Clear Sky Science · en
Rottlerin triggers dual degradation of SLC7A11 and GPX4 to drive ferroptosis and chemosensitization in hepatocellular carcinoma
Why this natural compound matters for liver cancer
Hepatocellular carcinoma, the most common form of primary liver cancer, is often diagnosed late and remains difficult to treat. This study explores whether a plant‑derived molecule called rottlerin, long known for its broad anticancer effects, can be harnessed to trigger a newer form of cell death called ferroptosis in liver cancer cells. By understanding how rottlerin works at the molecular level, the researchers hope to open new avenues for more effective and less easily resisted treatments.
A new way to kill cancer cells
Traditional anticancer drugs usually push tumor cells into well‑known death programs such as apoptosis. Ferroptosis is different: it is an iron‑dependent process driven by the buildup of toxic fats in cell membranes. Liver cancer cells are especially vulnerable to this type of damage because they rely heavily on antioxidant systems to keep these fat‑based toxins in check. The team asked whether rottlerin could sabotage those defenses and thereby force liver cancer cells into ferroptosis.
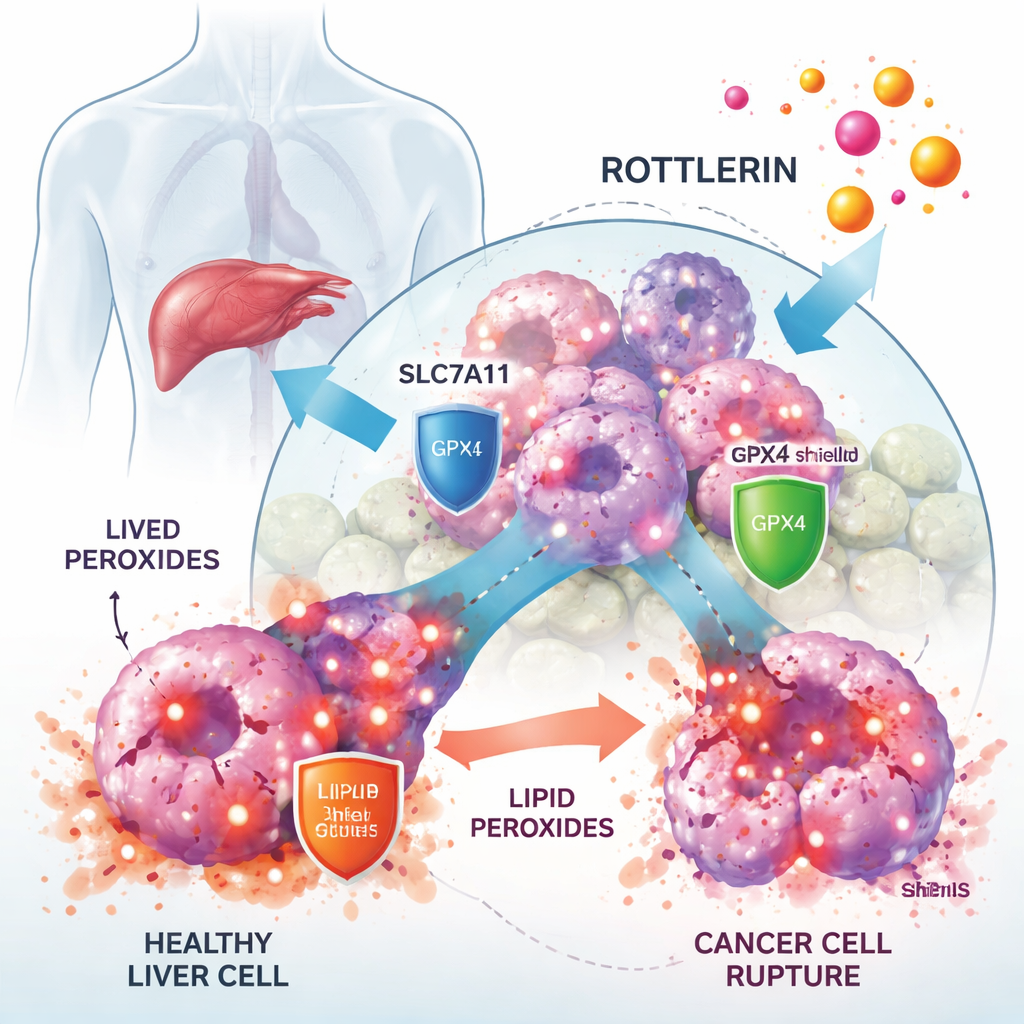
How rottlerin slows tumor growth
Working with human liver cancer cell lines, the researchers showed that rottlerin sharply reduced cancer cell growth at low micromolar doses, while normal liver‑related cells were less affected. When they added a specific ferroptosis blocker called Ferrostatin‑1, much of the rottlerin‑induced cell death was rescued, strongly suggesting that ferroptosis was the main mode of killing. Microscopy and biochemical tests backed this up: rottlerin‑treated cells accumulated high levels of lipid peroxides, showed altered mitochondria, and displayed reduced amounts of the antioxidant molecule glutathione, all hallmarks of ferroptosis.
Disarming the cells’ antioxidant shields
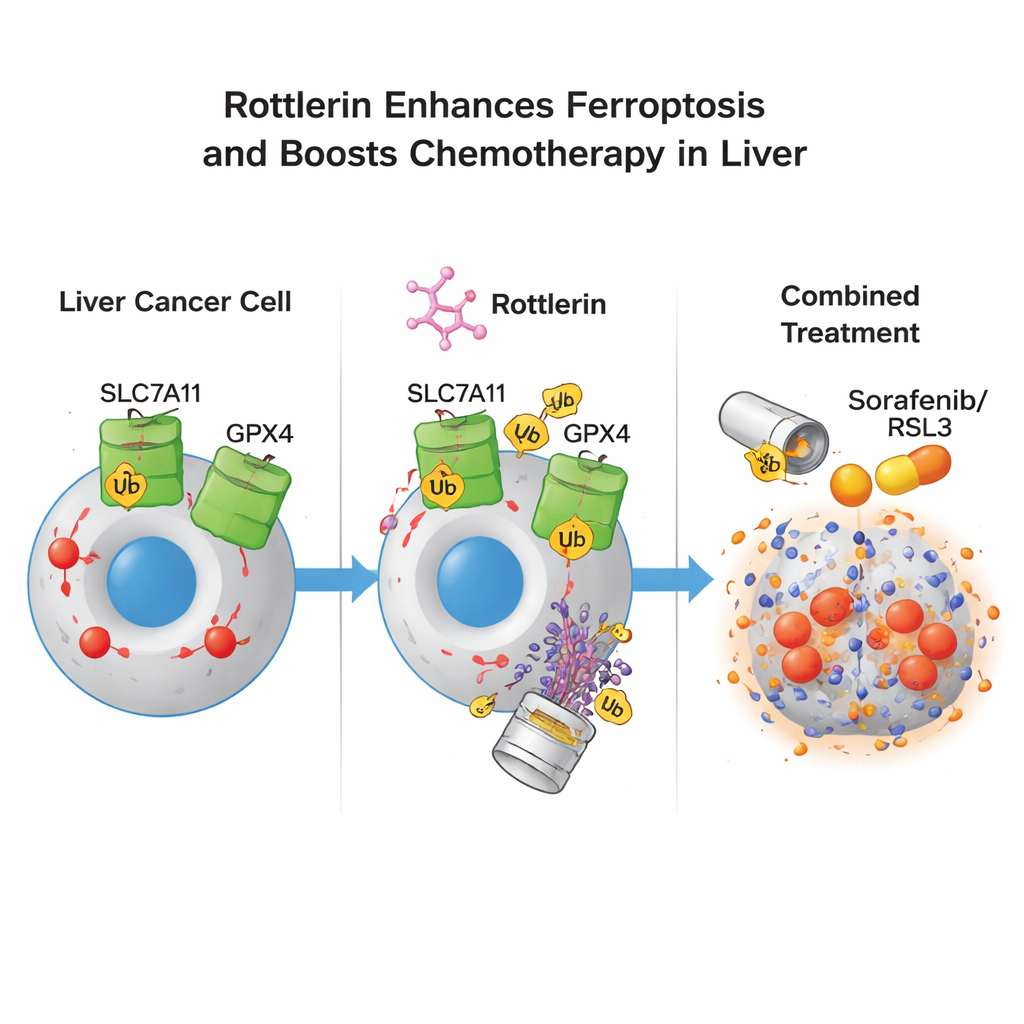
Liver cancer cells survive by leaning on a critical defense axis built from the transporter SLC7A11 and the enzyme GPX4. SLC7A11 imports cystine, a building block for glutathione, while GPX4 uses glutathione to neutralize lipid peroxides before they damage membranes. The study found that rottlerin caused both SLC7A11 and GPX4 protein levels to drop over time. This was not because the cells stopped making them, but because the proteins were being marked with small “destroy me” tags (ubiquitin) and fed into the cell’s protein‑shredding machinery, the proteasome. Blocking the proteasome reversed this loss, and artificially boosting SLC7A11 or GPX4 in the cells partially protected them from rottlerin. Together, these findings show that rottlerin acts as a dual degrader of two central ferroptosis defenses.
Boosting existing liver cancer drugs
Many patients with advanced liver cancer receive sorafenib, a standard first‑line drug that can itself induce ferroptosis, but resistance often limits its benefit. The authors tested whether low, otherwise weak doses of rottlerin could make tumor cells more sensitive to ferroptosis‑inducing drugs such as sorafenib and RSL3. In cell culture, combining low‑dose rottlerin with either drug markedly increased cancer cell death and shifted dose–response curves, indicating stronger potency. Importantly, this sensitizing effect persisted even when the classical rottlerin target protein PKCδ was genetically reduced, showing that the key action was not through this kinase but rather through degradation of SLC7A11 and GPX4. In mouse models bearing human liver tumors, combined treatment with rottlerin and sorafenib slowed tumor growth more than sorafenib alone and further lowered SLC7A11 and GPX4 levels in the tumors.
What this could mean for future treatments
For non‑specialists, the message is that rottlerin acts like a “two‑lock pick” on liver cancer cells: it simultaneously removes two crucial locks—SLC7A11 and GPX4—that normally protect cells from a lethal wave of fat‑based oxidation. Once those locks are gone, cancer cells become much more likely to undergo ferroptosis, especially when paired with existing drugs that push them in that direction. While more work is needed before rottlerin or related compounds can be used in patients, this dual‑degrader strategy offers a promising way to outmaneuver the adaptive defenses that often make liver tumors so hard to treat.
Citation: Luo, H., Jin, X., Gao, C. et al. Rottlerin triggers dual degradation of SLC7A11 and GPX4 to drive ferroptosis and chemosensitization in hepatocellular carcinoma. Cell Death Discov. 12, 89 (2026). https://doi.org/10.1038/s41420-026-02942-1
Keywords: liver cancer, ferroptosis, rottlerin, SLC7A11, GPX4