Clear Sky Science · en
Trim15 stabilizes VDAC3 via ubiquitination to suppress autophagy and enhance chemosensitivity in hypopharyngeal squamous cell carcinoma
Why this cancer study matters
Hypopharyngeal squamous cell carcinoma is a hard‑to‑treat throat cancer with low survival rates and few effective options when it spreads or stops responding to drugs. This study uncovers a hidden cellular pathway that helps decide whether these cancer cells die under treatment or quietly adapt and survive. By explaining how two proteins, Trim15 and VDAC3, control cellular self‑cleaning and stress responses, the work points to new ways to make existing chemotherapy drugs work better.
A deadly throat cancer that hides in plain sight
The hypopharynx lies deep in the throat, so tumors there often grow unnoticed until they reach an advanced stage. At that point, doctors must choose between aggressive surgery that can severely affect speech and swallowing, or organ‑preserving approaches such as chemotherapy and radiation that do not always cure the disease. Because survival has not improved much in recent decades, researchers are looking beyond traditional treatments to the underlying biology that allows these tumors to grow, spread, and resist drugs. The current study focuses on how cancer cells handle damaged mitochondria—their energy factories—and how that shapes their response to stress and therapy.
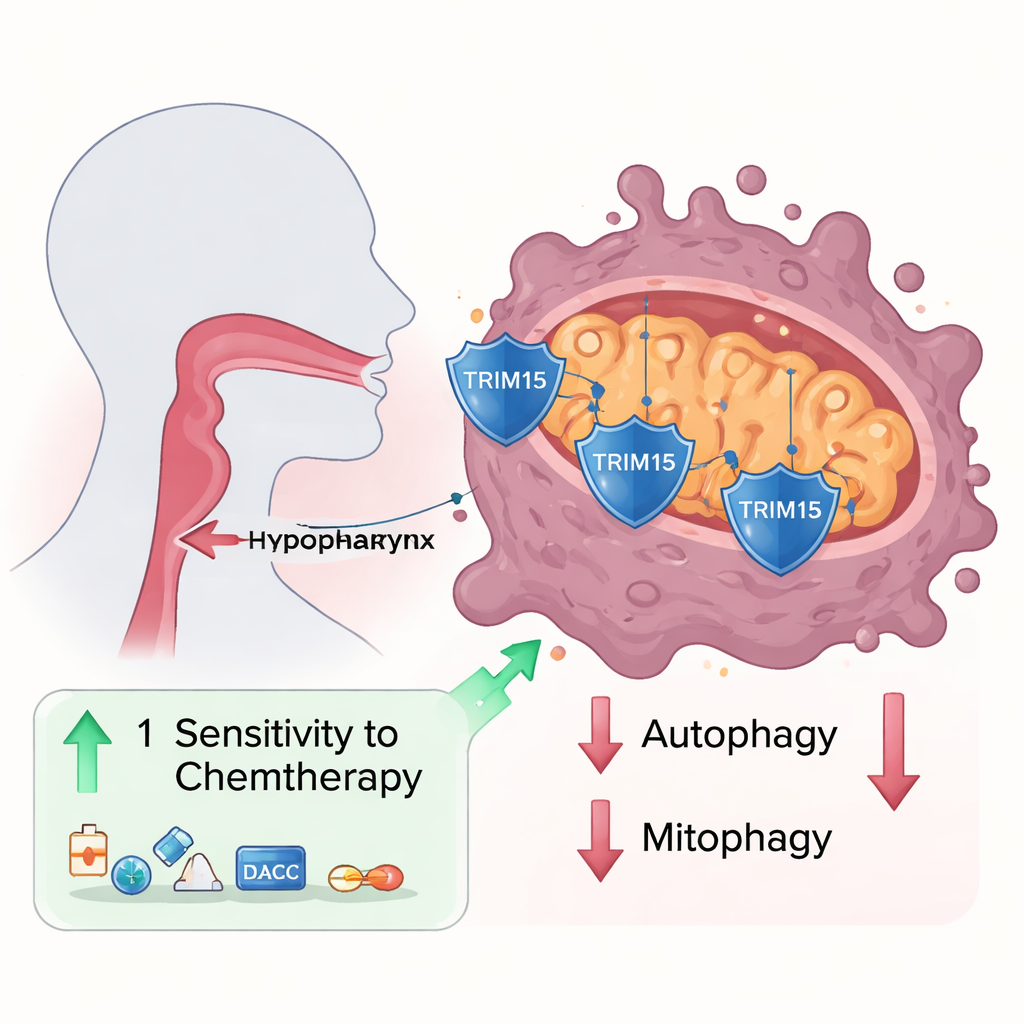
A guardian protein goes missing
The researchers examined tumor samples and cell lines from hypopharyngeal cancer and found that a protein called Trim15 was consistently reduced compared with nearby normal tissue. When they forced cancer cells to make more Trim15, the cells grew and migrated more slowly; when they removed it, the cells became more aggressive. Using biochemical pull‑down experiments, they discovered that Trim15 physically interacts with another protein, VDAC3, which sits in the outer membrane of mitochondria and acts as a gate for molecules moving in and out. Rather than marking VDAC3 for destruction, Trim15 adds a specific type of molecular tag that actually stabilizes VDAC3, keeping its levels high.
How cancer cells recycle their power plants
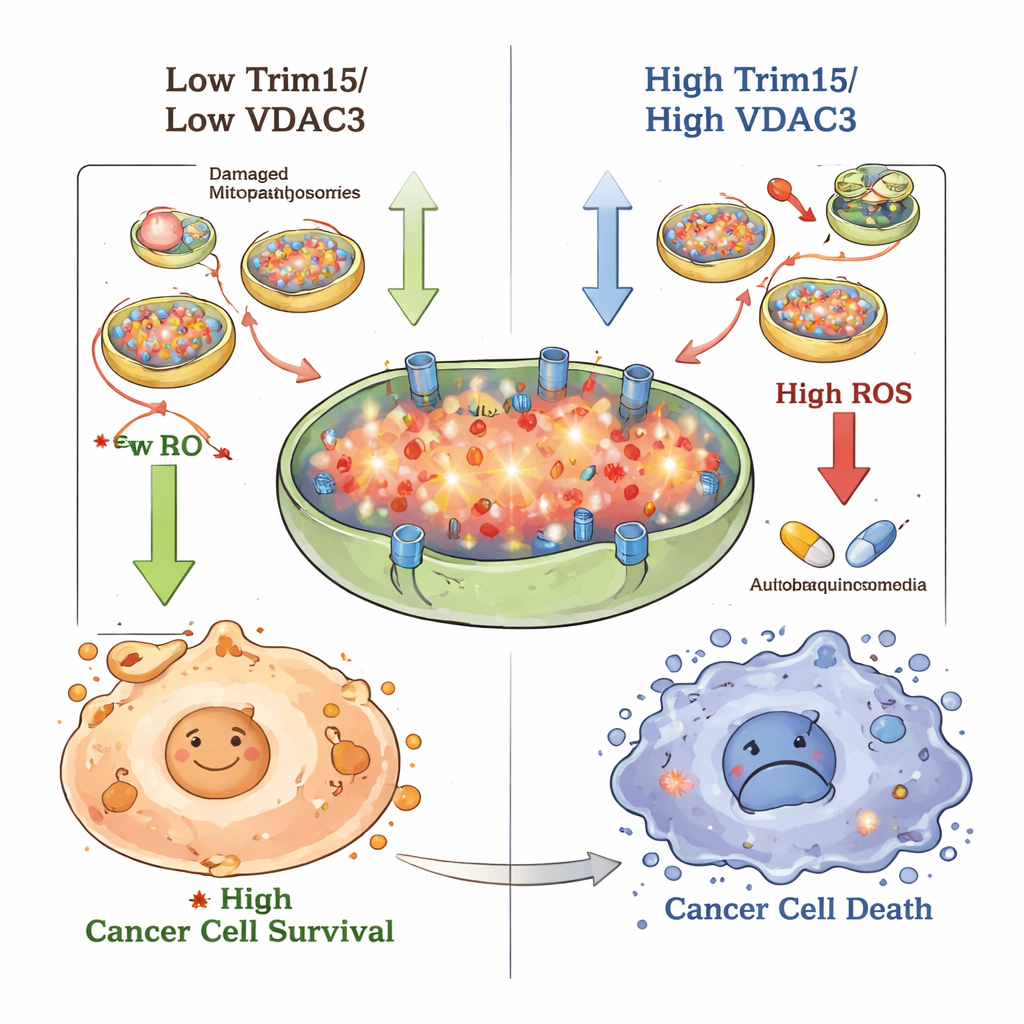
Mitochondria are continually monitored and, when damaged, can be removed through a specialized recycling process called mitophagy. The team showed that VDAC3 normally acts as a brake on this recycling. When VDAC3 was abundant, markers of general self‑digestion (autophagy) and mitophagy went down; when VDAC3 was reduced, these processes sped up, and more mitochondria were engulfed and broken down. This change also affected the balance of reactive oxygen species (ROS)—chemically reactive molecules often described as cellular exhaust. High VDAC3 pushed ROS levels up, making the cells more stressed, whereas low VDAC3 allowed the cells to clear damaged mitochondria, lower ROS, and survive better under harsh conditions.
Alcohol, oxidative stress, and drug resistance
Chronic alcohol exposure is a major risk factor for hypopharyngeal cancer, in part because concentrated ethanol repeatedly bathes the throat lining. When the researchers exposed cancer cells to alcohol in the lab, both Trim15 and VDAC3 levels fell. This drop appeared to be an adaptive move by the cancer cells: by weakening the Trim15–VDAC3 axis, they boosted mitophagy, kept ROS at safer levels, and tolerated alcohol‑induced damage. A similar response occurred with the standard chemotherapy drug 5‑fluorouracil (5‑FU): treatment lowered Trim15 and VDAC3, which likely helps tumor cells avoid lethal oxidative stress and contributes to chemoresistance.
Turning off self‑protection to boost chemotherapy
The team then asked whether blocking autophagy could strip away this protection. They used chloroquine, a long‑known antimalarial drug that interferes with the final steps of cellular self‑digestion. In cell culture, chloroquine on its own slowed the growth and movement of hypopharyngeal cancer cells and erased the growth advantage produced by VDAC3 loss. In mice bearing human tumor grafts, chloroquine and 5‑FU each shrank tumors, but the combination worked best, cutting tumor volume and weight more than either drug alone without obvious added toxicity. Importantly, forcing cells to overproduce Trim15 or VDAC3 made them more sensitive to 5‑FU, in part by raising ROS to levels the cells could no longer manage.
What this means for future treatments
To a layperson, the study shows that some hypopharyngeal cancer cells survive chemotherapy by becoming better recyclers: they rapidly clear damaged mitochondria and keep oxidative stress just low enough to avoid death. Trim15 and VDAC3 act together as a counterweight to this behavior—when present, they limit recycling and push stress higher, making cancer cells easier to kill. By targeting this Trim15–VDAC3–mitophagy axis, for example with drugs like chloroquine added to standard chemotherapy, doctors may eventually be able to make stubborn throat tumors more responsive to treatment and improve patient outcomes.
Citation: Wang, G., Shen, Y., Wang, L. et al. Trim15 stabilizes VDAC3 via ubiquitination to suppress autophagy and enhance chemosensitivity in hypopharyngeal squamous cell carcinoma. Cell Death Discov. 12, 88 (2026). https://doi.org/10.1038/s41420-026-02943-0
Keywords: hypopharyngeal cancer, autophagy, mitochondria, chemotherapy resistance, chloroquine