Clear Sky Science · en
Primed mesenchymal stem cells attenuate schistosomiasis fibrosis by enhancing macrophage subset switching and efferocytosis via Itgb2-Rac1 axis
Turning Down Liver Scars from a Common Parasite
Schistosomiasis, a parasitic disease that affects more than 200 million people worldwide, often leaves behind stubborn scars in the liver even after the worms are killed. These scars, known as fibrosis, can progress to cirrhosis and life‑threatening liver failure. This study explores a new cell‑based treatment that teaches the body’s own immune cells to clean up damage more efficiently, offering a fresh angle on how to reverse parasite‑induced liver scarring.
Why Liver Scars Are Hard to Heal
When Schistosoma worms lay eggs in the liver, the immune system builds tight clusters of cells—granulomas—around them. Over time, this protective response lays down excessive scar tissue, gradually stiffening the liver. A key role in this drama is played by macrophages, the body’s cleanup crew. One subtype, called “inflammatory” macrophages, fuels damage by releasing harsh chemical signals; another, more “resolving” type helps break down scar tissue and calm inflammation. The central question in this work is whether we can safely nudge macrophages from the harmful state to the healing one.
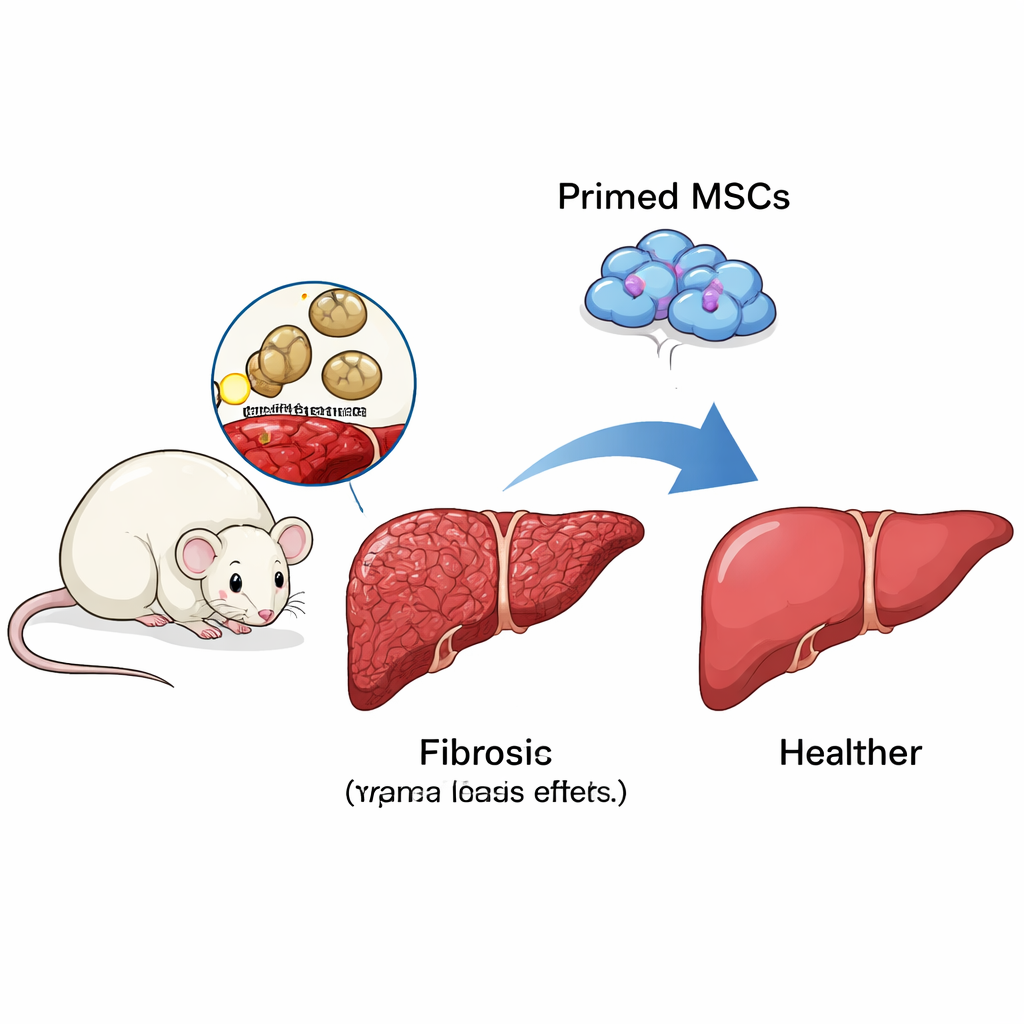
Training Stem Cells to Guide the Immune System
Mesenchymal stem cells (MSCs)—versatile cells found in bone marrow and other tissues—have attracted attention as a possible treatment for liver fibrosis. On their own, though, they do not always work well. The researchers found that “priming” MSCs in the lab with two immune signals, IFN‑γ and LPS, dramatically improved their performance in mice infected with Schistosoma japonicum. Compared with untreated animals or those given unprimed MSCs, mice receiving primed MSCs had livers that looked healthier to the naked eye, gained more weight, and showed much smaller granulomas and less collagen build‑up under the microscope. Markers of activated scar‑forming cells also fell, indicating a real drop in fibrosis, not just cosmetic changes.
Reprogramming the Body’s Cleanup Crew
Diving deeper, the team measured how primed MSCs affected macrophage behavior. In treated livers, there were fewer inflammatory macrophages and more of the pro‑resolving subtype that is better at dismantling scar tissue. At the same time, levels of classic inflammatory molecules dropped, while helpful, wound‑healing factors were kept in a balanced range rather than being driven to extremes. When the researchers blocked the arrival of new inflammatory macrophages into the liver, both the numbers of resolving cells and the benefits on fibrosis shrank. This suggested that primed MSCs do not simply add new cells, but instead convert incoming inflammatory macrophages into a more peaceful, repair‑oriented form.
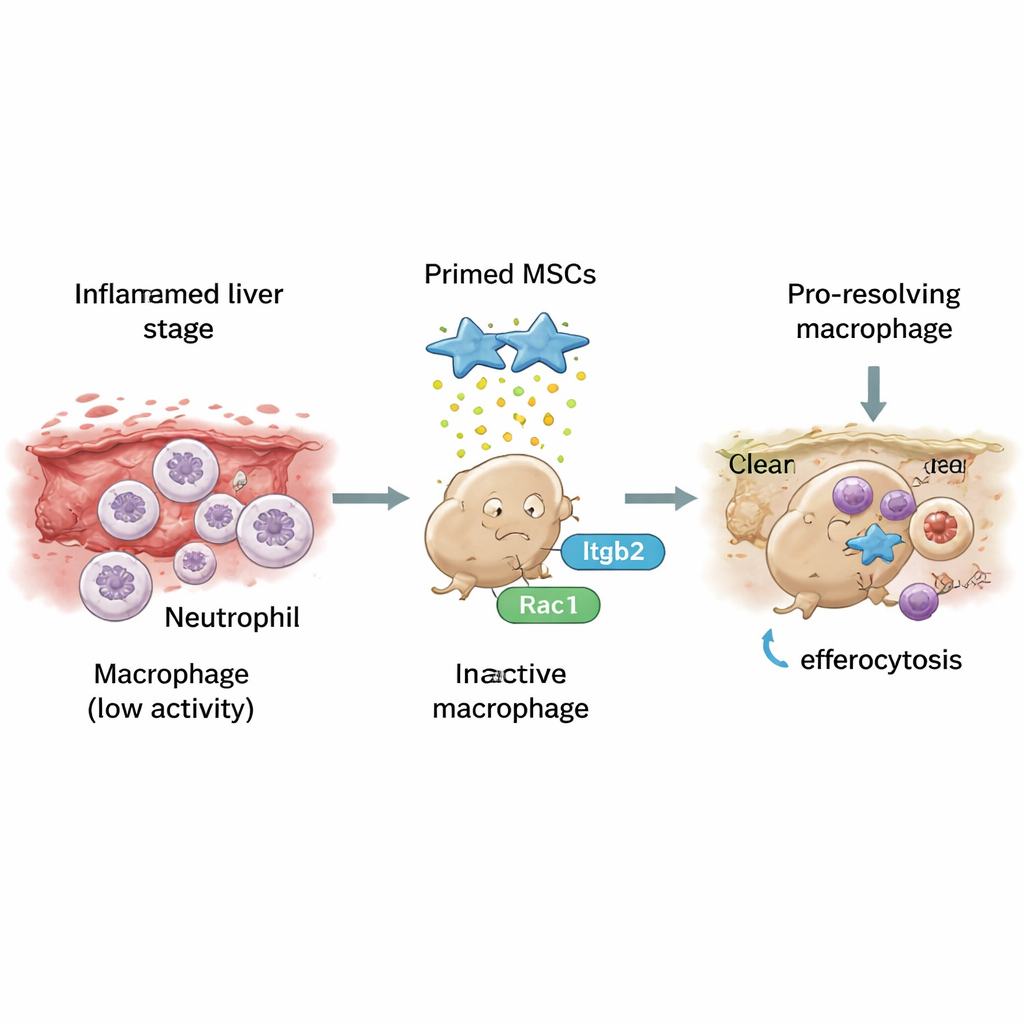
Helping Macrophages Eat Their Way to Healing
The study then turned to a process called efferocytosis—the orderly “eating” of dying cells, especially spent neutrophils, another type of immune cell. In infected mice given primed MSCs, liver macrophages were far more likely to be found with ingested neutrophils inside, a sign of active cleanup. In lab dishes, macrophages exposed to primed MSCs became better at swallowing labeled apoptotic (dying) neutrophils and, in parallel, shifted their gene activity toward a pro‑resolving profile. The researchers identified a key molecular handle for this change: a receptor called Itgb2 on macrophages, which communicates with an internal switch, Rac1, that controls the cell’s skeleton and engulfing motion. Primed MSCs boosted both Itgb2 and Rac1 activity; blocking either one sharply reduced efferocytosis and reversed the shift toward healing‑type macrophages.
What This Could Mean for Future Treatments
In plain terms, the work shows that specially primed stem cells can act like “coaches” for immune cells after schistosomiasis, encouraging them to stop fanning the flames and instead clear away debris and scar tissue more efficiently. By turning on the Itgb2–Rac1 pathway, these primed MSCs help macrophages smoothly dispose of dying cells, then adopt a scar‑resolving personality. While much remains to be done before this approach can be tried in people, the study provides a clear mechanistic roadmap for designing MSC‑based therapies that not only suppress damage, but actively help the liver rebuild itself after parasitic infection.
Citation: Lei, J., Ren, Y., Chen, Z. et al. Primed mesenchymal stem cells attenuate schistosomiasis fibrosis by enhancing macrophage subset switching and efferocytosis via Itgb2-Rac1 axis. Cell Death Discov. 12, 97 (2026). https://doi.org/10.1038/s41420-026-02947-w
Keywords: schistosomiasis, liver fibrosis, mesenchymal stem cells, macrophages, efferocytosis