Clear Sky Science · en
Hepatocyte TIA1 constrains metabolic steatohepatitis by translationally suppressing Srebf1 mRNA in stress granules
Why this liver study matters
Fatty liver disease is now one of the most common chronic liver problems worldwide, closely tied to obesity and type 2 diabetes. For many people it remains silent, but in some it progresses to a dangerous, inflammatory form that can lead to scarring, liver failure, and cancer. This study asks a simple but crucial question: do liver cells have an internal “brake” that helps keep fat buildup and inflammation in check—and what happens when that brake fails?
A growing problem in modern livers
The authors focus on a condition now called metabolic dysfunction-associated steatotic liver disease (MASLD), and its more severe inflammatory stage, MASH. These illnesses arise when energy intake chronically exceeds expenditure, driving fat accumulation inside liver cells, toxic stress, and persistent inflammation. Today, only one drug is approved and it offers only partial benefit, so researchers are hunting for molecular pathways that could be turned into better treatments. One promising area involves how cells fine-tune the use of their genetic messages after they are made, rather than just controlling which genes are switched on or off.
An unexpected protector inside liver cells
The team homed in on a protein called TIA1, which binds RNA and helps assemble tiny droplets in the cell called stress granules. These granules temporarily store stalled genetic messages when cells are under duress. By analyzing human liver samples and several mouse models of diet-induced fatty liver, the researchers found that TIA1 levels rise in liver cells during early MASLD and MASH. Paradoxically, people and mice with more advanced liver cancer tend to have lower TIA1 levels, hinting that it may act as a protector early in disease but be lost later on. In mice fed different liver-damaging diets, TIA1 was especially abundant in hepatocytes, the main metabolic workhorses of the liver.
What happens when the brake is removed
To test TIA1’s function, the scientists created mice lacking TIA1 specifically in hepatocytes and exposed them to high-fat and other liver-stressing diets. Compared with normal littermates, these knockout mice developed much larger, paler livers loaded with fat, accompanied by more inflammation, oxidative stress, and fibrous scarring. Markers of liver injury surged in their blood. In contrast, restoring extra TIA1 in the liver using a gene therapy vector reduced fat accumulation, dampened inflammation, and improved liver chemistry. Parallel experiments in cultured mouse liver cells exposed to a toxic saturated fat showed that TIA1 is required to form stress granules and that these granules help limit fat buildup and cellular stress.
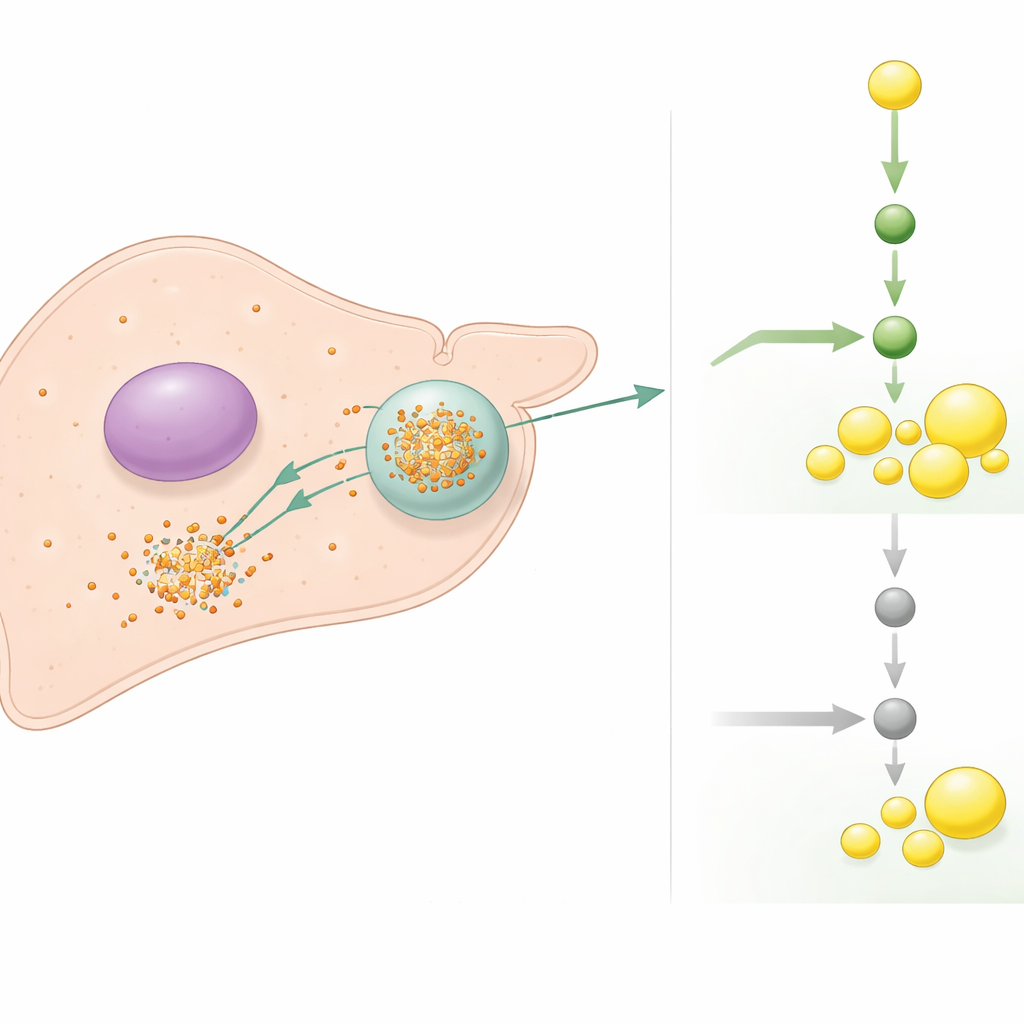
A molecular traffic stop for fat-making signals
Diving deeper, the authors discovered that TIA1 directly recognizes and binds the tail end of the messenger RNA that encodes SREBP1, a master switch for new fat production in liver cells. Under lipotoxic stress, TIA1 herds this SREBP1 message into stress granules, where it is both prevented from being translated into protein and more rapidly degraded. When TIA1 is removed, the SREBP1 message escapes this sequestration, persists longer, and is translated more efficiently, driving a strong rise in fat-making enzymes and worsening steatosis and inflammation. Drugs that block SREBP1 activity were able to rescue much of the damage caused by TIA1 loss in both liver cells and mice, underscoring that this fat-production pathway is the main culprit downstream of TIA1 failure.
What this means for patients and future treatments
Taken together, the study reveals a built-in safety system in hepatocytes: during metabolic stress, TIA1 organizes stress granules that capture and silence the message for SREBP1, restraining fat synthesis and inflammatory damage. When this TIA1–granule–SREBP1 axis is impaired, fatty liver disease progresses more quickly toward steatohepatitis and scarring. Although much work remains—including confirming these mechanisms in larger patient cohorts and in females as well as males—the findings point to new therapeutic angles. Boosting TIA1 activity, stabilizing protective stress granules, or more directly dialing down SREBP1 could all offer ways to reinforce the liver’s own defenses against modern metabolic strains.
Citation: Liu, R., Chen, J., Wang, J. et al. Hepatocyte TIA1 constrains metabolic steatohepatitis by translationally suppressing Srebf1 mRNA in stress granules. Cell Death Dis 17, 357 (2026). https://doi.org/10.1038/s41419-026-08682-5
Keywords: fatty liver disease, stress granules, RNA-binding proteins, lipid metabolism, SREBP1