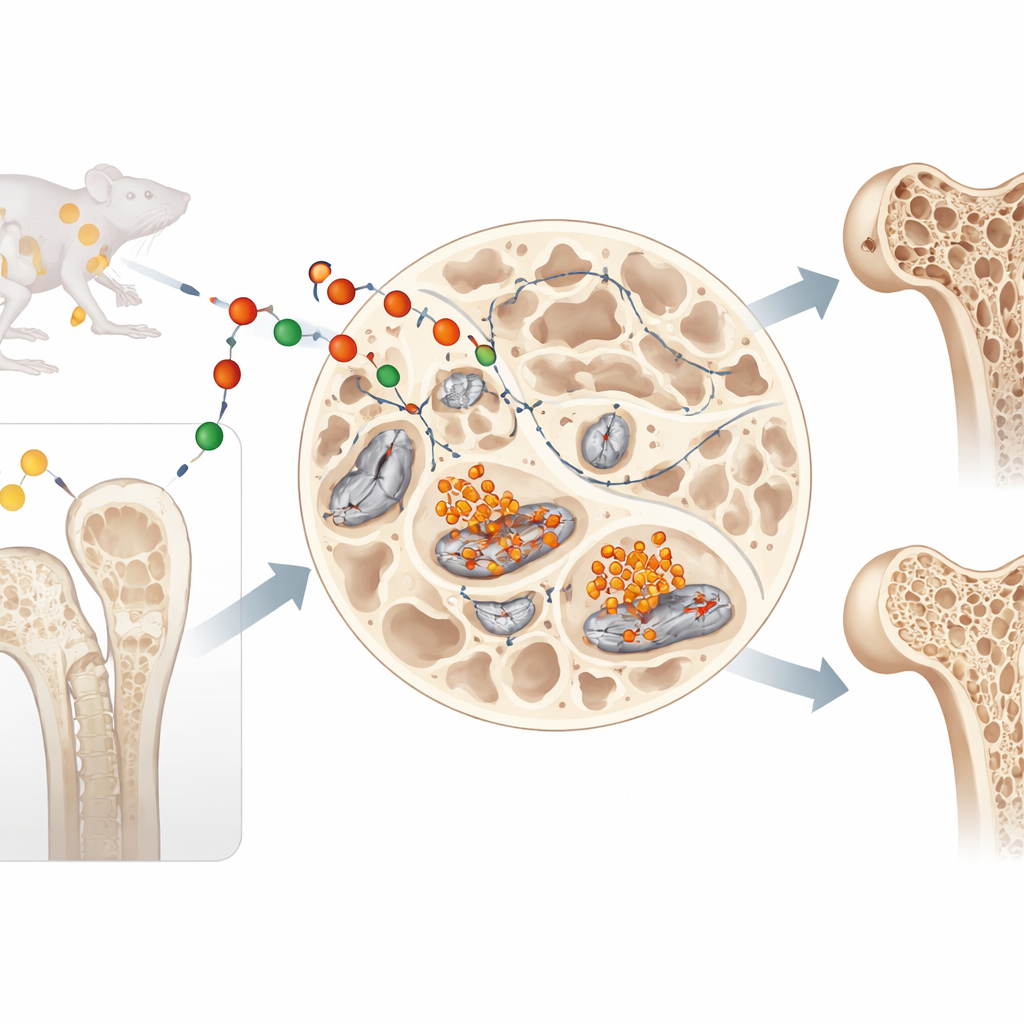
A Messenger Protein with a Double Life
Lipocalin-2 has been known as a roaming hormone that travels through the blood, helping control appetite and body weight and defending against infection by binding iron. But the authors of this paper asked a different question: what does lipocalin-2 do right where it is made, inside bone itself? They focused on osteocytes, the long-lived cells buried deep in bone that sense mechanical forces and direct bone remodeling. The team found that osteocytes not only keep making lipocalin-2 as they mature, they actually increase its production, suggesting a strong local role inside the skeleton.
Iron Overload and a Rust-Like Cell Death
Because lipocalin-2 can ferry iron, the researchers tested whether it drives iron buildup in osteocytes. In cultured bone cells, adding lipocalin-2 caused iron to accumulate inside the cells, boosted harmful reactive oxygen molecules, and triggered heavy damage to cell membranes rich in fats. These are hallmarks of a recently recognized form of cell death called ferroptosis, in which iron acts a bit like rust, sparking destructive chemical reactions. Blocking iron with a chelating compound completely prevented the damage and cell death, confirming that the process was iron-dependent.
The Gatekeeper Receptor and Mitochondrial Fallout
To understand how lipocalin-2 gets its iron cargo into osteocytes, the team turned to a specific cell-surface receptor, SLC22A17. When they reduced this receptor in osteocyte-like cells, lipocalin-2 no longer drove iron entry, oxidative stress, or ferroptotic death, even under conditions of iron overload. The study also showed that lipocalin-2 undermines the cell’s powerhouses: mitochondria. Extra lipocalin-2 caused mitochondrial membranes to lose their normal voltage and reduced energy production, while removing lipocalin-2 restored mitochondrial function. In mouse bones lacking lipocalin-2 in osteocytes, genes that support mitochondrial respiration and quality control were switched on, hinting that these cells were more energetically robust.
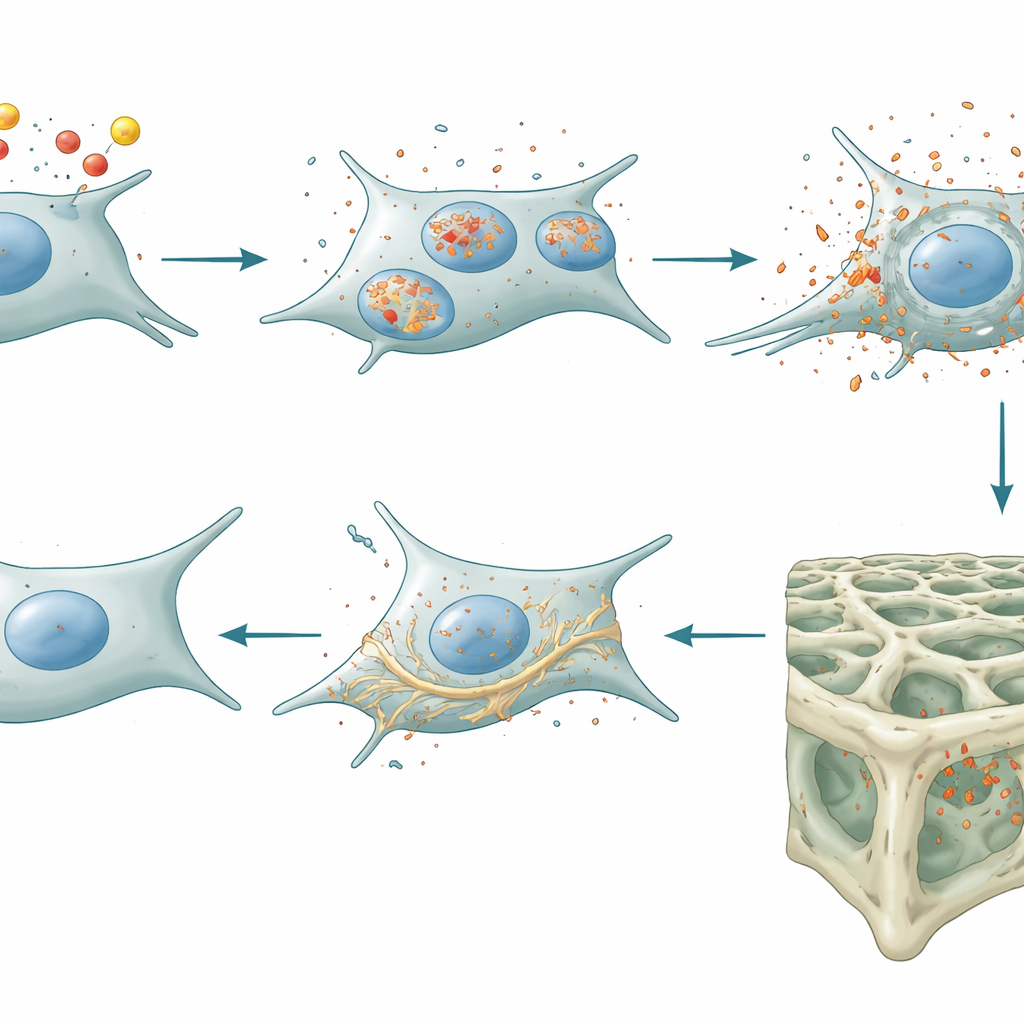
From Cell Stress to Weaker Bone, and How Removing the Brake Helps
The researchers next looked at intact mice engineered to delete lipocalin-2 specifically in late-stage bone-forming cells and osteocytes. These animals accumulated less iron inside bone cells and showed stronger antioxidant defenses that counteract ferroptosis. Importantly, they also produced lower levels of two key proteins—sclerostin and DKK1—that normally act as brakes on the Wnt/β-catenin pathway, a major driver of bone formation. With those brakes eased, signaling that encourages bone-building osteoblasts increased. Micro–CT scans and bone labeling experiments confirmed the outcome: mice without osteocytic lipocalin-2 built more and better-connected spongy (trabecular) bone, mainly by ramping up bone formation rather than slowing bone breakdown. Despite these skeletal changes, their body weight, blood sugar control, metabolism, and activity levels remained normal, showing that the effect was local to bone rather than a whole-body hormone effect.
What This Means for Keeping Bones Strong
Put simply, this work shows that lipocalin-2 inside bone cells acts like a local switch that raises iron levels, strains mitochondria, primes cells for ferroptotic damage, and dampens a key growth pathway, together limiting new bone formation. When this internal brake is removed, osteocytes stay healthier, send more pro-building signals, and trabecular bone becomes denser and better connected without disturbing overall metabolism. Because lipocalin-2 levels and iron imbalance both rise with aging and chronic inflammation, targeting the lipocalin-2–receptor system could offer a new way to protect bones from “rusting” from the inside out.
Citation: Khanal, V., Carroll, M., Moradi, F. et al. Osteocytic Lipocalin-2 regulates bone formation locally through iron-dependent ferroptosis and Wnt suppression. Cell Death Discov. 12, 113 (2026). https://doi.org/10.1038/s41420-026-02956-9
Keywords: bone health, osteocytes, iron metabolism, ferroptosis, Wnt signaling