Clear Sky Science · en
Colorectal cancer-derived osteopontin rewires macrophages into a pro-metastatic M2 state via the PI3K/AKT/CSF1-CSF1R axis
Why This Matters for People With Colon Cancer
Most deaths from colorectal (colon and rectal) cancer are caused not by the original tumor, but by its spread to other organs. This study looks at how cancer cells "talk" with nearby immune cells to help tumors spread. By uncovering a key message that cancer cells send to reprogram immune cells into helpers rather than fighters, the work points to new treatment strategies that might slow or stop metastasis in advanced colorectal cancer.
A Sticky Protein With a Dark Side
The researchers focused on a protein called osteopontin, or OPN, which is produced by both cancer cells and certain immune cells inside tumors. OPN has long been linked to worse outcomes in many cancers, but exactly how it promotes spread in colorectal cancer was unclear. By analyzing large patient databases and tumor samples, the team found that OPN levels were much higher in colorectal cancer tissue than in normal colon tissue, especially in patients with advanced disease and distant metastases. Patients whose tumors had more OPN generally had poorer survival, suggesting that OPN is closely tied to aggressive, hard-to-treat cancer.
It’s Not Just the Cancer Cells Themselves
Surprisingly, when the scientists forced colorectal cancer cells in the lab to make more or less OPN, the cells did not become more invasive or mobile on their own. Their growth, migration, and ability to invade through artificial membranes changed very little. This challenged the common idea that OPN mainly works by directly boosting the cancer cells’ own strength. Instead, the team looked at the tumor’s neighborhood—the so-called tumor microenvironment—especially a group of immune cells called macrophages, which can either attack tumors (an “M1” state) or support them (an “M2” state).
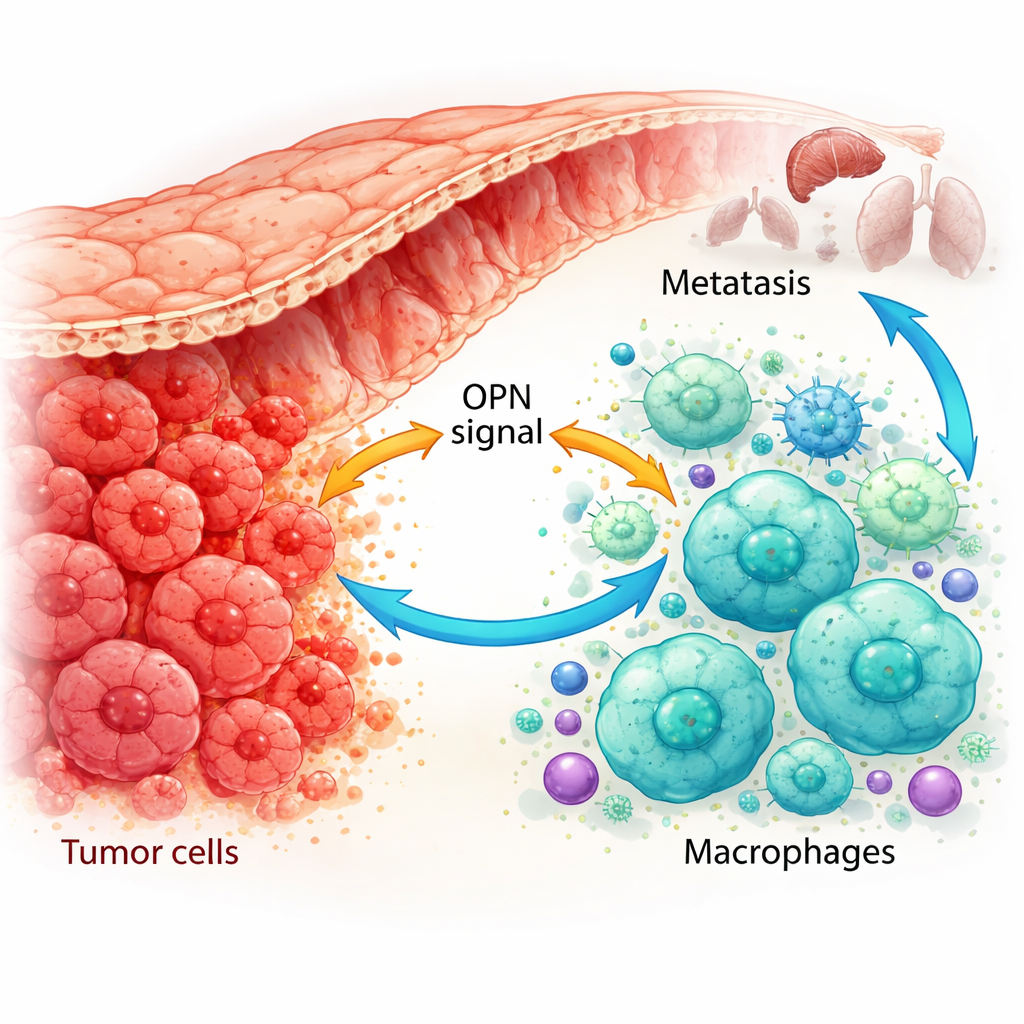
How Immune Cells Get Rewired Into Tumor Helpers
Using single-cell genetic maps and advanced staining of patient tissues, the researchers showed that OPN is produced not only by cancer cells but also by tumor-associated macrophages. In tumors that had already spread, both the cancer cells and a specific subtype of macrophages were rich in OPN. These macrophages tended to be in the M2-like, tumor-supporting state and were more abundant when tumor OPN levels were high. In mouse models, tumors engineered to produce more OPN attracted more of these M2-like macrophages and grew faster, while tumors with reduced OPN had fewer M2 macrophages and grew more slowly. This suggested that OPN is a powerful recruiter and reprogrammer of macrophages in the tumor environment.
The Hidden Signaling Chain: From OPN to Metastasis
To figure out the molecular chain of events, the team grew human macrophages together with colorectal cancer cells making different amounts of OPN. They found that high-OPN cancer cells pushed macrophages toward the M2 state and encouraged a two-way movement: macrophages migrated toward the tumor, and tumor cells became more invasive in response to signals from these macrophages. Digging deeper, the scientists discovered that OPN activates a signaling pathway inside macrophages called PI3K/AKT. This activation, in turn, makes macrophages release large amounts of another molecule, CSF1, and increase the receptor for that molecule, CSF1R, on their surface. CSF1 then acts in a loop to further enforce the M2, pro-metastatic state and keep drawing macrophages into the tumor.
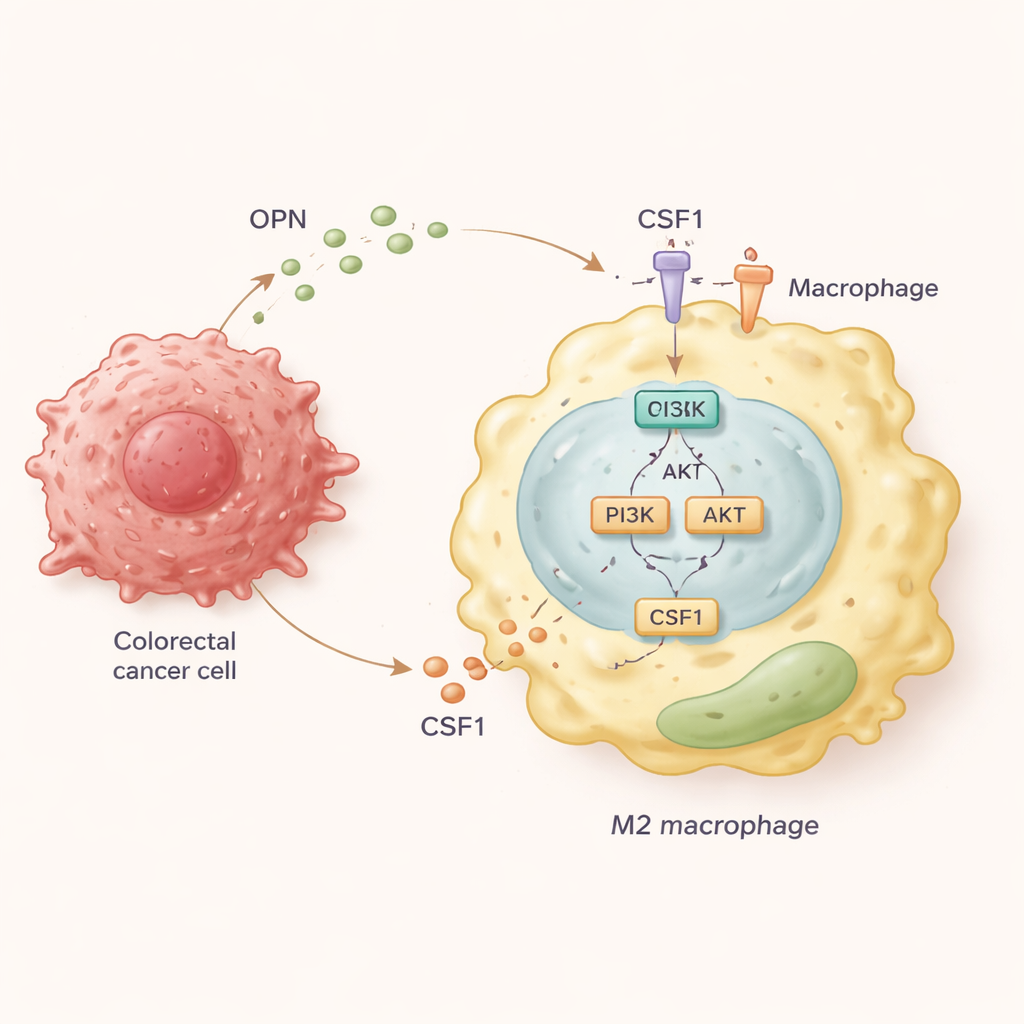
Blocking the Loop to Slow Spread
In mouse models of colorectal cancer that had spread inside the abdomen, the researchers tested a CSF1R-blocking drug called PLX3397. Mice with OPN-high tumors developed many metastatic nodules, but treatment with the CSF1R inhibitor significantly reduced both the number of metastases and the presence of M2-like macrophages in the tumors. Tumor cells showed less growth and more signs of programmed cell death. Importantly, the drug mainly depleted the tumor-promoting macrophages without greatly affecting the more inflammatory type, suggesting a relatively targeted way to weaken the tumor’s support system.
What This Means for Future Treatments
For a layperson, the main message is that some colorectal cancers succeed in spreading not just because the cancer cells are aggressive, but because they hijack nearby immune cells and turn them into accomplices. In this study, cancer-cell–derived OPN acts like a master switch that reprograms macrophages through the PI3K/AKT and CSF1/CSF1R signaling chain, creating a fertile ground for metastasis. By breaking this chain—particularly at the CSF1R step—researchers were able to reduce tumor spread in mice. This suggests that measuring OPN levels could help identify patients who might benefit from drugs targeting macrophages, and that combining such drugs with existing treatments or immunotherapies could offer new hope for people with advanced colorectal cancer.
Citation: Liang, X., Qin, F., Yuan, Z. et al. Colorectal cancer-derived osteopontin rewires macrophages into a pro-metastatic M2 state via the PI3K/AKT/CSF1-CSF1R axis. Cell Death Discov. 12, 92 (2026). https://doi.org/10.1038/s41420-026-02945-y
Keywords: colorectal cancer, osteopontin, tumor-associated macrophages, metastasis, CSF1R inhibition