Clear Sky Science · en
BMSC-derived extracellular vesicles enhance osteosarcoma proliferation and metastasis via the circRNA-0010220/β-catenin pathway
Why this bone cancer story matters
Osteosarcoma is the most common bone cancer in children and young adults, and it often spreads to the lungs. Even with modern chemotherapy and surgery, many patients still face relapse and life‑threatening metastasis. This study digs into the hidden “conversations” between bone cancer cells and nearby stem cells in the bone marrow, uncovering a tiny molecular messenger that helps tumors grow and spread—and that could become a new therapeutic target.
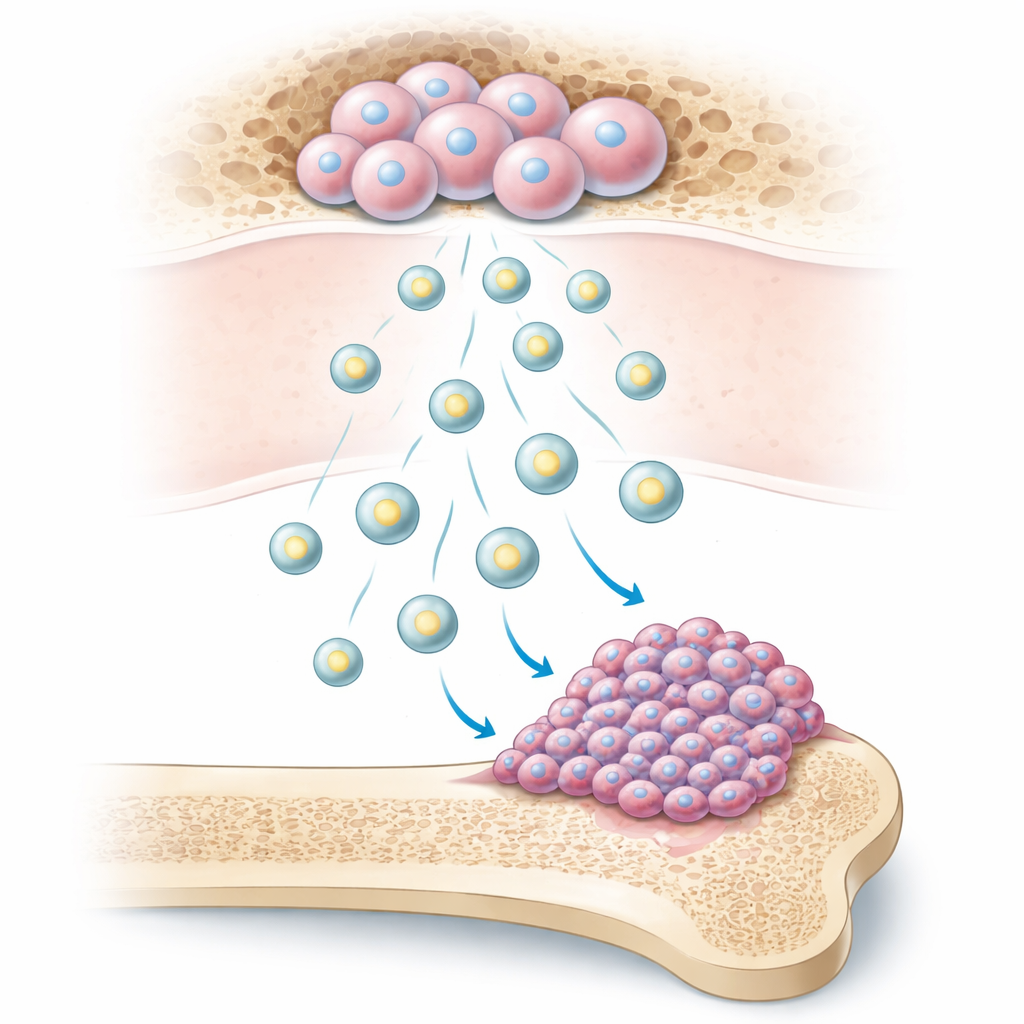
Tiny packages with big influence
Our bones contain bone marrow mesenchymal stem cells, versatile cells that help maintain and repair bone. These stem cells release minuscule, membrane‑wrapped bubbles called extracellular vesicles. The researchers show that osteosarcoma cells readily absorb these bubbles. When that happens, the cancer cells begin to divide faster, form more colonies, move more quickly across surfaces, and invade through barriers in lab tests. In mice, tumors exposed to these vesicles grew larger and produced more lung metastases, indicating that these microscopic packages powerfully boost the cancer’s aggressiveness.
A circular message that strengthens cancer
The team then examined which genetic messages inside the vesicles changed osteosarcoma cells the most. They focused on circular RNAs, a recently discovered class of RNA molecules that form closed loops and are unusually stable. Sequencing revealed one standout, called circRNA‑0010220, which was strongly increased in osteosarcoma cells treated with stem‑cell vesicles and in patient tumor samples. When the scientists reduced circRNA‑0010220 levels in the stem cells, their vesicles lost much of their ability to stimulate cancer cell growth, movement, and invasion—both in cultured cells and in mouse models. This pinpointed circRNA‑0010220 as a key driver of the harmful effects.
An epigenetic brake is turned off
Diving deeper, the researchers mapped how this circular RNA rewires cancer cell behavior. Inside osteosarcoma cells, circRNA‑0010220 travels into the nucleus, the cell’s control center. There it latches onto a protein called EZH2, an enzyme that adds chemical tags to histones—the spool‑like proteins around which DNA is wrapped. Acting together, circRNA‑0010220 and EZH2 increase a specific tag known as H3K27me3 on the DNA control region of a gene called CTNNBIP1. This modification tightly packs the chromatin in that region, effectively turning the gene off. Because CTNNBIP1 normally acts as a brake on a powerful growth‑promoting pathway, silencing it removes an important safeguard against runaway cancer behavior.
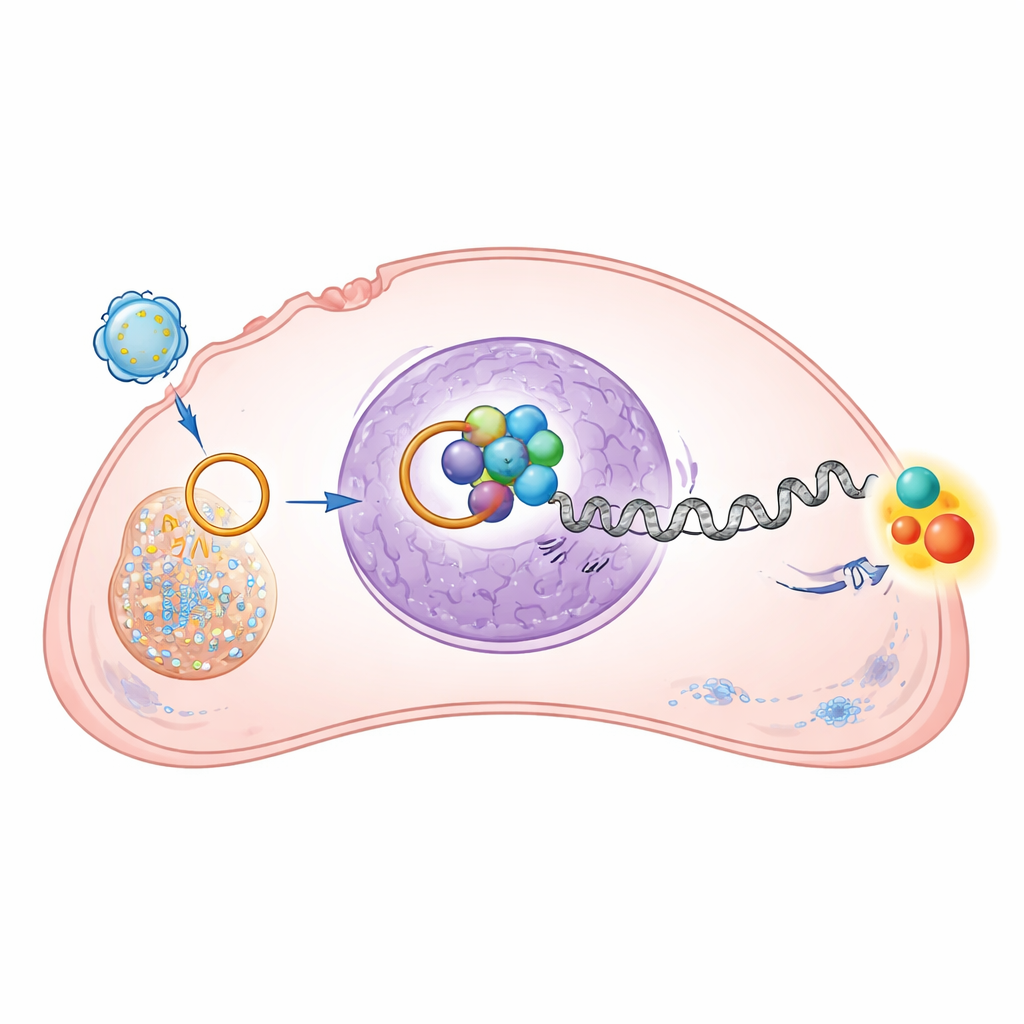
Switching on a powerful growth pathway
CTNNBIP1 restrains the Wnt/β‑catenin pathway, a well‑known signaling system that, when overactive, encourages cells to divide, migrate, and survive. By shutting down CTNNBIP1, circRNA‑0010220 indirectly boosts levels and nuclear activity of β‑catenin in osteosarcoma cells. The study showed that genes controlled by this pathway—linked to cell cycle progression, stem‑like traits, and invasion—became more active when vesicle‑borne circRNA‑0010220 was present. When the researchers used a drug that specifically blocks β‑catenin, the pro‑growth and pro‑migration effects of circRNA‑0010220 were largely reversed. This confirms that the circular RNA operates through this growth pathway to drive tumor progression.
What this means for future treatment
Taken together, the work outlines a full chain of events: bone marrow stem cells send vesicles loaded with circRNA‑0010220 to osteosarcoma cells; the circular RNA teams up with EZH2 in the nucleus to chemically silence CTNNBIP1; this releases the brakes on the Wnt/β‑catenin pathway, ultimately fueling faster tumor growth and spread. For patients, this suggests several new angles for therapy. Interrupting vesicle transfer, blocking circRNA‑0010220, interfering with its partnership with EZH2, or dampening β‑catenin signaling could all help slow osteosarcoma progression. While these ideas remain to be tested clinically, the study exposes a previously hidden conversation within the bone tumor environment and highlights a concrete set of molecular targets for future drugs.
Citation: Pan, R., Pan, Y., Ruan, W. et al. BMSC-derived extracellular vesicles enhance osteosarcoma proliferation and metastasis via the circRNA-0010220/β-catenin pathway. Cell Death Dis 17, 376 (2026). https://doi.org/10.1038/s41419-026-08655-8
Keywords: osteosarcoma, extracellular vesicles, circular RNA, Wnt beta-catenin, epigenetic regulation