Clear Sky Science · en
Targeting NXPH4/ALDH1L2 signaling suppresses enzalutamide resistance in prostate cancer
Why this matters for prostate cancer patients
Many men with advanced prostate cancer rely on a powerful hormone-blocking drug called enzalutamide. At first it often works well, shrinking tumors and slowing the disease. But in most patients, the cancer eventually finds a way around the drug and starts growing again. This study digs into how that escape happens and points to a new weak spot in drug‑resistant tumors—offering a potential path to make existing treatments work longer and better.
A common treatment meets a stubborn disease
Prostate cancer growth is closely tied to male hormones, or androgens, which act through a protein called the androgen receptor. Enzalutamide is designed to shut down this receptor and is a standard therapy when tumors stop responding to traditional hormone‑lowering treatment. Unfortunately, many tumors adapt, becoming what doctors call castration‑resistant prostate cancer. The authors of this paper set out to discover which genes help cancer cells survive enzalutamide, hoping that blocking these helpers might restore the drug’s punch.
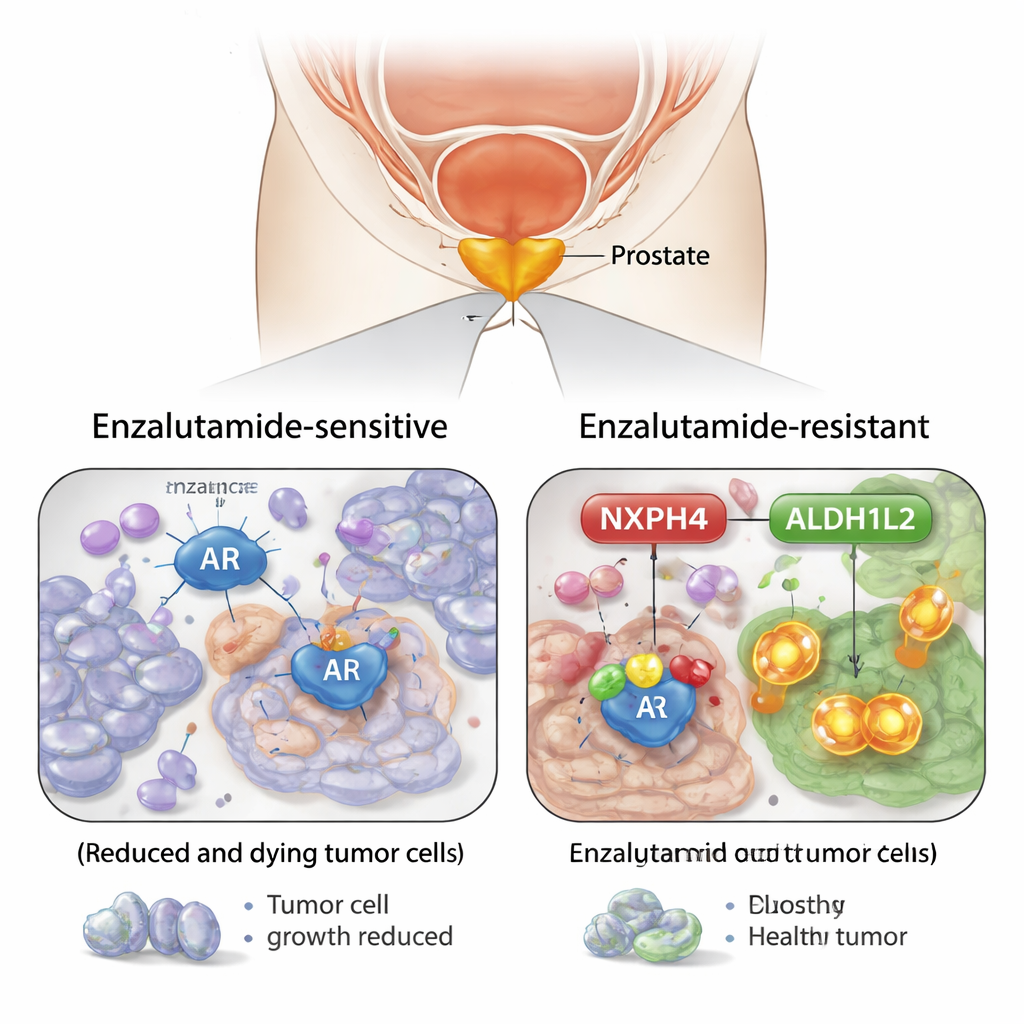
Spotlighting a surprising helper molecule
By comparing prostate cancer cells that were sensitive to enzalutamide with cells that had become resistant, the team identified a protein called NXPH4 as a key player. Resistant cells produced much more NXPH4, and its levels rose steadily when cells were exposed to enzalutamide over time. Tissue samples from patients showed the same pattern: higher NXPH4 in more aggressive tumors and in cancers that no longer responded to the drug. When the researchers reduced NXPH4 levels in resistant cells, the cells became easier to kill with enzalutamide and formed fewer colonies, both in dishes and in mice. In contrast, forcing cells to make extra NXPH4 made them harder to treat.
How hormones push NXPH4 into action
The study also asked why NXPH4 is so abundant in these tumors. The answer again pointed to the androgen receptor. The researchers showed that when cells were treated with the hormone dihydrotestosterone, NXPH4 levels went up. When they added extra androgen receptor, NXPH4 increased; when they blocked the receptor, NXPH4 fell. Detailed DNA experiments confirmed that the androgen receptor physically binds to the control region of the NXPH4 gene, turning it on. This means the same hormone‑driven system that fuels prostate cancer growth also primes cells with NXPH4, setting the stage for later drug resistance.
Powering up the cell’s energy factories
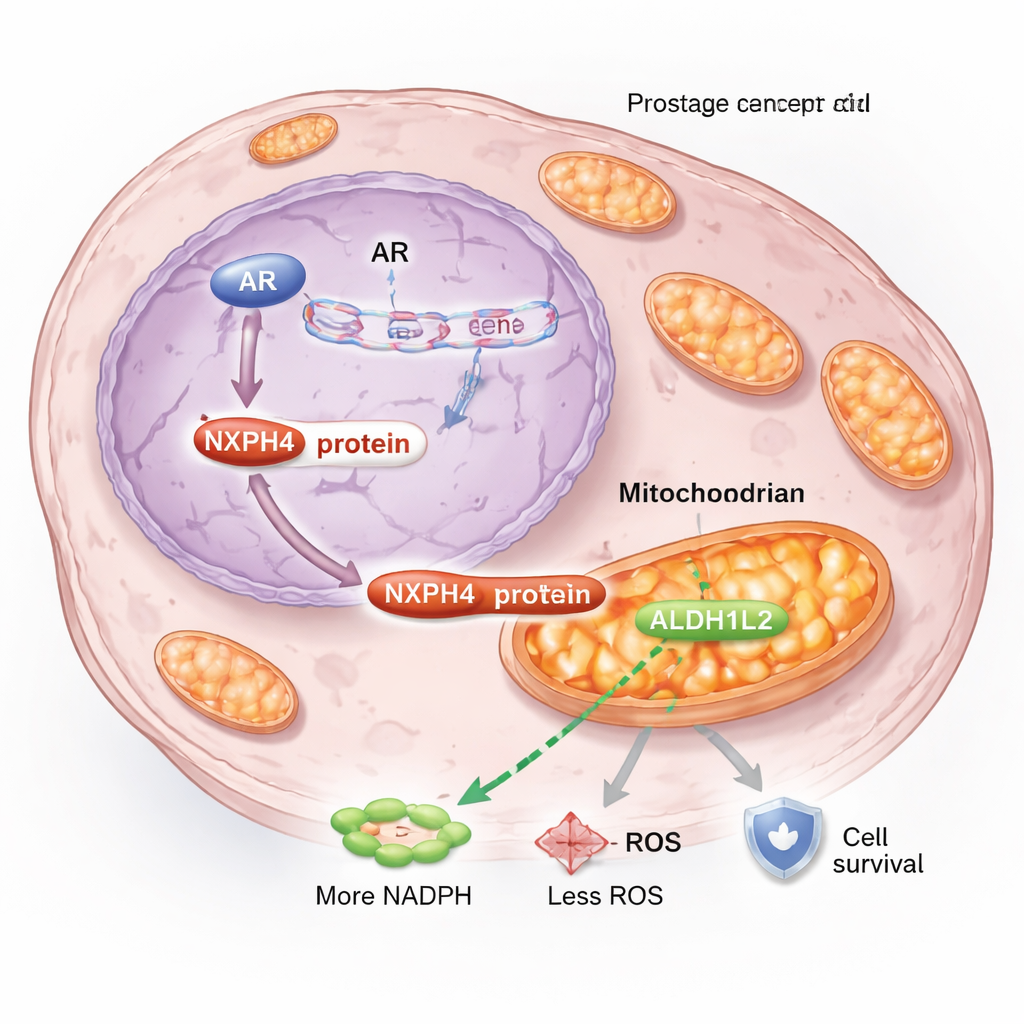
Digging deeper, the team found that NXPH4 changes how cancer cells manage energy and stress inside their mitochondria, the tiny power plants of the cell. Under hormone‑blocking conditions, some NXPH4 moves into the mitochondria and binds to another protein, ALDH1L2, which is crucial for generating NADPH, a key molecule that helps control damaging reactive oxygen species, or ROS. In resistant cells, NXPH4 and ALDH1L2 form a complex that boosts mitochondrial breathing, maintains membrane potential, increases NADPH, and lowers ROS levels. This combination keeps mitochondria healthy and allows cancer cells to withstand the stress caused by enzalutamide, rather than dying as they normally would.
Breaking the resistance circuit
When the researchers disrupted this mitochondrial support system, the results were striking. Knocking down NXPH4 in resistant cells reduced mitochondrial activity, raised oxidative stress, slowed growth, and made cells far more vulnerable to enzalutamide. Silencing ALDH1L2 had a similar effect and erased the protective benefits of extra NXPH4, confirming that these two proteins work together. In mouse models, tumors lacking NXPH4 grew more slowly, and combining NXPH4 loss with enzalutamide shrank tumors even more than either approach alone. These findings suggest that the NXPH4–ALDH1L2 partnership is not just a side effect of resistance but a driving force behind it.
What this could mean for future treatment
Put simply, this work shows that some prostate cancers survive enzalutamide by rewiring their energy supply. NXPH4, turned on by the androgen receptor, teams up with ALDH1L2 inside mitochondria to keep them running hot, protect against oxidative damage, and help tumor cells ride out hormone‑blocking therapy. Targeting NXPH4, or its interaction with ALDH1L2, could weaken this survival circuit. While drugs against NXPH4 do not yet exist, the study highlights this molecule pair as a promising new target. In the future, combining enzalutamide with medicines that disrupt NXPH4‑ALDH1L2 signaling may help patients stay ahead of resistant prostate cancer for longer.
Citation: Sun, X., Zhang, Y., Zhang, W. et al. Targeting NXPH4/ALDH1L2 signaling suppresses enzalutamide resistance in prostate cancer. Cell Death Discov. 12, 91 (2026). https://doi.org/10.1038/s41420-026-02944-z
Keywords: prostate cancer, enzalutamide resistance, mitochondrial metabolism, NXPH4, ALDH1L2