Clear Sky Science · en
Mitophagy in pancreatic cancer: mechanistic insights and implications for novel therapeutic strategies
Why our cells’ power plants matter in pancreatic cancer
Pancreatic ductal adenocarcinoma is one of the deadliest cancers, in part because tumors quickly learn to resist nearly every treatment doctors throw at them. This review article explores a surprising player in that resistance story: mitophagy, the cell’s built‑in system for identifying and recycling worn‑out mitochondria, the tiny “power plants” that make energy. Understanding how pancreatic cancer hijacks this cleanup process could open new paths to more effective, longer‑lasting therapies.
Cellular cleanup crews and cancer’s survival tricks
Mitochondria do far more than generate energy; they help control metabolism, cell death, and the handling of harmful molecules called reactive oxygen species. When mitochondria are damaged or too abundant, cells use mitophagy to tag and remove them. This can happen through two main routes. One, known as the PINK1/Parkin pathway, relies on a damage sensor (PINK1) and a tagging enzyme (Parkin) to mark faulty mitochondria for destruction. The other uses receptor proteins such as BNIP3, NIX, and FUNDC1 sitting on mitochondrial surfaces to directly connect them to the cell’s waste‑disposal sacs, called autophagosomes, without the same tagging step. These pathways often work together, giving cells flexible ways to adapt their mitochondrial stock to stress like low oxygen or exposure to drugs. 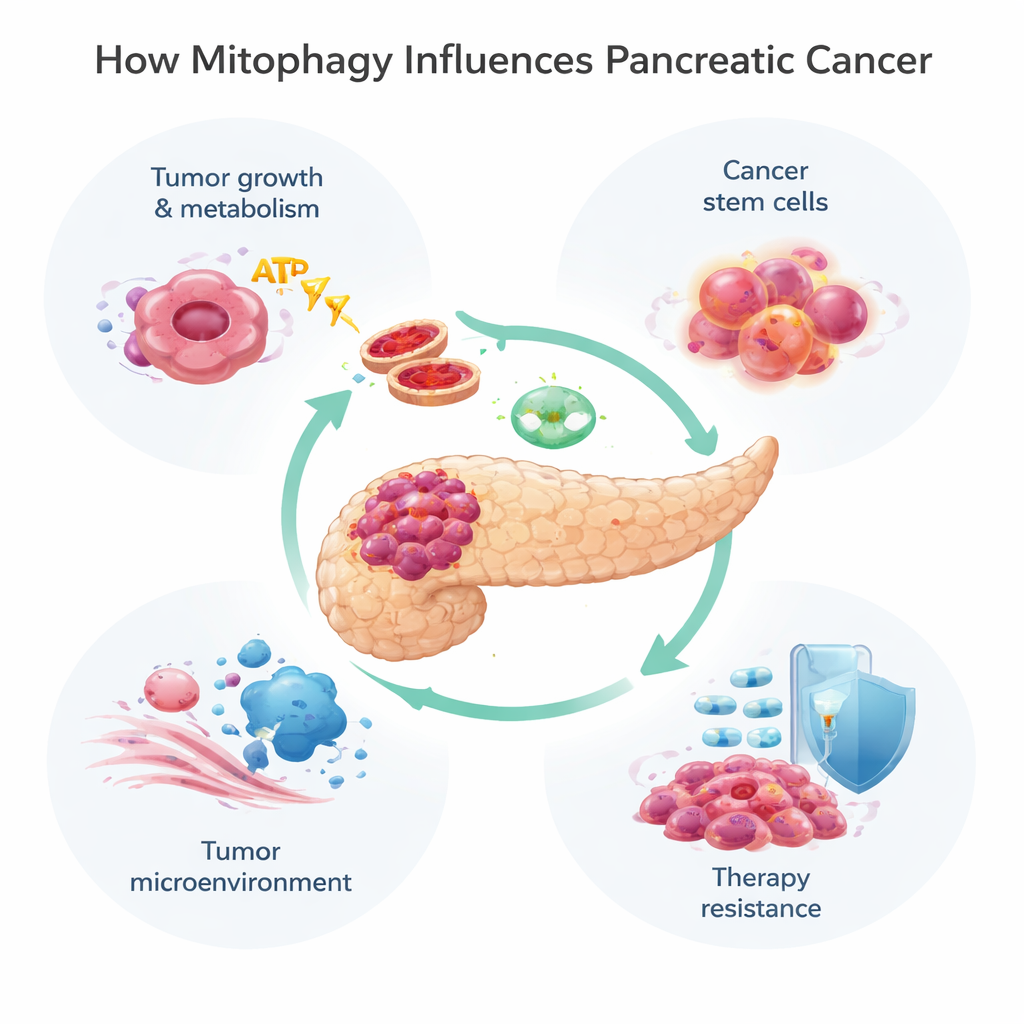
How pancreatic tumors reshape their energy use
Pancreatic tumors live in a harsh neighborhood: they are poorly supplied with blood and oxygen and surrounded by dense scar‑like tissue. To survive, cancer cells rewire their energy systems, often shifting from oxygen‑based respiration in mitochondria to a more primitive sugar‑burning process known as glycolysis. Mitophagy helps fine‑tune this shift. In early tumor development, loss of PINK1 or Parkin speeds up cancer formation by allowing iron buildup, excess reactive oxygen species, and a push toward glycolysis. At the same time, another mitophagy protein, NIX, can help remove still‑functional mitochondria, nudging cells further toward glycolytic metabolism and faster growth. By contrast, BNIP3 shows a more complex pattern: it appears early, is later silenced in many advanced tumors, and when restored can slow cancer cell growth, suggesting some mitophagy routes restrain cancer while others fuel it.
Cancer stem cells and the tumor’s neighborhood
Not all cancer cells are equal. A small subset, often called cancer stem cells, can self‑renew, seed new tumors, and weather chemotherapy particularly well. In pancreatic cancer, these cells depend on mitophagy to stay metabolically flexible. A modifier protein called ISG15 supports this process and helps maintain their stem‑like traits, linking mitophagy directly to relapse and treatment failure. Beyond the cancer cells themselves, mitophagy also shapes the tumor microenvironment—the mix of support cells, immune cells, and connective tissue around the tumor. Cancer‑associated fibroblasts, for instance, can be driven into a “reverse Warburg effect,” where they ramp up their own mitophagy and glycolysis, then feed the tumor with energy‑rich by‑products. Mitophagy also influences how immune cells recognize and attack cancer, by altering the display of immune flags such as MHC‑I and the brakes like PD‑L1 that tumors use to hide.
Why blocking or boosting cleanup can change drug response
Many standard and experimental treatments for pancreatic cancer, including the chemotherapy drug gemcitabine and a new targeted drug against the common KRAS‑G12D mutation, injure mitochondria. Tumor cells often respond by turning on mitophagy, clearing the damage and escaping death. Studies show that PINK1‑driven mitophagy can blunt the killing effects of chemotherapy and some natural compounds, helping cancer cells survive. On the other hand, under certain conditions, pushing mitophagy too far—especially through BNIP3 and NIX—may strip cells of too many functioning mitochondria, tipping them into an energy crisis and promoting cell death. This dual nature means that mitophagy can either shield tumors from treatment or, if pushed in the right way, make them more vulnerable. 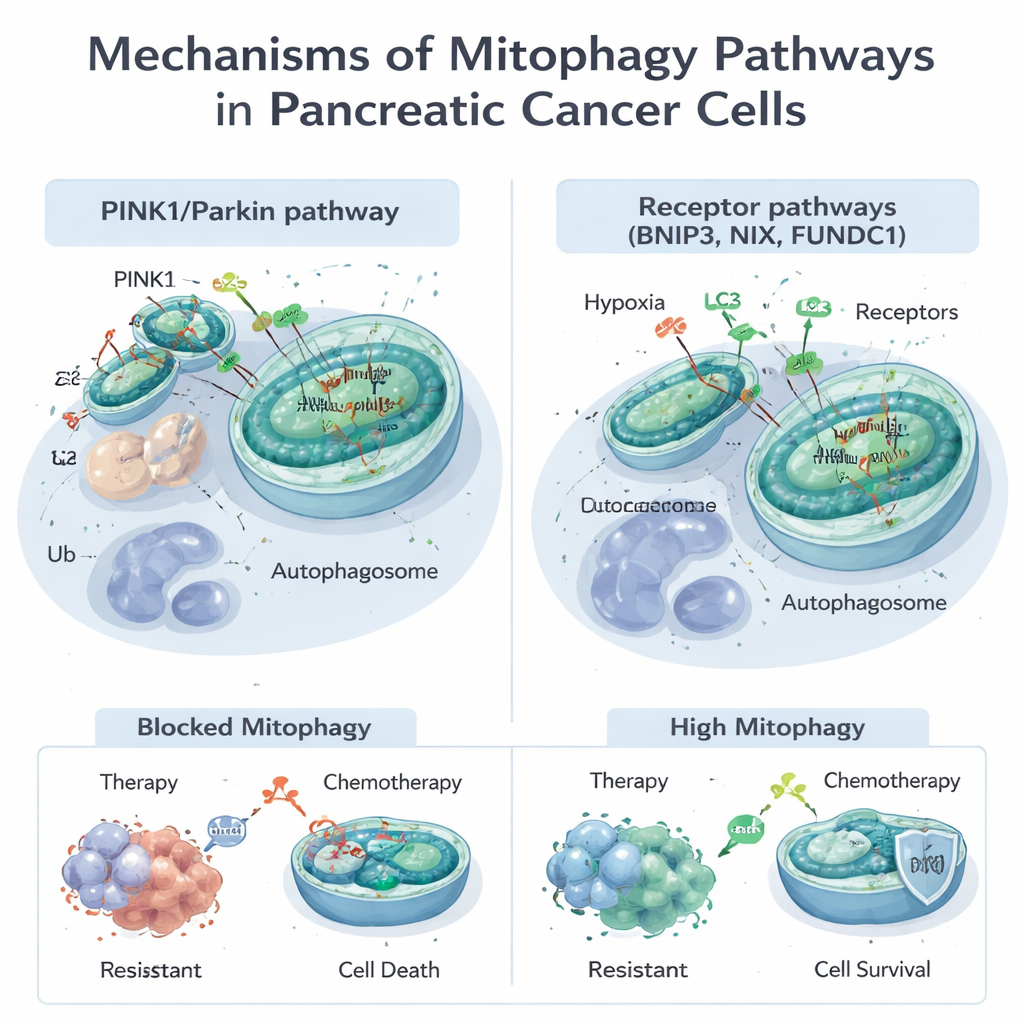
New treatment ideas and the need for smart patient matching
Because mitophagy sits at the crossroads of metabolism, stress responses, and cell death, it is an attractive target for combination therapies. Current drug strategies mostly act indirectly: some compounds interfere with mitochondrial fission and fusion; others, like chloroquine and hydroxychloroquine, block the last stages of the recycling process by disabling the fusion of waste sacs with acidic breakdown compartments. Several clinical trials are testing these drugs alongside standard chemotherapy in pancreatic cancer. Yet the results so far are mixed—some patients benefit, others do not. Part of the problem is that tumors differ widely in their baseline mitophagy and metabolic profiles: some rely heavily on mitochondrial respiration, others on glycolysis. The authors argue that future success will depend on better biomarkers and gene signatures to group patients by their mitophagy activity and energy use, and on the development of more precise drugs that selectively adjust specific mitophagy pathways rather than bluntly turning autophagy on or off. For lay readers, the key takeaway is that learning to read and carefully tweak this mitochondrial cleanup system could help convert pancreatic cancer from a stubborn foe into a more manageable disease.
Citation: Wang, Z., Lyu, Z., Palmen, R. et al. Mitophagy in pancreatic cancer: mechanistic insights and implications for novel therapeutic strategies. Cell Death Discov. 12, 93 (2026). https://doi.org/10.1038/s41420-026-02948-9
Keywords: pancreatic cancer, mitophagy, mitochondria, chemotherapy resistance, tumor metabolism