Clear Sky Science · en
Targeting metabolic vulnerabilities: REV-ERB agonist SR9009 potentiates sorafenib efficacy in liver cancer
Why starving cancer cells of energy matters
Liver cancer is one of the deadliest cancers worldwide, and many patients with advanced disease still rely on an older drug called sorafenib. While sorafenib can slow tumor growth, cancer cells often adapt and become resistant, limiting patients’ survival. This study explores a new way to make sorafenib work better by attacking how liver cancer cells fuel themselves, offering a glimpse of future treatments that could hit tumors harder without adding serious side effects.
How current liver cancer drugs fall short
Sorafenib has long been a mainstay treatment for patients whose liver tumors cannot be removed surgically. Newer immunotherapies now help some people live longer, but many patients either do not qualify for these treatments or do not respond to them. For those patients, sorafenib remains important, yet resistance often appears within months. The researchers asked a simple but powerful question: what changes inside liver cancer cells allow them to escape sorafenib’s effects, and can those changes be turned into new weak spots for therapy?
Cancer cells turn up their power plants
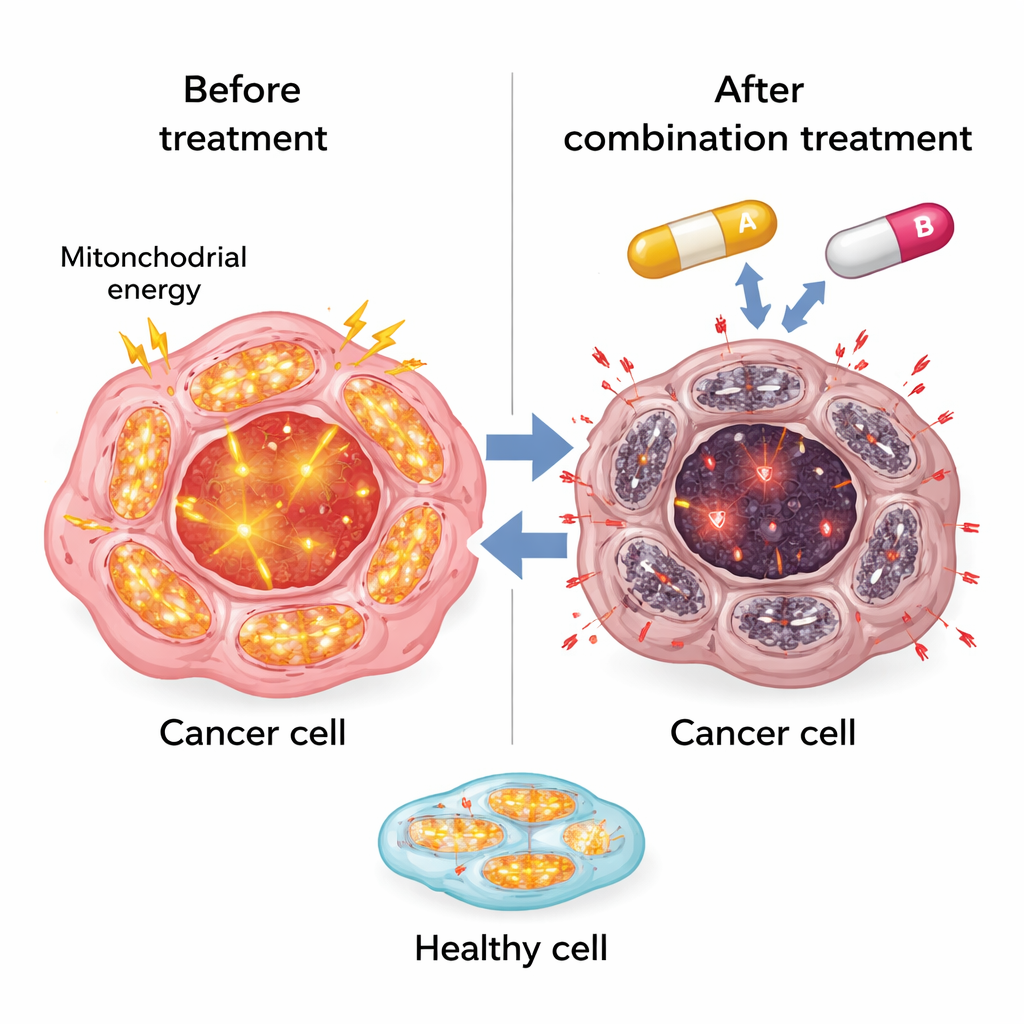
To find answers, the team compared liver cancer cells that still responded to sorafenib with cells that had learned to survive the drug. Using large-scale RNA sequencing, they discovered that resistant cells greatly increased the activity of genes involved in mitochondrial oxidative phosphorylation—the main way cells generate energy in their tiny “power plants,” the mitochondria. Other stress-protection genes, including those tied to handling a molecule called heme, were also boosted. Together, these changes suggested that resistant cancer cells cope with sorafenib by rewiring their energy production and stress defenses rather than simply ignoring the drug’s original targets.
A second drug that cuts off the power
The researchers then turned to SR9009, an experimental compound originally designed to act on clock-related proteins called REV-ERBs that help control metabolism. SR9009 is known to interfere with mitochondrial activity and to be more harmful to tumor cells than to normal cells. In both mouse and human liver cancer cells, combining SR9009 with sorafenib sharply reduced cell survival compared with either drug alone. The two drugs together cut mitochondrial energy production almost to zero, forcing the cells to rely on backup sugar-burning pathways that could not fully compensate. Lab tests showed steep drops in cellular ATP—the universal energy currency—and fewer colonies of surviving cancer cells. Importantly, normal fibroblasts and noncancerous liver cells were much less affected, hinting that the strategy may selectively strike tumor tissue.
Disrupting heme balance and triggering toxic stress
The study also uncovered a second vulnerability: how resistant cells manage heme, a small iron-containing molecule essential for many enzymes, including those in mitochondria. Resistant liver cancer cells turned on high levels of HO-1, an enzyme that breaks down free heme, and increased ferritin, which safely stores released iron. This appeared to shield them from heme-related damage. SR9009 changed this balance. In resistant cells, SR9009 raised levels of free heme and boosted markers of oxidative stress such as reactive oxygen species and lipid damage, especially when combined with sorafenib. At the same time, mitochondrial proteins decreased, reinforcing the idea that overwhelming the cell’s energy factories and stress defenses pushes cancer cells past a point of no return.
Stronger tumor control in animal models
To see if these effects translated beyond dishes of cells, the team tested the drug pair in mice. In animals carrying both ordinary and sorafenib-resistant liver tumors, only the combination of sorafenib plus SR9009 shrank the resistant tumors. In a separate model where liver cancers arose spontaneously after a cancer-causing chemical exposure, adding SR9009 to a reduced dose of sorafenib led to tumor shrinkage in more than 90% of nodules, while tumors in mice treated with sorafenib alone largely continued to grow. Remarkably, the combination did not increase overall toxicity: mice kept stable body weight, blood counts remained normal, and liver injury markers were lower when SR9009 was included, suggesting a protective effect on healthy liver tissue.
What this could mean for patients
For a general reader, the key message is that this work outlines a clever way to make an existing liver cancer drug more effective by exploiting how tumors adapt. Sorafenib-resistant cells survive in part by boosting their energy factories and stress-control systems. SR9009 knocks out these supports, leaving cancer cells starved of energy and overloaded with toxic by-products, while sparing normal cells. Although SR9009 itself is not yet an approved medicine and these findings come from preclinical models, the study provides a roadmap: pairing standard cancer drugs with agents that target tumor-specific metabolism could overcome resistance and allow lower, safer doses. In the long run, such combination strategies might give patients with advanced liver cancer more time and better quality of life.
Citation: Sabbioni, S., Guerriero, P., Shankaraiah, R.C. et al. Targeting metabolic vulnerabilities: REV-ERB agonist SR9009 potentiates sorafenib efficacy in liver cancer. Cell Death Discov. 12, 86 (2026). https://doi.org/10.1038/s41420-026-02940-3
Keywords: liver cancer, drug resistance, cancer metabolism, combination therapy, mitochondria