Clear Sky Science · en
Activating GCN2 and subsequently the Unfolded Protein Response with the small oral molecule NXP800 delays tumor growth in osteosarcoma
Turning Cancer’s Stress into a Weakness
Osteosarcoma is a rare but aggressive bone cancer that mostly strikes children and young adults. For decades, treatment has relied on intense chemotherapy and surgery, yet survival for patients with advanced disease has barely improved. This study explores a new pill, called NXP800, that aims to exploit a hidden weakness of cancer cells: their constant struggle with internal stress. By pushing this stress beyond a tipping point, NXP800 may force tumor cells to self-destruct while sparing most healthy cells.
A Tough Bone Cancer in Need of New Options
Osteosarcoma usually grows in the long bones of the arms and legs and can quickly spread to the lungs. Standard treatment combines several powerful chemotherapy drugs with surgery to remove the tumor. While this approach cures many patients with localized disease, outcomes are poor for those whose tumors return or have already spread at diagnosis. Because current drugs have reached their limits, researchers are searching for therapies that attack osteosarcoma in entirely new ways, ideally by targeting traits that distinguish cancer cells from normal tissues.
Making Stress Work Against Tumor Cells
Cancer cells live under constant pressure. They divide rapidly, use large amounts of energy and oxygen, and often experience harsh conditions such as low oxygen or exposure to chemotherapy. To cope, they rely on emergency systems that help them fold and process proteins correctly and adjust their metabolism. One of these systems is known as the “unfolded protein response,” which is triggered when the cell’s protein factory, the endoplasmic reticulum, is overloaded. Another, the “integrated stress response,” senses different kinds of stress and slows down protein production. Normally, these pathways help cancer cells survive—but if pushed too hard for too long, they can flip from protecting the cell to triggering its death.
NXP800 Hits a Key Stress Sensor
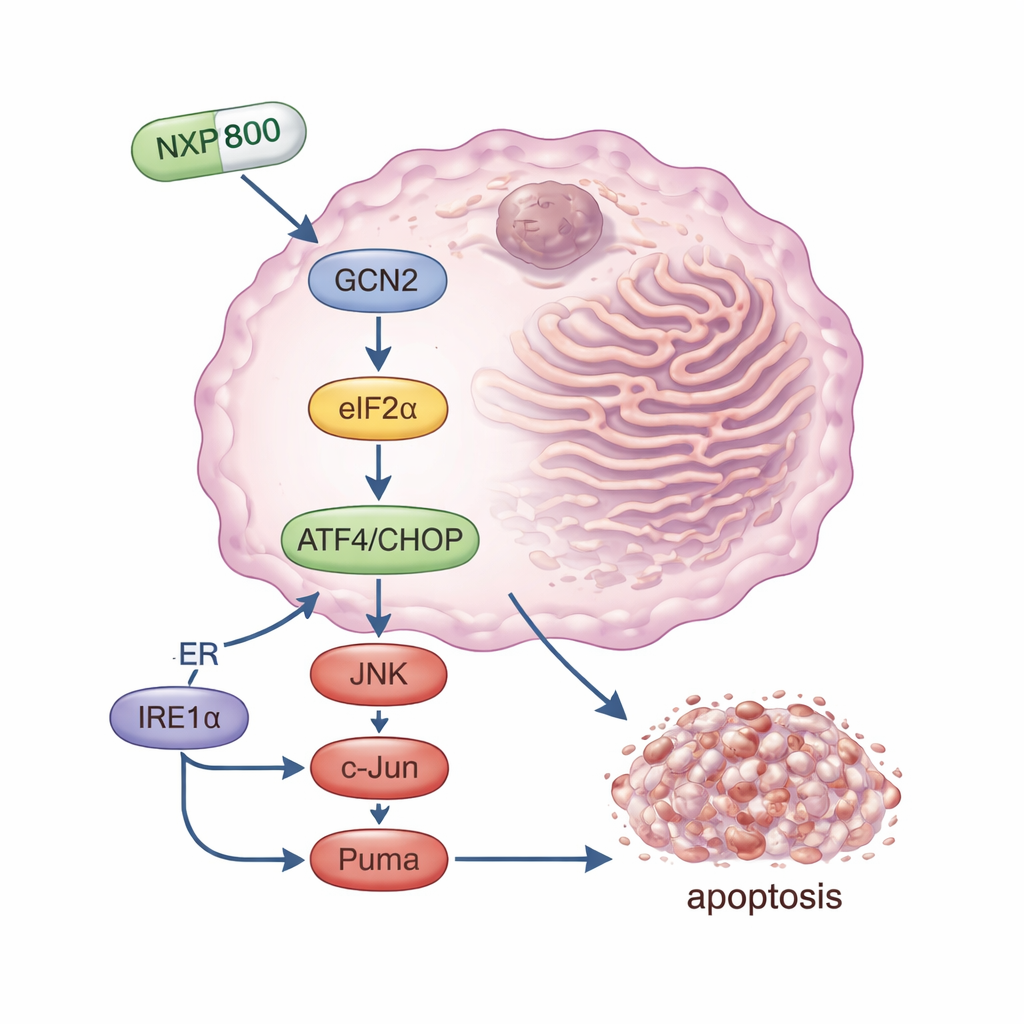
NXP800 is an oral small molecule that was first developed to block a heat shock pathway involved in cancer survival. In this study, researchers discovered that in osteosarcoma cells its main effect is different: it strongly activates a stress-sensing protein called GCN2. Once switched on, GCN2 modifies another factor, eIF2α, which rapidly reduces overall protein production while boosting the levels of specific stress “master switches,” especially a protein named ATF4 and its partner CHOP. Together, these molecules drive the cell toward programmed death. The team showed that when they blocked GCN2—either by silencing its gene or with a separate inhibitor—NXP800 could no longer trigger this chain reaction effectively, and cancer cells became far less sensitive to the drug. This pinpoints GCN2 as a crucial target of NXP800 in osteosarcoma.
Engaging Multiple Death Pathways Inside the Tumor
Beyond the GCN2 arm of the stress network, NXP800 also switched on another route linked to cell death. It activated a sensor called IRE1α, which in turn stimulated signaling proteins JNK and c-Jun. This branch is known to promote the production of Puma, a powerful pro-death molecule that damages the cell’s internal survival machinery. In laboratory experiments, NXP800 slowed osteosarcoma cell growth, arrested their cell cycle, and activated enzymes and protein changes that mark apoptosis, or controlled cell death. Importantly, normal bone-related stem cells were much less affected, suggesting a degree of selectivity for tumor cells that are already living at the edge of tolerable stress.
From Lab Bench to Living Models
To see whether these cellular effects translated into real tumor control, the researchers tested NXP800 in mice implanted with human osteosarcoma cells near the tibia. Mice that received NXP800 by mouth developed tumors that grew significantly more slowly and remained roughly half the volume of tumors in untreated mice by the end of the study. Tumor samples from treated animals showed clear signs of the drug’s mechanism: higher levels of the stress markers eIF2α and ATF4, fewer cells actively dividing, and more cells undergoing apoptosis. Separate safety experiments in healthy mice revealed no major organ damage, and liver tests were not worsened, supporting a manageable safety profile at the doses used.
What This Could Mean for Patients
Taken together, this work shows that NXP800 can delay osteosarcoma growth by deliberately overactivating the cancer cell’s own stress defenses until they trigger self-destruction. By targeting the GCN2 stress sensor and related pathways, the drug offers a brand-new way to weaken tumors that have resisted traditional chemotherapy. Because NXP800 is already being tested in early clinical trials for other cancers, these findings raise the possibility that it could be repositioned for osteosarcoma, either alone or combined with existing drugs. While human trials will be needed to confirm benefit and safety, this study provides a compelling proof-of-principle that turning cellular stress against the tumor may open a new therapeutic chapter for patients with this difficult bone cancer.
Citation: Racineau, E., Lallier, M., Postec, A. et al. Activating GCN2 and subsequently the Unfolded Protein Response with the small oral molecule NXP800 delays tumor growth in osteosarcoma. Cell Death Discov. 12, 94 (2026). https://doi.org/10.1038/s41420-026-02941-2
Keywords: osteosarcoma, NXP800, stress response, GCN2 pathway, apoptosis