Clear Sky Science · en
SUMOylation is destined for regulatory T cell-related immune dysregulation
Keeping the Immune Peace
Our immune system walks a tightrope: it must attack dangerous invaders without turning on our own tissues. A small group of white blood cells called regulatory T cells, or Tregs, act as peacekeepers on this line. This article explains how a tiny, reversible chemical tag—called SUMO—helps Tregs sense stress, adjust their behavior, and, when things go wrong, contribute either to autoimmune disease or to cancer’s ability to hide from immunity.
A Molecular Velcro for Cell Control
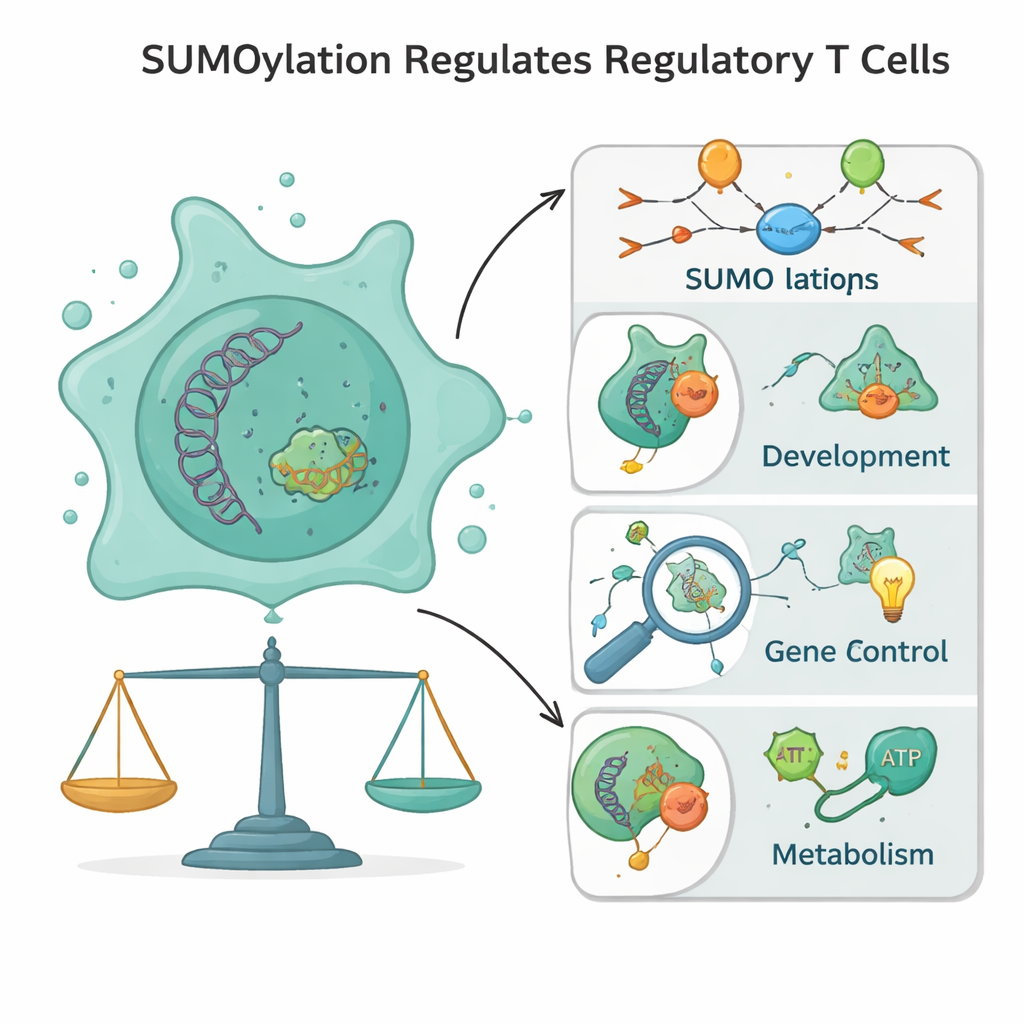
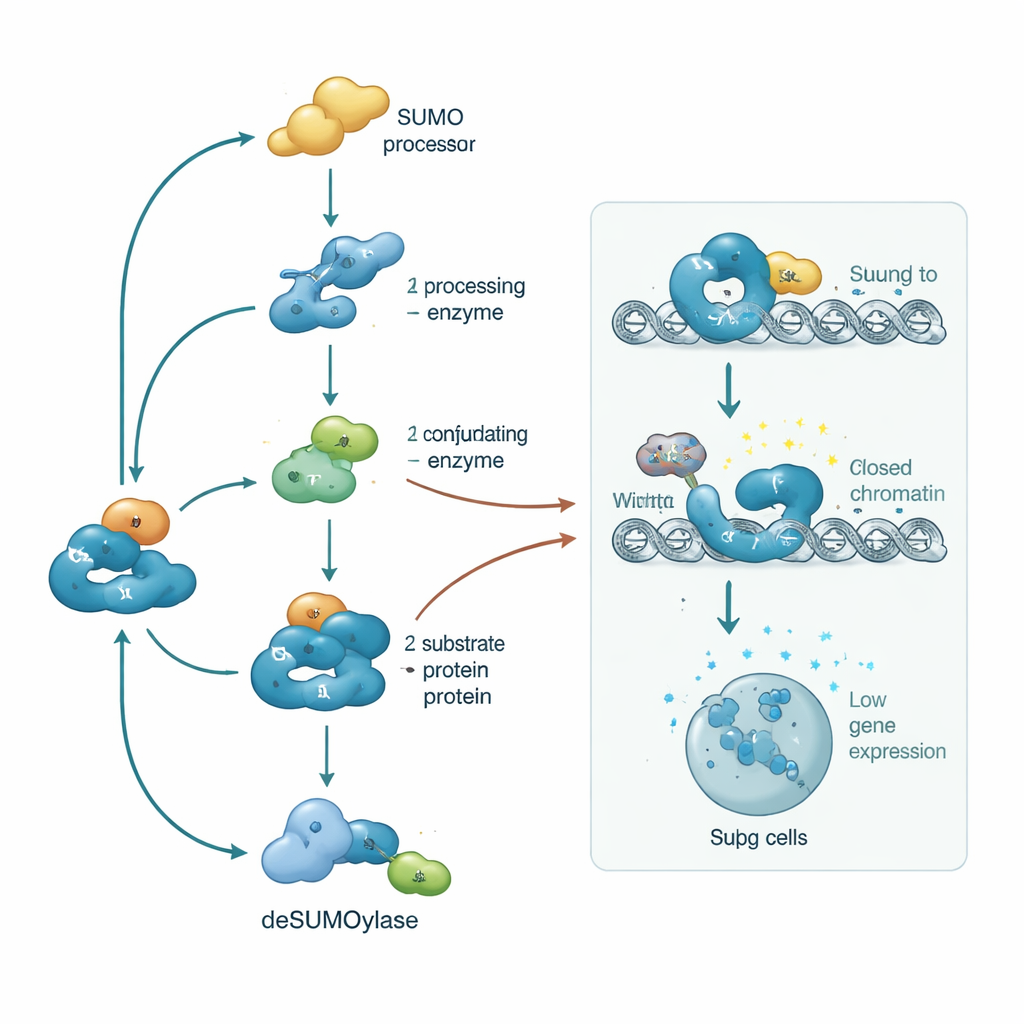
SUMOylation is the process of attaching small proteins named SUMO to other proteins, like snapping on and off molecular badges. Unlike related tags that mark proteins for destruction, SUMO tags mainly change how proteins interact, where they sit in the cell, and how active they are. Because these tags can be rapidly added and removed, they act like a molecular dimmer switch rather than an on–off breaker. In Tregs, SUMOylation is especially concentrated in the cell nucleus, where DNA is stored. There it shapes crucial processes such as DNA repair, cell division, and the reading of genes. SUMO tags also talk to other modifications—such as phosphorylation, acetylation, methylation, and ubiquitination—forming an information-rich code that lets Tregs integrate many signals at once.
Building and Protecting Regulatory T Cells
From their earliest days in the thymus, future T cells depend on SUMOylation to develop safely. When immune gene segments are cut and rejoined to build a diverse T cell receptor, SUMO tags help guide accurate DNA repair and prevent dangerous breaks from lingering. Later, as T cells are selected to avoid attacking the body, SUMO-regulated transcription factors and signaling pathways fine-tune which cells survive as Tregs. In mature Tregs, SUMO keeps chromosomes stable during cell division, supports the formation of structures like the nucleolus that are needed for protein production, and helps safeguard genome integrity under the oxidative and metabolic stress found in inflamed tissues and tumors. In this way, SUMO serves as a quiet architect of Treg differentiation and survival.
Switching Genes On and Off
Tregs are defined by a distinct gene program: they must steadily express “identity” genes such as FOXP3, BACH2, IKAROS, and IRF4 while keeping inflammatory genes shut down. SUMOylation shapes both sides of this equation. It supports a stable, Treg-specific pattern of DNA hypomethylation and activating histone marks around key regulatory regions, helping FOXP3 and its partners stay on over the long term. At the same time, SUMO tags on transcription factors like NF-κB, STATs, NFAT, and AP-1 recruit co-repressors and chromatin-modifying complexes that compact DNA into heterochromatin, making inflammatory genes harder to access. If this SUMO balance is disturbed, Tregs can lose FOXP3, gain inflammatory traits, and even convert into harmful effector cells—a shift linked to autoimmunity and loss of immune tolerance.
Fuel Choices and Survival in Harsh Environments
Tregs are metabolically flexible: they can use sugar-burning glycolysis, but they especially rely on mitochondrial pathways that burn fats and other fuels. SUMOylation acts on many of the master switches that control these choices, including AMPK, mTOR, LKB1, HIF‑1α, PPARs, and SREBPs. By tuning these factors, SUMO can shift Tregs away from excessive glycolysis toward fatty acid oxidation and oxidative phosphorylation, a strategy that helps them thrive in low-glucose, high-lactate, and oxygen-poor niches such as tumors. SUMO also regulates antioxidant systems and mitochondrial quality-control enzymes, making Tregs unusually resistant to oxidative stress that would damage other T cells. In cancers, this same machinery can be hijacked to feed and protect Tregs, allowing them to suppress anti-tumor immunity.
When Regulation Goes Wrong—and How We Might Fix It
Overall, the review argues that Tregs are effectively "addicted" to SUMOylation: they depend on this reversible tagging system to coordinate development, gene regulation, and metabolism in response to constant stress. When SUMO pathways are overactive or misdirected, they can drive Treg-mediated immune escape in tumors or, when weakened, contribute to autoimmune and inflammatory disease. For a lay reader, the key message is that a seemingly minor protein tag has outsized influence on whether immune cells calm inflammation or allow disease to flourish. Because SUMO enzymes are few in number yet control many targets, drugs that tweak this system could offer powerful new ways to either bolster Treg restraint in autoimmunity or loosen it in cancer therapy.
Citation: Qian, J., Yu, L., Tian, M. et al. SUMOylation is destined for regulatory T cell-related immune dysregulation. Cell Death Discov. 12, 90 (2026). https://doi.org/10.1038/s41420-026-02946-x
Keywords: regulatory T cells, SUMOylation, immune tolerance, autoimmunity, tumor microenvironment